- Submissions

Full Text
Novel Techniques in Nutrition and Food Science
Characteristic Analysis, Antioxidant Components, and Benefits of Pomegranate Peel and Red Onion Skin as Natural Antioxidants on Sunflower Oil Stability
Abd El-Rahman SN* and Sandak RN
Crops Technology Research Department, Food Technology Research Institute, Agricultural Research Center, Giza, Egypt
*Corresponding author:Soheir N Abd El-Rahman, Crops Technology Research Department, Food Technology Research Institute, Agricultural Research Center, Giza, Egypt
Submission: December 14, 2022;Published: January 09, 2023

ISSN:2640-9208Volume6 Issue5
Abstract
One of the most significant advancements in food technology recently is the management of natural antioxidants produced from plant waste to address issues with its sustainable production. The aim of the present study is to determine the chemical compositions, total tannin, phenolic, α-tocopherol, and antioxidant yields of red onion skin and pomegranate peel. Polyphenols compounds were identified by HPLC. Moreover, Sunflower oil was treated with aqueous and ethanolic extracts of red onion skin, and pomegranate peel as natural antioxidants at various concentrations compared with synthetic antioxidants (BHT and Catachin) at 80 °C for five hours each day for seven days. According to the findings, pomegranate peel and red onion skin had higher antioxidant yields from ethanol than from aqueous solution; also, pomegranate peel has a higher content of total tannin, and phenolic, while, red onion skin has a higher level of α-tocopherol. In comparison to these chemicals from red onion skin extract, the pomegranate peel extract utilizing HPLC contained the highest amounts of Syringic acid, Ferulic acid, Protocattechuic acid, Salicylic acid, and Resorcinol, respectively. On other hand, red onion skin extract contained the highest amounts of Coumarin, Caffic acid, P-Coumaric acid, Vanilne, Cinnamic acid, Quercetin, and Kaempferol. With time, lower acid values, peroxide values, and TBARS, also, higher iodine numbers were noticed than without it when sunflower oil was treated with increasing concentrations of extracts (ethanol and aqueous). The best results were noticed at 500ppm concentration. Pomegranate peel and red onion skin antioxidant extracts gave the best or the same effects recorded by synthetic antioxidants. It is therefore advised to use these edible extracts (natural antioxidants) extracted from onion skin and pomegranate peel instead of these toxic synthetic antioxidants.
Keywords:Red onion skin; Pomegranate peel; Antioxidants; Sunflower oil; HPLC; BHT; TBABS; Peroxide value
Abbreviations: BHT: Butylated Hydroxyl Toluene; TBARS: Thiobarbituric Reactive Substances; TBHQ: Tert-Butyl Hydroquinone
Introduction
The loss of a vital source of nutrients and phytochemicals makes food waste a serious issue for food processing businesses. The challenge of avoiding waste by turning processed food into usable items is brought on by rising customer demand for them [1]. One of the earliest consumed fruits, the pomegranate (Punica granatum) is a member of the Punicaceae family. Pomegranate peel has high concentrations of phenolics, flavonoids, complex polysaccharides, and minerals like potassium, calcium, phosphorus, magnesium, and salt [2]. Pomegranate fruit is recognized as a potent nutritional anticancer agent, according to numerous recent research [3]. According to Howell & D’Souza [4], pomegranate fruit (Punica granatum L.) has been found to have medicinal properties that include antioxidants, anti-carcinogens, antiinflammatory, antibacterial, and anti-parasite properties. When compared to other recognized antioxidants like ascorbic acid, vitamin E, and beta-carotene, polyphenols in pomegranate fruit have a wider variety of effects on numerous types of free radicals. Because of their antioxidant properties, phytochemicals (such as phenolic compounds, anthocyanins, tannins, and others) found in plants have medicinal effects [5]. Recently, there has been a lot of focus on the inclusion of polyphenols in meals, possibly because of their potent antioxidant properties (scavenger of free radicals). The pomegranate peel has the strongest antioxidant capacity, according to recent studies [6]. Onion is a crop that is grown all over the world for its medicinal and functional benefits in addition to being used for basic human consumption [7]. Due to behavioral