- Submissions

Full Text
Modern Concepts & Developments in Agronomy
Citric Acid Acidification of Wheat Straw Derived Biochar for Overcoming Phosphorus Deficiency in Soil
Adil Mihoub*
Biophysical Environment Station, Algeria
*Corresponding author: Adil Mihoub, Biophysical Environment Station, Touggourt 30240, Algeria
Submission: July 22, 2022;Published: September 15, 2022

ISSN 2637-7659Volume11 Issue 4
Abstract
Phosphorous (P) fixation in soils is a serious concern worldwide, and Biochar (B) is gaining attention daily due to its potential benefits for improving the agronomic benefits of applied P. The present study aims to enhance understanding of the P transformation process in a deprived sandy soil following biochar amendments (no-acidified Wheat Straw Biochar (WSB) and chemically modified (acidification with 0.01M C6H8O7 (AWSB)) along with or without P at 250mgkg-1.. A 54-day pot experiment was conducted with two bio-chars levels of 4% (B1), 8% (B2) w/w and control (CK, unamended soil), and two P-levels (without or with P). The results indicate that integration of AWSB with P resulted in increased available P in the soil. We conclude that incorporating acidified wheat straw biochar is a promising practice to potentially improve P availability in deprived soils. Further research is needed to explore site-specific P management for sustainable crop production.
Keywords: Acidified wheat straw biochar; P adsorption; P availability; Low P soil; Sustainable agriculture
Introduction
Phosphorus (P) is an irreplaceable element that has no substitute to sustain life on Earth [1]. Most importantly, it is obtained from limited phosphate rock resources [2]. In the soil, the available phosphorus of plants is relatively low because most of the phosphorus is usually combined with Calcium (Ca) and Magnesium (Mg) in calcareous soils and combined with Iron (Fe) and Aluminum (Al) in acid soils, Resulting in a decrease in the availability of phosphorus in this plant [3]. The fixed phosphorus in the soil is prone to runoff loss, leading to eutrophication of freshwater bodies [4].
Therefore, under acidic and alkaline soil conditions, a large amount of phosphate fertilizer (>90%) becomes unavailable, and the effectiveness of phosphorus in farmland systems becomes the primary limiting factor limiting phosphate fertilizer [5]. Therefore, exploring advanced phosphorus recycling technologies and management strategies is inevitable. These technologies and management strategies can provide phosphorus in the form available to plants and reduce the loss of available phosphorus to meet the increasing demand for phosphorus and food [6]. One option to promote phosphorus management in alkaline soils is to use biochar, a carbon-rich solid product at high temperatures, by exposing organic waste (such as wood chips, crop residues, or fertilizers) under anaerobic conditions [7]. Applying biochar to the soil can replace or partially reduce the use of inorganic phosphate fertilizers and may be one of the lowest costs, most efficient and most sustainable methods to save P resources and prevent P pollution [8].
In a recent Meta study with 108 pairwise comparisons of peer-reviewed studies on biochar effects on P availability in varying soils suggested that: P availability was increased by a factor of 5.1 and 2.4 is acidic (pH<6.5) and neutral (pH 6.5-7.5) soils, respectively and no significant effect was observed in alkaline soils [6]. Thus, the biochar amendment effects on P availability are inconsistent and regulated by changes in soil chemical properties, specifically soil pH. Recently, biochar feedstock/biochar modifications have been proposed to produce biochar with superior characteristics and improve its agronomic benefits [9,10]. However, chemical modifications using Citric Acid (CA) are not well studied. Therefore, improving our understanding of the potential effects of citric acid-modified biochar on P processes in soil and plant bioavailability in agricultural systems is mandatory.
The current study aimed to evaluate how biochar produced from wheat straw, and their chemical modification, treating with 0.01 N-Citric acid, and P application (control No P application or yes, P at 250mgkg-1 soil (in the form of KH2PO4.3H2O solution) affect P availability under alkaline soil condition.
Materials and Methods
Biochar production and properties
Figure 1:Scheme for the preparation of CA-modified biochar.
