- Submissions

Full Text
Novel Research in Sciences
Nebulized Therapy with Plasma-Platelet Growth Factors in Control of Cytokin Storm Hyperinmune Phenomenon in Lungs of Patients Affected by Covid-19
J alcaraz* and T Holland
Medical advisory of Bridging Biosciences, USA
*Corresponding author: J alcaraz, Medical advisory of Bridging Biosciences, FRISCO, Tx, USA
Submission: April 07, 2020;Published: May 08, 2020
.jpg)
Volume3 Issue4May, 2020
Opinion
In Regenerative Medicine, application of Plasma Growth Factors (PRP) represents a great scientific advance that opens a new knowledge in the functioning of platelets and their function on the immune system, intervening not only in bactericidal processes, but in complex mechanisms to regulate processes related to inflammation and cellular immunity through a large number of cytokines and interleukins [1,2].
The scarcity of adequately structured and designed clinical trials together with a proliferation of processes for obtaining PRP not tested in the scientific literature, means that this Biotechnology has been relegated to the background in Cell Therapy, fostered by a lucrative desire fierce for the different clinical applications that it is intended to give; losing the true essence of it, which is none other than being an economic procedure, simple to carry out with adequate training and with a potential for application in different fields of medicine thanks to its direct effect on the mechanisms that regulate apoptosis or perpetuation in survival, differentiation and multiplication within the cell cycle and on the immune system.
On the other hand, today it is known that these proteins have an extraordinary diffusion capacity through the tissues, in such a way that can obtain maximum pharmacokinetic levels within 30-40 minutes of their local application and stable for 5-6 hours in tissue. This allows its administration by different routes: infiltration, intravenous infusion, in the form of eye drops or even NEBULIZED.
The existence of automated software for obtaining high concentration platelet plasma growth factors (between 1,500,000-3,000,000 platelets / mm3) could be a great scientific advance in having a useful, reliable and reproducible tool by any scientific working group to test its benefit for various clinical applications in the form of randomized clinical trials.
Unfortunately, these days we have had to experience the scourge of a pandemic caused by COVID-19 that is reaping tens of thousands of lives, especially those patients with a more compromised immune system, such as elderly patients or patients with various comorbidities, reaching figures of case fatality around 20% in these risk groups; and although every day we know more about its mechanisms of infection and symptom development, we currently do not have a specific treatment or vaccine that can confront it safely and efficiently [3,4].
It is known that an immunological phenomenon occurs between the 5th and 7th day of infection, which has become known as “CYTOKIN STORM”, which radically worsens the prognosis of patients, developing a phenomenon of lethal pneumonitis. in up to 35% of patients with pneumonia as a consequence of viral infection. This Fact is defined by the activation of the pulmonary monocyte-macrophage system, mediated by a series of interleukins, the best known being Interleukuna-6 (IL-6) [3,4].
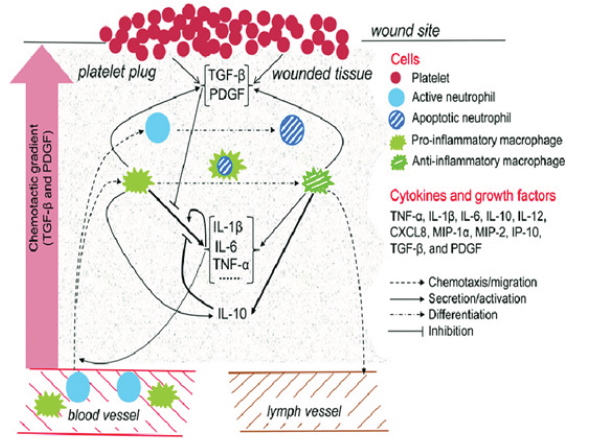
Until this moment, therapeutic options have been considered like immunomodulators-immunosuppressants, such as certain monoclonal antibodies or high doses of corticosteroids. From the point of view of regenerative medicine, the organism itself has means of regulating these phenomena of immune and inflammatory hyperreactivity. And at this point platelets have been shown to play a crucial role in controlling this inflammatory response in various murine and even human models. This explains that in histological samples of patients with bronchial and pulmonary hyperreactivity, a multitude of platelet aggregates has been observed in the affected areas; and recently it has been possible to objectify as two of the growth factors found in platelet alpha granules, known as Plateletderived growth factor (PDGF), as well as Transforming Factor alpha / beta (TGF a/b ), would improve the phagocytosis capacity of both dendritic cells and lung macrophages locally, inducing a suppression of IL-6 secretion, which would reduce the phenomena of inflammation, hemorrhage and necrosis at the lung tissue level (Figure 1). Similarly, it has been observed in vitro that approximate levels around 10 ng/ml of TGF (a/b) or 15 ng/ml of PDGF, would induce overexpression of IL-13, which in turn would decrease CD-4 + T lymphocytic population in lung owing to an apoptotic effect, favoring an attenuation of cellular immunosuppression.
Figure 1:
References
- Burnouf T, Chang CW, Kuo YP, Wu YW, Tseng YH, et al. (2011) A chromatographically purified human TGF‐beta1 fraction from virally inactivated platelet lysates. Vox Sang 101: 215‐ 220.
- Stiles CD (1983) The molecular biology of platelet-derived growth factor. Cell 33: 653-655.
- Alcaraz‐Rubio J, Oliver‐Iguacel A, Sánchez‐López J (2015) Plasma rico en factores de crecimiento plaquetario. Una nueva puerta a la Medicina regenerativa. Revista de Hematología 16( 2): 128-142.
- Alcaraz JR (2019) What Therapy with Plasma Growth Factors: Controversies of Key at Cellular Therapy. Haematol Int J 3(1): 000140.
© 2020 J alcaraz. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