changes that increased the demand for freshly cut and cooked veggies (onion), more waste has been produced globally, which has led to a 25% increase in onion output. According to Sagar et al. [8], the majority of onion processing waste is skin (peel), which consists of two outermost leathery layers, undersized roots, and malformed bulbs. is still an underutilized commodity while having the potential to be a source of biologically active substances like phenols and flavonoids [9]. Peel also has a variety of medicinal effects and has antioxidant, antibacterial, and antimutagenic properties [10]. The majority of the by-products produced during the processing of onions are not used, despite the fact that they are packed with phytochemicals and bioactive substances. Due to a rising interest in sustainable resource utilization and the circular economy, nutritional supplements and medicinal treatments can still be further reformulated even though the mechanism of action of the compounds contained in onions has been investigated. A wide range of pharmacological pathways are used by the different onion chemical components to prevent disease [11]. Since the onion skin is not edible, it is quickly removed before processing and selling, but due to its composition and smell, it cannot be used as animal feed or disposed of in a landfill. As a result, the onion processing process generates a significant amount of waste. In order to produce allium sustainably, onion peel must be turned into a “food by-product” by valorizing it. In the food, cosmetics, and pharmaceutical industries, by-products of food processing frequently contain useful compounds that can be employed as functional additives [12]. Therefore, the current research is carried out to shed light on the pomegranate peel and red onion skin chemical composition, identification of polyphenolic, quercetin, kaempferol, and ᾳ-tocopherol by HPLC, and utilization of these compounds to enhance the shelf life of the oil by way of adding natural antioxidants which extracted from pomegranate peel and red onion skin to the sunflower oil.
Materials and Methods
Materials
The pomegranate specimen needed for the planned study was bought from an Egyptian fruit store. The needed fruit rind was cut and removed from the pomegranate fruits after they had been easily peeled. The fruit rind was then dried in an oven at 40 °C for 24 hours. The fruit rind (pomegranate peel) was mechanically ground into a fine powder and stored at -18 °C until used [13]. The skin of a red onion Allium cepa L was obtained as a waste (by-product) from the Dehydro-Foods Manufactory for Onion Dried Production (Olam Food Manufactory Co.), Sixth Industrial Area of 6th October City, Giza Governorate, Egypt, and was then washed, hand peeled, naturally dried, as well as before being ground and sieved powder according to Marotti & Piccaglia [14]. El-Nasr Pharmaceutical Chemical, El-Ameriea, Cairo, Egypt provided the ethanol, as well as Butylated Hydroxyl Toluene (BHT), and catachin as synthetic antioxidants were bought from Sigma Chemical Co. in St. Louis, Missouri, the United States, including flavonoids and phenolic compounds. Sunflower oil was purchased from Tanta Company for Oils and Soaps in Tanta, Egypt.
Methods
Determination of the chemical analysis of raw materials: In the raw materials, proximate analysis including crude protein, crude lipids, ash, and crude fibers was determined in accordance with AOAC [15], total carbohydrates were calculated by deference.
Antioxidant extraction: Red onion skin and pomegranate peel that had been weighed out (10g each) were extracted for 24 hours with 50ml each of water, and ethanol. The antioxidant extracts were filtered and dried out at 40 °C in a rotary evaporator (RE 300/MS).
Determination of total phenolic, tannins, and α-tocopherol: Using the Folin-Ciocalteu reagent and the method outlined by Muscolo et al. [16], the total phenolic content of the raw materials was assessed. The results are reported as Gallic Acid Equivalents (GAE) mg/100g of dry weight. Additionally, the method of Çam & Hişil [17] was used to determine the total tannins content. Vitamin E (α-Tocopherol) was extracted separately from raw materials with 10mL of a 3:2 v/v hexane: isopropanol solution stirred for 5h and centrifuged at 3000rpm for 10 minutes. According to Prieto et al. [18], the supernatant was utilized to determine the amount of vitamin E.