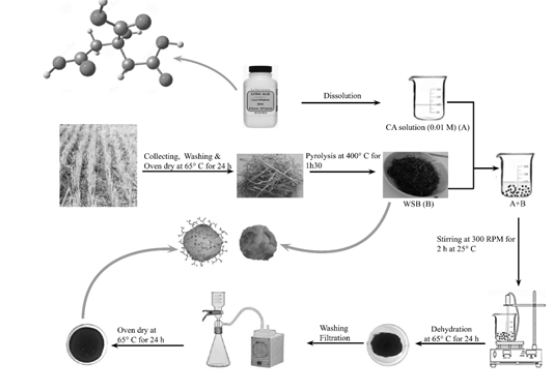
Wheat straw was crushed into powder and dried to constant weight at 65°C in a thermo-ventilated oven and then pyrolyzed in a muffle furnace at atmospheric pressure by applying 400°C for 1h30 under limited oxygen condition in a 250-c. The detailed preparation process of AWSB described above is shown in Figure 1. The resulting biochar materials were grounded and sieved through a 250μm square-mesh sieve prior to analysis. pH and Electrical Conductivity (EC) were measured in distilled water at 1:10 biochar to water mass ratio after shaking for 30 min according to ASTM D1762-84 2007 (Standard test method for chemical analysis of wood charcoal [11]. Biochar Organic Carbon Content (OC) was determined by the Walkley-Black method. For the determination of total P in biochars, the samples were digested in sulfuric acid. Later, the P concentration was measured following the method of [12] by using a using spectrophotometer at 882nm.
Incubation experiment
Table 1:Treatment details used for the experimentation.
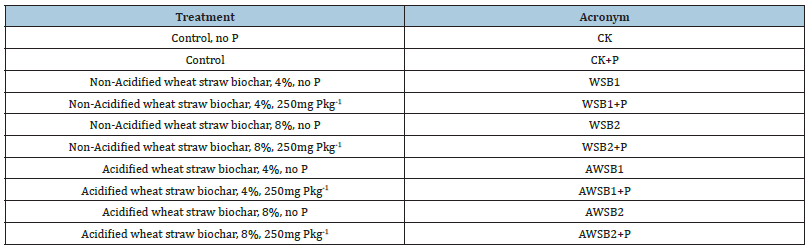
The top 0-30cm composite soil samples were collected from a deprived area. The soil was sandy, neutral, calcareous in nature (CaCO3~15%) and poor in organic matter as well as in available P. The incubation experiments included ten treatments, and each treatment was carried out with 100g portions of the air-dried soil packed uniformly in plastic cups. In eight treatments, each soil was amended with one of biochar’s produced: acidified or no-acidified biochar at rates of 4%, and 8 % (w/w) of initial soil in dry weight, with or without addition of KH2PO4 at a P rate of 250mgkg-1 soil. Two controls without biochar input, one with the unfertilized soil and the other with only KH2PO4, were included. The different treatments used were as follows (Table 1). In the experimentation, two replicates of each treatment were prepared, randomly placed and incubated in the laboratory at ambient temperature 25± 2°C and 80% soil moisture (v/w) for 9, 18, 27, 36, 45 and 54 days. At the end of each incubation period, samples (~20g) were removed from each plastic cup and analyzed for available P (Olsen-P).
Data analysis
Average and stranded deviation were calculated. Statistical analysis was performed using Stat-soft Statistica Software, 10th Edition (Stat-soft, Tulsa OK). In addition, Duncan’s multiple range test, which combines means of similar values into ordered homogenous groups, was applied.
Results and Discussion
The wheat straw that was charred at 400 °C yielded an alkaline biochar (pH= 8.9), rich in OC (55.4 %), moderately low in available P (9mgkg-1) and total P (68.64mgkg-1). However, a higher EC value was found (26.12dSm-1), indicating the existence of water-soluble salts. In this experiment, the CA modification/ amelioration of biochar focused on enhancing P availability and its half-life in low-P soils. It is clearly demonstrated that application of 4-8% biochar significantly increased P availability in studied soil. All biochar treatments individually or in combination with or without phosphorus input yielded a significant increase in the available P in studied sandy soil. When compared with the control, content of available P in soil samples increased significantly (p<0.001) after amendment with WSB compared with the controls without biochar amendment (Table 2).
Incubation experiment
Table 2:Evolution of soil available P of different treatments during the incubation time.
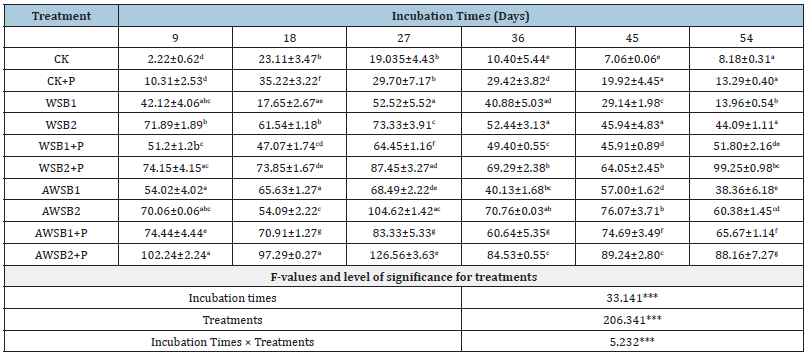
Values represent mean of two replicates ± SD. Values followed by different letters are significantly differences among each other as determined by the DMR test (P<0.05). ***Stands significant at P<0.001.
