- Submissions

Full Text
Novel Research in Sciences
Titanium Porous Plates for Electrolyzer- Series Production
Thorsten Hickmann*
Managing Director of Eisenhuth Gmbh & Co. KG, Germany
*Corresponding author:Thorsten Hickmann, Managing Director of Eisenhuth Gmbh & Co. KG, Germany
Submission: March 22, 2021;Published: April 15, 2021
.jpg)
Volume7 Issue2April, 2021
Mini Review
The discussion of renewable energy goes on, and Electrolyzers come more and more an important issue in this renewable world. Electrolyzers cells are basically galvanic cells converting chemical energy directly into electrical energy. Depending on the specific technology, this reaction may take place at elevated temperatures, thus technical requirements for materials and components of the stack become more and more challenging [1]. Porous titanium (sintered titanium, titanium foam) is widely used in many fields, such as pharmaceutical industry, water treatment industry, food industry, biological engineering, chemical industry purification, petrochemical industry, metallurgical industry and gas processing etc. Porous titanium can also be used as Gas Diffusion Layer (PDL) in new generation fuel cells (PEMFC). Manufacturing process for our porous titanium (sintered porous titanium). Raw titanium powder preparation-cold isostatic pressing-sintering at high temperature & to size-assembled vacuum-machined to filter if required Properties of porous titanium (sintered titanium) is however very cost-effective.
As an alternative, the material based on a titanium compound can be used easily. With this titanium compound it’s possible to produce a material with the following characteristics: [2]
a. uniform structure, narrow pore size distribution, high deposition efficiency.
b. High porosity, low filtration resistance, high penetration performance.
c. Can be high temperature resistance, usually sued up to 150 degrees.
d. good chemical stability, acid and alkali corrosion, antioxidant properties.
e. good mechanical properties, can be used in suction & pressure filtration.
Application of porous titanium resident on the above properties: [2]
i. Porous titanium sheet & porous stainless steel sheet can be used as Gas Diffusion
Layer (GDL) in polymer electrolyzes.
ii. Porous titanium sheets & tubes can be assembled as titanium filters, can be used for
filtration under high temperature & corrosive environment.
iii. Porous titanium can be coated with pint and others, which can be used as anode The
main product we provide for porous titanium is porous titanium & microporous titanium
sheet & disk and porous titanium tube. We provide porous titanium tubes manufactured
titanium filter assemblies. In this context porous titanium soften used.
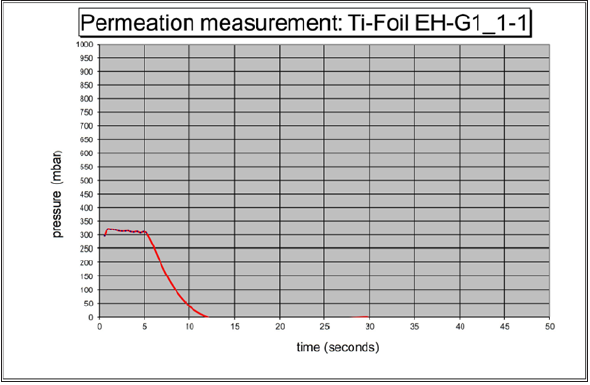
The suitable compounds are an essential starting material in electrochemical applications. These are plastics which are highly filled with titanium powder and thus exhibit electrically and thermally conductive properties. In the case of the thermoplastic method a thermoplastic binder with a defined degree of polymerization is compounded with the titanium powder material. The chemical structure of the polymer remains unchanged during compounding or molding process. The polymer has to be selected with sufficient chemical and thermal stability (e.g. data from [3]. Several material candidates are available on the market in high quality and well defined configurations. The porosity measurement device can be shown in Figure 1. Figure 1 shows that the special configurated measurement instrument. The measurement instrument works as follows: The Titanium-plate is put between 2 glass plates. In the 2 plates there is integrated a sensor on the one side. Then, a vacuum is applied. And on the other side the change of pressure is measured. As a next step, the plate is now measured. The results of this measurement can be seen in Figure 2. Figure 2 shows the results of the permeability of a porous plate. It takes approx. 10 second for the 500mbar pressure. So, it can be concluded that this plate is permeable.
Figure 1: Permeability measurement device of Titanium porous plate.

Figure 2:Permeability measurement device of Titanium porous plate.
However, the production of titanium-bipolar plates is quite challenging. When it comes to the processability of highly filled plastic compounds, a compromise must be found between good flowability on the one hand and special properties such as electrical conductivity on the other hand. A common way to develop novel material solutions for polymer composites characterized by high thermal and/or electrical conductivity is the use of conductive graphite up to 70-85wt.% filler content in composites for Titanium or alternatively Nickel powder. Such highly filled graphite-thermoplastic composites can be used as bipolar plates in electrolyzes [4]. For a comparison, the electrical conductivity=resistivity is measured. Here, the results can be seen in Figure 3. Figure 3 shows the electrical conductivity of the porous titanium compound plate. The actual value is approximately five times higher than pure titanium. However, an improvement work is going on. As a next step it is planned to change the ingredients of the raw material, so the permeability can be changed, and the electrical conductivity can be improved. The above-mentioned work is realized under a public funded project. The project is funded by the state of Lower Saxony. The realization of the funding is realized by the N-Bank under the funding code ZW 3 85116052, for which the project partner would like to thank them.
Figure 3:electrical resistivity of a compound plate and a pure titanium plate.

References
- Pitschak B, Mergel J, Muller M (2017) Electrolysis process. In: Töpler J & Lehmann J (Eds.), Hydrogen and Fuel Cell, Heidelberg, Germany.
- Chang L, Carmoa M, Bender G, Everwanda A, Lickert T, et al. (2018) Performance enhancement of PEM electrolyzers through iridium-coated titanium porous transport layers. Electrochemistry Communications 97: 96-99.
- Bonnet M (2013) Kunststofftechnik. Springer Verlag, Hamburg, Germany.
- Apelt S, Hickmann T, Marek A, Widdecke H (2006) How conductive compounds work in plastics. 12: 86-90.
© 2021 Thorsten Hickmann. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