Determination of antioxidants by HPLC: Polyphenolic compounds were determined by HPLC (HP 1050) according to the method described by Hajnos et al. [19].
Addition of Antioxidant to Sunflower Oil
Studies on oxidations were conducted using sunflower oil as a substrate. According to Buford [20], in order to examine the antioxidant potency of natural antioxidants extracted (aqueous and ethanolic) from pomegranate peel and red onion skin powder as well as synthetic antioxidants (BHT and catachin), antioxidants were added at concentrations of 100, 200, and 500ppm on a dry weight basis. The same procedures were used to prepare an additive-free control sample. In a 500ml glass beaker, sunflower oil with and without antioxidants (natural or synthetic) was heated intermittently for five hours per day for seven days at a temperature of 80±2 oC.
Determination of oxidative status in sunflower oil
The Thio Barbituric Reactive Substances (TBARS) were determined in oils. Malonaldehyde (MA) is an aldehyde produced during the breakdown of unsaturated fatty acids which gives a pink color and can be measured using spectrophotometers Jung et al. [21] and Zeb & Ullah [22]. The peroxide value, and the acid value as well as iodine number, was determined in accordance with AOAC [15].
Statistical analysis
The SPSS10 program was used to conduct statistical analysis. Data were presented as means and Standard Error of the Means (SEM), and statistical analysis was carried out using a one-way analysis of variance and Duncan’s tests [23].
Results and Discussion
Nutritional composition of raw material
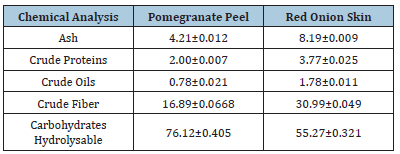
(Table 1) lists the chemical compositions of the raw materials. The analysis revealed that the ash, protein, oil, crude fiber content, and total carbs for pomegranate peels were 4.21, 2.00, 0.78, 16.89, and 76.12%, respectively, and for red onion skin they were 8.19, 3.77, 1.78, 30.99, and 55.27%. In comparison to pomegranate peel, the red onion skin was subsequently thought to be a better supplier of chemical components. [24] indicated that the protein level of pomegranate peel and onion skin was higher than that of garlic peel, although the latter’s carbohydrate amount (93.26%) was higher. [25] found that the best source of dietary fiber, polyphenols, and antioxidants is onion skin. In contrast, pomegranate fruit peel powder should be used to fortify foodstuffs. These findings concur with those of Essa & Mohamed [26], who discovered that pomegranate peels contain a healthy amount of crude fiber (12.17%).
Table 1:Chemical composition of pomegranate peel, and red onion skin on a dry weight basis. Values are mean±SD, n=3.
Yields of antioxidant extracts and phytochemical content in raw materials
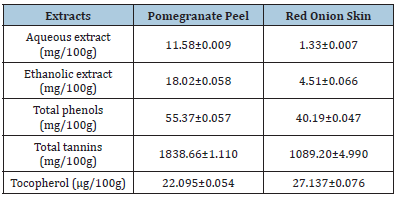
Pomegranate peel, and red onion skin yields in aqueous and ethanol extracts, as well as total phenols, tannins, and α-Tocopherol contents were determined. The results are shown in (Table 2). The results showed that the yield extract in pomegranate peel and red onion skin from ethanol was higher than aqueous, which may be because the solvent’s polarity was increased. According to the findings, pomegranate peel had a higher concentration of ethanol extract (18.02mg/100g) than red onion skin (4.51mg/100g). The ethanol extract from pomegranate peel may have produced a larger quantity of components with potent antioxidant activity than the water extract, which may account for these results. The choi of solvent is important in extraction operations since selectivity is directly impacted, which influences the chemical makeup and functional characteristics of the final extract. The desired compound’s solubility in the chosen solvent generally determines this choice. It is generally accepted that a polar solvent would encourage the solubilization and extraction of polar compounds, whilst a less polar solvent would fit for less polar molecules since solubilization involves electrostatic repulsions and attractions between the solvent and the solute [27]. The results from the same Table revealed that the pomegranate peel’s ethanol extract had the highest levels of total phenolic and total tannins at 55.37 and 1838.66mg/100g, respectively. Red onion skin’s phytochemical levels were reported to be 40.19 and 1089.20mg/100g, while vitamin E (α-Tocopherol) was higher at 27.137μg/100g in red onion skin than pomegranate peel was 22.095μg/100g, respectively. These findings demonstrated that when compared to red onion skin, which contained the highest levels of vitamin E, as well as the pomegranate peel enhanced the highest yield in phytochemicals, indicating that it contained a potent antioxidant. Pomegranate, or Punica granatum L, is a fruit that has long been used as a traditional micronutrient medicine and is a significant source of vitamins, phytochemicals, and bioactive components (such as phenolic acids, flavonoids, and tannins) [28]. In addition, the onion skin is also a rich source of total phenolics (9.4-52.7mg Gallic acid equivalent (GAE) /g dm) [11].