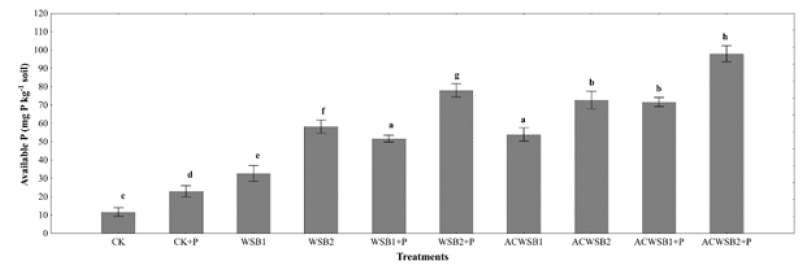
The addition of biochar alone without P amendment increases the availability of P in soil. Our results are similar to previous studies which indicated that the addition of biochar increases the availability of P even without adding P [13]. In addition, the amount of soil available P in AWSB1 and AWSB2 amended treatments is higher compared to the control (without biochar input) and even compared with WSB treatments (Figure 2). This could probably be attributed to the acidification of soil/biochar by citric acid which can compete strongly with P for adsorption sites by blocking phosphate sorption sites on soil thereby reducing phosphate adsorption [14]. Organic acids have been reported to strongly enhance the mobilization of P from soils [15,16]. The low pH of the soil plays a vital role in the dissolution of immobilized soil P [17]. Addition of organic acids to the soil caused an immediate pH drop and the extent of pH decrease depended on both soil type and organic compound [18,14]. According to Hopkins et al. [19], the decrease of soil pH (6.5) will increase the mobility of fixed soil P. In the case of low soil pH, the activity of H+ will decompose the bond of calcium and phosphorus in the calcareous soil. Breaking of the bond between calcium and phosphorus leads to the dissolution and availability of phosphorus in the soil [20]. In addition, P is also part of the biochar structure [21]. As observed in the AWSB of this study, lowering the pH of biochar also increases the release of phosphorus from the biochar into the soil solution [22]. Biochar improves P availability in calcareous soils mainly due to changes in soil microbial community [23,24]. Indirect application of biochar can also promote the secretion of phosphomonoesterase by soil microorganisms, thereby enhancing the mineralization of soil (Paz-Ferreiro et al., 2012). According to Laird [25], the application of biochar can increase the Cation Exchange Capacity (CEC) of the soil by 20%. The CEC is an indirect measure that can increase the retention rate of water and nutrients by reducing its leaching loss. The modification of various bio-sorbents (such as biochar) by citric acid may change its quality by introducing additional carboxyl groups on its surface Wang et al., 2014 & [26], thereby increasing the availability of soil and nutrients availability [10].
Biochar production and properties
Figure 2:Effect of wheat straw biochar acidification (yes/no) and phosphorous application (no application or 250mgPkg-1 soil) on P availability under alkaline condition. Bars represent means ± S.E and values followed by different letters are significantly differences among each other as determined by the Duncan’s test (P<0.05).
Conclusion
Results of the incubation experiment showed that the acidification of biochar exerted a positive effect by enhancing the availability of soil P. A greater Olsen’s P availability was obtained with acidified bio-chars instead of non-acidified ones. The capacity for a reduction in P adsorption and increase in P availability appears to follow the order of AWSB treatments > WSB treatments > control treatments. This indicates that treating biochar with an acidifying agent such as citric acid is effective enough to reduce the rate of orthophosphate conversion to unavailable form. However, these findings need to be further confirmed by field experiments over the long term, considering the possible adverse effects of biochar application at high rates.
References
- Rafique M, Ortas I, Ahmed IA, Rizwan M, Afridi MS, et al. (2019) Potential impact of biochar types and microbial inoculants on growth of onion plant in differently textured and phosphorus limited soils. J Environ Manag 247: 672e680.
- Chowdhury RB, Moore GA, Weatherley AJ, Arora M (2017) Key sustainability challenges for the global phosphorus resource, their implications for global food security, and options for mitigation. J Clean Prod 140(2): 945-963.
- Gyaneshwar P, Kumar GN, Parekh LJ, Poole PS (2002) Role of soil microorganisms in improving P nutrition of plants. In: Adu-Gyamfi JJ (Ed.), Food Security in Nutrient-Stressed Environments: Exploiting Plants’ Genetic Capabilities. Springer, Netherlands, pp. 133-143.
- Siddique MT, Robinson JS (2003) Phosphorus sorption and availability in soils amended with animal manures and sewage sludge. J Environ Qual 32(3): 1114.