Table 2:Antioxidant of aqueous and ethanolic yields extract, total phenols, tannins, and α-tocopherol contents in pomegranate peel, and red onion skin. Values are mean±SD, n=3.
Identification of polyphenols for pomegranate peel, and red onion skin by HPLC
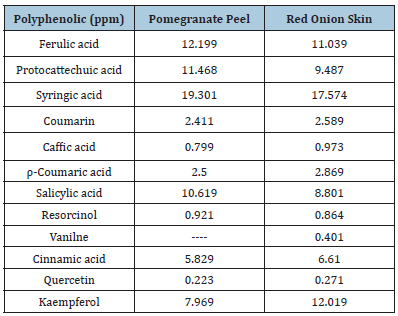
Using HPLC, the polyphenolic content of pomegranate peel and red onion skin extracts was determined. The results are shown in (Table 3). According to the findings, syringic acid, kaempferol, ferulic acid, protocatechuic acid, salicylic acid, and cinnamic acid were the polyphenolic components found in the red onion skin at the greatest concentrations (17.574, 12.019,11.039, 9.487, 8.801, and 6.610ppm, respectively). In terms of polyphenolic quantities, coumarin and p-coumaric acid were the two medium sources (2.589 and 2.869ppm). The lowest concentrations of polyphenolics were caffeine acid, resorcinol, vanillin, and quercetin, all of which were less than one. Onion plant skin is a biodegradable by-product.
By removing the dyestuffs from these wastes, it is feasible to assess their usage in the manufacturing of pigments and natural colors. There are some naturally occurring flavonoid (quercetin, for example) and anthocyanidin (pelargonidin, for example) colors in the skin that makes up this outermost section. The majority of the flavonols in plants are flavonoids called quercetin glycosides. From the interior to the exterior of the onion plant, the amount of quercetin may increase [29]. Syringic acid, ferulic acid, protocatechuic acid, Salicylic acid, kaempferol and cinnamic acid were the greatest concentrations of polyphenolics in the pomegranate peel extract, were present in concentrations of 19.301, 12.199, 11.468, 10.619, 7.969, and 5.829ppm, respectively. The moderate levels of polyphenolics were coumarin and p-coumaric acid (2.411 and 2.500ppm). The pomegranate peel extract had no Vanilne and contained the least quantities of Caffic acid, Resorcinol, and Quercetin of all the polyphenolics, this results agree with Soheir & Sara [30]. According to Saeed et al. [31], pomegranate peel (Punica granatum L.) extract by HPLC analysis revealed that the amount of ascorbic acid was higher than the phenolic acids (gallic acid and protocatechuic acid) and flavonoids (quercetin and kaempferol) in all extracts. The highest amount of ascorbic acid was discovered in the aqueous extract, followed by ethanolic. The amount of protocatechuic acid in the aqueous extract was larger than the number of other polyphenols, and the amount of quercetin was the lowest, despite the fact that phenolic acids were higher than flavonoids in all extracts. Gallic acid (3,4,5-trihydroxy benzoic acid), protocatechuic acid, and other phenolic acids are secondary metabolites that are naturally occurring molecules found in the plant kingdom with distinctive structural characteristics. Phenolic acids are one of the most prevalent classes of phytochemicals (2,4-dihydroxy benzoic acid). These substances are essential for growth and reproduction, protecting against infections, and sensory qualities may have a significant influence in determining the antioxidant capacity of meals, making them a possible natural source of antioxidants [32].
Table 3:Analysis of the HPLC polyphenols, quercetin,and kaempferol for pomegranate peel, and red onion skin.
Adding aqueous and ethanolic pomegranate peel, red onion skin extracts, catachin and BHT to sunflower oil
Table 4:Effect of natural (red onion skin and pomegranate peel) and synthetic antioxidants on acid value, Peroxide value, TBARS, and iodine number during heating sunflower oil at 80 °C/5hrs daily for 7 days. Values are mean±SD, n=3 control sunflower oil free antioxidants.
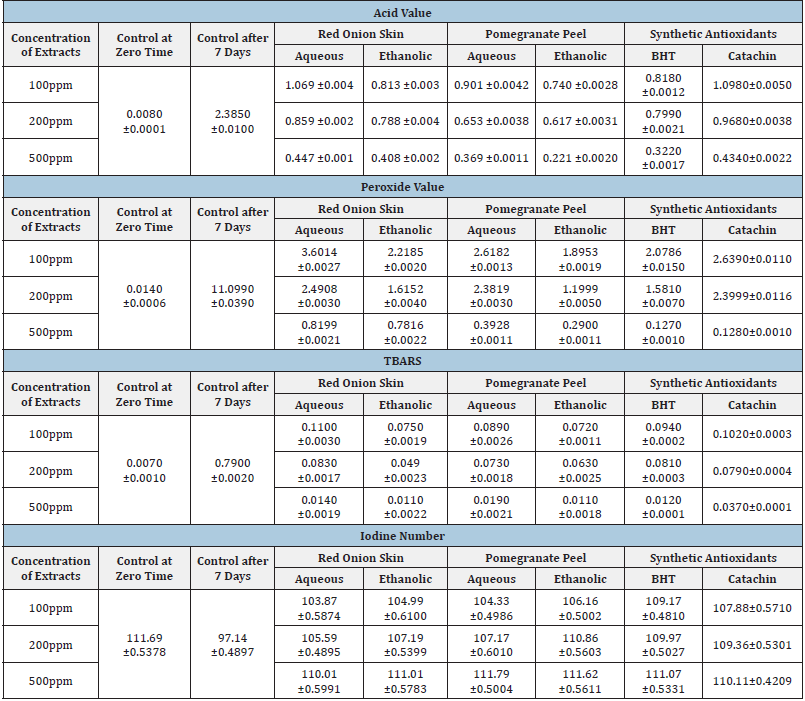
Sunflower oil was heated at 80 oC. for five hours each day for seven days with various concentrations (100, 200, and 500ppm) of pomegranate peel, and red onion skin extract, and compared to BHT and Catachin as synthetic antioxidants. The stability of all samples was assessed by measuring changes in acid value, peroxide value, Thiobarbituric Acid Reactive Substances (TBARS), and iodine number. (Table 4) demonstrated that pomegranate peel, red onion skin, and synthetic antioxidants had the best outcomes when sunflower oil was treated with extracts (ethanol and aqueous) at 500ppm concentration. With the passage of time, lower acid value, peroxide value, and TBARS were detected than without it, meanwhile, the iodine number increased gradually during seven days. This suggests that red onion skin, pomegranate peel extracts, and synthetic antioxidants like BHT and Catachin were all behaving as strong antioxidants. Additionally, there were noticeable changes in the three concentrations utilized in this study 100, 200, and 500ppm, thus the effect of natural and synthetic antioxidant extracts on the acid value, peroxide value, TBARS, and iodine number in sunflower oil depended on how much extract was added. In the presence of pomegranate peel, red onion skin, and synthetic antioxidants at a greater concentration of 500ppm, the autoxidation of sunflower oil was gradually suppressed. These results are consistent with data reported recently by Sarojini et al. [33] who found that Pomegranate Peel Extract (PPE) contains a considerable number of phenolic compounds that have the ability to scavenge free radicals during the oxidation process explaining the slow-down formation of peroxide in sardine fish oil compared to control. Antioxidants are considered a group of compounds that can counteract oxidation by acting as reducing agents, free radical scavengers, and quenchers of radical species, such as metals [34]. In comparison to synthetic antioxidants, the Peroxide Value (PV) and ThioBarbituric Acid (TBA) at 0.05% and 0.1% of pomegranate peel aqueous extract (POP-Ax) produced the best results [30]. Plants contain many natural antioxidant compounds, including polyphenols (anthocyanins, flavonols, and flavones), carotenoids, tannins, lignin, phenolic acids, and vitamins. However, these antioxidants can lose their activity under adverse conditions, such as high temperatures, extreme pH, and strong lights [35].