- Rafique M, Ortas I, Rizwan M, Chaudhary HJ, Gurmani AR, et al. (2020) Residual effects of biochar and phosphorus on growth and nutrient accumulation by maize (Zea mays) amended with microbes in texturally different soils. Chemosphere 238: 124710.
- Glaser B, Lehr VI (2019) Biochar effects on phosphorus availability in agricultural soils: a meta-analysis. Sci Rep 9: 1e9.
- Qayyum MF, Liaquat F, Rehman RA, Gul M, ul Hye MZ, et al. (2017) Effects of co-composting of farm manure and biochar on plant growth and carbon mineralization in an alkaline soil. Environ Sci Pollut Res 24(33): 26060-26068.
- Raj SN, Shetty HS, Reddy MS (2006) Plant growth promoting rhizobacteria: potential green alternative for plant productivity. In: Siddiqui ZA (Ed.), PGPR: Biocontrol and Biofertilization. Springer, Netherlands, pp. 197-216.
- Takaya CA, Fletcher LA, Singh S, Okwuosa UC, Ross AB (2016) Recovery of phosphate with chemically modified biochars. Journal of Environmental Chemical Engineering 4(1): 1156-1165.
- Ding Y, Liu Y, Liu S, Li Z, Tan X, et al. (2016) Biochar to improve soil fertility. A review. Agronomy for Sustainable Development 36: 1-18.
- ASTM standard (2009) Standard test method for chemical analysis of wood charcoal. American Society for Testing and Materials (ASTM) International: Conshohocken, Pennsylvania USA.
- Shaheen SM, Tsadilas CD, Eskridge KM (2009) Effect if common ions on phosphorus sorption and availability in Greek alfisols with different pH. Soil Science 174(1): 21-26.
- Deluca TH, Gundale MJ, Mackenzie MD, Jones DL (2015) Biochar effects on soil nutrient transformations. Biochar for environmental management: science, technology and implementation 2: 421-454.
- Mihoub A, Bouhoun MD, Naeem A (2018) Short-term effects of phosphate fertilizer enriched with low molecular weight organic acids on phosphorus release kinetic and availability under calcareous conditions in arid region. Journal of Scientific Agriculture 2: 66-70.
- Gerke J (1994) Kinetics of soil phosphate desorption as affected by citric acid. Zeitschrift für Pflanzenernährung und Bodenkunde 157(1):17-22.
- Mihoub A, Daddi Bouhoun M, Naeem A, Saker ML (2017) Low-molecular weight organic acids improve plant availability of phosphorus in different textured calcareous soils. Archives of Agronomy and Soil Science 63(7): 1023-1034.
- Sultan H, Ahmed N, Mubashir M, Danish S (2020) Chemical production of acidified activated carbon and its influences on soil fertility comparative to thermo-pyrolyzed biochar. Scientific Reports 10: 595.
- Zhi-An Li, Bi Zou, Han-Ping Xia, Yong-Zhen Ding, Wan-Neng Tan, et al. (2008) Role of low-molecule-weight organic acids and their salts in regulating soil pH. Pedosphere 18(2): 137-148.
- Hopkins B, Ellsworth J (2005) Phosphorus availability with alkaline/calcareous soil. In Western Nutrient Management Conference 6: 88-93.
- Dorozhkin SV (2012) Dissolution mechanism of calcium apatites in acids: A review of literature. World Journal of Methodology 2(1): 1-17.
- Danish S, Younis U, Akhtar N, Ameer A, Ijaz M, et al. (2015) Phosphorus solubilizing bacteria and rice straw biochar consequence on maize pigments synthesis. International Journal of Biosciences 5(12): 31-39.
- Silber A, Levkovitch I, Graber ER (2010) pH-dependent mineral release and surface properties of cornstraw biochar: agronomic implications. Environmental Science & Technology 44(24): 9318-9323.
- Deb D, Kloft M, Lässi, J, Walsh S (2016) Variable effects of biochar and P solubilizing microbes on crop productivity in different soil conditions. Agroecology and Sustainable Food Systems 40(2): 145-168.
- Amin Aeeaz, Mihoub A (2021) Effect of sulfur-enriched biochar in combination with sulfur-oxidizing bacterium (Thiobacillus Spp.) on release and distribution of phosphorus in high calcareous p-fixing soils. Journal of Soil Science and Plant Nutrition 21(3): 2041-2047.
- Laird DA, Fleming P, Davis DD, Horton R, Wang B, et al. (2010) Impact of biochar amendments on the quality of a typical Midwestern agricultural soil. Geoderma 158(3-4): 443-449.
- Zhou Y, Gu X, Zhang R, Lu J (2014) Removal of aniline from aqueous solution using pine sawdust modified with citric acid and β-cyclodextrin. Industrial and Engineering Chemistry Research 53(2): 887-894.
© 2022 Adil Mihoub. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