The antioxidant capacity of certain compounds can be measured directly or indirectly. The direct method determines the ability of antioxidants to intervene or stop the lipid oxidation process in meat homogenates, meat products, oil emulsion, and liposome systems in the presence of antioxidant compounds. Numerous studies have demonstrated that adding commercially available synthetic or natural antioxidants can slow down or prevent lipid and protein oxidation in processed beef products [36]. Because of their toxicity and potential health dangers, synthetic antioxidants including Butylated Hydroxyl Anisole (BHA), 2,6-di-tert-butyl- 4-methyl phenol (BHT), and Tert-Butyl Hydroquinone (TBHQ) have had their use in industrial processing prohibited. As a result, using naturally occurring compounds instead of manufactured additions is becoming more and more common. For instance, the food sector has made substantial use of tea polyphenols as antioxidants. It has also been demonstrated that their antioxidant impact is comparable to that of synthetic antioxidants. Many studies have shown that certain natural substances can effectively prevent the lipid oxidation of foods that are consumed for muscle growth [37]. Numerous natural substances, like ᾳ-tocopherol and certain polyphenols, have shown benefits in delaying protein oxidation during the processing of meat and meat products [38]. An antioxidant inhibitor is added to the system to stop or detect the chain of oxidation, stabilize the radical that is produced, and lessen the amount of oxidative damage that occurs in the body. Antioxidant molecules play a function in exerting a protective impact against oxidative damage by interacting with reactive radicals to create non-radical products inactively. The majority of these tests are carried out by accelerating the chain reaction’s induction phase by raising the temperature or adding more oxygen. A large number of pure chemicals’ and plant extracts’ antioxidative properties have been identified thanks to these experiments. These techniques for evaluating oxidation stability speed up oxidation under too severe circumstances and are too general and insensitive to be useful [39].
Conclusion
From the current investigation, it can be inferred that the skins of red onions and pomegranate peels may effectively stabilize sunflower oil at all concentrations. Pomegranate peel extract exhibits stabilizing effectiveness comparable to traditional synthetic antioxidants, such as BHT, and catachin at its legal limit, at a concentration of 500ppm. It increases sunflower oil’s resilience to thermally destructive changes. Pomegranate peel and red onion skin can be suggested as powerful sources of antioxidants for the stability of food systems, particularly vegetable oils.
Funding
No specific grant was provided for this research by funding institutions in the public, private, or not-for-profit sectors.
References
- Celano R, Docimo T, Piccinelli AL, Gazzerro P, Tucci M, et al. (2021) Onion peel: turning food waste into a resource. Antioxidants 10(2): 304-322.
- Jalal H, Pal MA, Ahmad SR, Rather M, Andrabi M, et al. (2018) Physico-chemical and functional properties of pomegranate peel and seed powder. The Pharma Innovation Journal 7(4): 1127-1131.
- Uddandrao VVS, Parim PRB, Nivedha K, Rameshreddy PS, Vadivukkarasi S, et al. (2019) Anticancer activity of pomegranate extract: effect on hematological and antioxidant profile against ehrlich-ascites-carcinoma in Swiss albino mice. Orient Pharm Exp Med 19(3): 243-50.
- Howell AB, D’Souza DH (2013) The pomegranate: Effects on bacteria and viruses that influence human health. Evid Based Complement Alternat Med 2013: 606212.
- Censits JH, Fu M (2013) Chemotherapy and targeted therapies: Are we making progress in castrate-resistant prostate cancer. Semin Oncol 40(3): 361-374.
- Ranjitha J, Bhuvaneshwari G, Jagadeesh SL (2018) Effect of different treatments on quality of nutri-enriched cookies fortified with pomegranate peel powder and defatted soybean flour. Int J Curr Microbiol App Sci 7(2): 3680-3688.
- Aslam R, Alam MS, Pandiselvam R (2022) Aqueous ozone sanitization system for fresh produce: Design, development, andoptimization of process parameters for minimally processed onion. Ozone Sci Eng 44: 3-16.
- Sagar NA, Pareek S, Aguilar GAG (2020) Quantification of flavonoids, total phenols and antioxidant properties of onion skin: A comparative study of fifteen Indian cultivars. J Food Sci Technol 57(7): 2423-2432.
- Kumar M, Barbhai MD, Hasan M, Punia S, Dhumal S, et al. (2022) Onion (Allium cepa L.) peels: A review on bioactive compounds and biomedical activities. Biomed Pharmacother 146: 112498.
- Kumari N, Kumar M, Lorenzo JM, Sharma D, Puri S, et al. (2022) Onion and garlic polysaccharides: A review on extraction, characterization, bioactivity, and modifications. Int J Biol Macromol 219: 1047-1061.
- Shabir I, Pandey VK, Dar AH, Pandiselvam R, Manzoor S, et al. (2022) Nutritional profile, phytochemical compounds, biological activities, and utilization of onion peel for food applications: A review. sustainability 14(19): 1-15.
- Gontard N, Sonesson U, Birkved M, Majone M, Bolzonella D, et al. (2018) A research challenge vision regarding management of agricultural waste in a circular bio-based economy. Critical Reviews in Environmental Science and Technology 48(6): 614-654.
- Dahham SS, Ali MN, Tabassum H, Khan M (2010) Studies on antibacterial and antifungal activity of pomegranate (Punica granatum L.). Am-Euras J Agric & Environ Sci 9(3): 273-281.
- Marotti M, Piccaglia R (2002) Characterization of flavonoids in different cultivars of onion (Allium cepa L). J Food Sci 67(3): 1229-1232.
- (2012) Official methods of analysis, (19th edn), Association of Official Analytical Chemists. Washington DC, US.
- Muscolo A, Papalia T, Settineri G, Mallamaci C, Panuccio MR (2019) Sulfur bentonite-organic-based fertilizers as tool for improving bio-compounds with antioxidant activities in red onion. J Sci Food Agric 100(2): 785-793.
- Çam M, Hışıl Y (2010) Pressurised water extraction of polyphenols from pomegranate peels. Food Chem 123(3): 878-885.
- Prieto P, Pineda M, Aguilar M (1999) Spectrophotometric quantitation of antioxidant capacity through the formation of aphosphomolybdenum complex: specific application to the determination of vitamin E. Anal Biochem 269(2): 337-341.
- Hajnos MW, Oniszczuk A, Szewczyk K, Wianowska D (2007) Effect of sample-preparationmethods on the HPLC quantitation of some phenolic acids in plant materials. Acta Chromatographica 19: 227-237.
- Buford R (1988) Extending shelf life by using traditional phenolic antioxidants. Cereal Food World 32(2): 207-212.
- Jung S, Nam KC, Jo C (2016) Detection of malondialdehyde in processed meat products without interference from the ingredients. Food Chem 209: 90-94.
- Zeb A, Ullah FA (2016) A simple spectrophotometric method for the determination of thiobarbituric acid reactive substances in fried fast foods. J Anal Methods Chem pp. 1-5.
- SPSS (2000) Statistical package for Social Sciences. SPSS for Windows, Version 10, SPSS Inc, Chicago, IL, USA.
- Ifesan BOT, Fadipe EA, Ifesan BT (2014) Investigation of antioxidant and antimicrobial properties of garlic peel extract (Allium sativum) and its use as the natural food additive in cooked beef. Journal of Scientific Research and Reports 3: 712-721.
- Benítez V, Mollá E, Cabrejas MAM, Aguilera Y, Andréu FJL, et al. (2011) Characterization of industrial onion wastes (Allium cepa L.): Dietary fiber and bioactive compounds. Plant Foods Hum Nutr 66(1): 48-57.
- Essa RY, Mohamed EE (2018) Improvement of functional and technological characteristics of spaghetti by the integration of pomegranate peel powder. American Journal of Food Technology 13(1): 1-7.
- Navarro MJ, Laguerre M, Tixier ASF, Tenon M, Feuillëre N, et al. (2018) What is the best ethanol-water ratio for the extraction of antioxidants from rosemary? Impact of the solvent on yield, composition, and activity of the extracts. Electrophoresis.
- Abdu OH, Saeed AA, Fdhel TA (2020) Polyphenols/flavonoids analysis and antimicrobial activity in pomegranate peel extracts. Electronic Journal of University of Aden for Basic and Applied Sciences 1(1): 14-19.
- Deveoglu O (2022) A review on onion skin, a natural dye source. J Text Color Polym Sci 19 (2): 307-319.
- Soheir NA, Sara A (2017) The preventive effect of pomegranate peel extract as a natural antioxidant on sunflower oil oxidative rancidity. IJFNPH 9(1): 45-54.
- Saeed AAM, Abdu OH, Salem TAF (2020) HPLC Analysis and DPPH assay of some bioactive compounds in pomegranate peel extracts. Research and Reviews: Journal of Medicinal Chemistry 2(1): 10-23.
- Grijalva EPG, Pére DLA, López NL, López RIC, Heredia JB (2016) Dietary phenolic compounds, health benefits and bioaccessibility. Arch Latinoam Nutr 66(2): 87-100.
- Sarojini A, Raju CV, Lakshmisha IP, Amitha, Gajendra (2019) Effect of pomegranate (Punica granatum) peel extract on lipid oxidation in sardine fish oil. Journal of Entomology and Zoology Studies 7(2): 140-144.
- Loi M, Paciolla C (2021) Plant antioxidants for food safety and quality: Exploring new trends in research. Antioxidants (Basel) 10(6): 972.
- Maqsood S, Benjakul S (2010) Comparative studies of four different phenolic compounds on in vitro antioxidative activity and the preventive effect on lipid oxidation of fish oil emulsion and fish mince. Food Chem 119(1): 123-132.
- Cheng JR, Xiang R, Tang DB, Zhu MJ, Liu XM (2021) Regulation of protein oxidation in Cantonese sausages by rutin, quercetin and caffeic acid. Meat Sci 175: 108422.
- Özünlü O, Ergezer H, Gökçe R (2018) Improving physicochemical, antioxidative and sensory quality of raw chicken meat by usingacorn extracts. LWT Food Sci Technol 98: 477-484.
- Khan IA, Xu WM, Wang DY, Yun A, Khan A, et al. (2020) Antioxidantpotential of chrysanthemum morifolium flower extract on lipid and protein oxidation in goat meat patties during refrigerated storage. J Food Sci 85(3): 618-627.
- Zrelli H, Matsuoka M, Kitazaki S, Zarrouk M, Miyazaki H (2011) Hydroxytyrosol reduces intracellular reactive oxygen species levels in vascular endothelial cells by upregulating catalase expression through the AMPK-FOXO3 a pathway. Eur J Pharmacol 660(2-3): 275-282.
© 2023 Abd El-Rahman SN. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






