- Submissions

Full Text
Modern Concepts & Developments in Agronomy
Biosorption of Chromium (VI) in Aqueous Solution by Ananas comosus Biomass Shell
Acosta Rodríguez I1, Cárdenas González JF2*, Galaviz Orocio K1, Rodríguez Pérez A2, Michel Cuello C2 and Muñoz Morales A2
Bulgaria
*Corresponding author: Cárdenas González JF, Unidad Académica Multidisciplinaria Zona Media, México
Submission: April 28, 2019;Published: May 10, 2019

ISSN 2637-7659Volume4 Issue2
Abstract
Effluents from tanneries are one of the main sources of contamination with Chromium (VI) of water and soils. This metal is used in tanning leather and skins, steel alloys, electroplating, textile staining and as a biocide in water cooling systems in nuclear plants, discharging the metal to the environment representing a great risk to the health of living organisms, so in this work, we studied the Chromium (VI) removal in aqueous solution by the Ananas comosus biomass shell, by the Diphenylcarbazide method to evaluate the metal concentration in solution. Biosorption at different pH (1, 2, 3 and 4) was evaluate for different times. We too studied the effect of temperature in the range of 28 to 60 °C and the removal at different initial concentrations of Chromium (VI) of 200 to 1000mg/L. Therefore, the highest biosorption of the metal (100mg/L) occurs within 10 hours, at pH of 1.0, 28 °C with 5g of natural biomass. According to temperature, the highest removal was observing at 60 °C, in 3 hours, when the metal is completely adsorbed. At the analyzed concentrations of Cr (VI), fungal biomass, showed excellent removal capacity at 60 °C, it removal 1g/L of the metal in 45 minutes, besides it removal efficiently the metal in situ (100% and 97.2% removal in 100mL of water and 10g of earth contaminated, after 9 and 10 days of incubation, with 10g of biomass; so, it can be used to eliminate it from industrial wastewater.
Keywords:Biosorption; Chromium (VI); Pineapple; Biomass; Bioremediation
Introduction
Currently, one of the main environmental problems is heavy metal pollution. These elements alter the equilibrium of ecosystems by persisting indefinitely in the environment, because they are not degrading by biological or chemical means [1]. Its accumulation in the organisms of the different links of the trophic chain, its mobility in natural aquatic ecosystems and its toxicity, make its elimination a global concern [2]. The introduction and redistribution of metal ions in the biosphere have their origin from natural and anthropogenic sources [3]. However, the main direct as well as indirect cause of the metal contamination are the urban sources, being the industrial operations with a deficient or absent treatment of its residual waters and solid waste, its main emitter. Some of the industries that generate waste contaminated with these elements are sugar, oil, brewing, textiles, cellulose and paper, metal finishing, copper and its alloys, tannery, food and of iron and steel, legally classified as point sources of pollution [2]. Hexavalent chromium [Cr (VI)] is an important water pollutant. Even at metals levels measuring in the parts per billion (ppb), research has shown it to be toxic [4]. Cr (VI) can originate from different anthropogenic activities such as chromite mining, leather tanning, pigment synthesis, electroplating and metal finishing [4]. The primary forms of chromium found in nature are Cr (III) and Cr (VI) and these forms are converting to each other depending on environmental conditions [5]. Cr (VI) is consider the most toxic form of chromium and is usually associated with oxygen as chromates (CrO4-2) and dichromate (Cr2O7-2) [4]. It has been established now that various chromium compounds as oxides, chromates and dichromate, are environmental contaminants in water, soil, and industrial effluents, because this metal is widely used in various manufacturing, such as electrolytic plating, explosives manufacturing, leather tanning, metal alloy, dyes and pigments manufacturing, etc., [2,3&5].
There are studies of the current technologies that are being used to effectively reduce Cr (VI) present in aqueous solutions by means of chemical, electrochemical and biological methods, for example: ion exchange on resins, coagulation-flocculation, adsorption on activated carbon, reduction, chemical precipitation, sedimentation, etc., [6], which in most cases are expensive or inefficient, especially when the concentration of these ions is very low [7]. Therefore, arise emerging technologies such as biosorption, the process of attracting various chemical species by biomass (live or dead), by physicochemical mechanisms as adsorption or ion exchange [8]. Recently, varieties of low-cost materials have been study for their ability to remove Cr (VI) from aqueous solution and promising results are shown. Among these low-cost adsorbents are dead microorganisms, clay minerals, agricultural wastes, industrial wastes and various other low-cost materials [4,6-9]. Thus, there is a need to develop or find innovative low-cost adsorbents with an affinity towards metal ions for the removal of Cr (VI) from aqueous solution, which leads to high adsorption capacity [4,6-8]. The objective of this study was to analyze in vitro biosorption of Cr (VI) by pineapple (Ananas comosus) biomass shell.
Materials and Methods
Preparation of the bio sorbent
The Ananas comosus shell, was obtained from the fruits harvested and offered in the market place Republic, between the months of July to August in 2017, of the capital city of San Luis Potosí, S.L.P. México. To obtain the biomass, the shells was washed with trideionized water during 72 hours at constant stirring, with water changes every 12 hours. Subsequently, it was boiling 1 hour to removal traces of the fruit and dust and were dry at 80 °C for 12 hours in an oven, ground in blender and stored in amber vials until use.
Biosorption studies and determination of hexavalent, trivalent and total Cr
For these studies, was used 5g of dried biomass mixed with 100mL of trideionized water containing 100mg/L of the metal, in an Erlenmeyer flask at the desired temperature and pH. The flasks were agitated on a shaking bath Yamato BT-25 model at different times. Samples of 5mL were taken at different times and centrifuged at 3000rpm for 5min. The supernatant liquid was separated and analyzed for Cr (VI) ions. Hexavalent Chromium and trivalent Chromium were quantifying by a Spectrophotometric method employing Diphenylcarbazide and chromazurol S, respectively [10,11], total Chromium was determining by electrothermal atomic absorption spectroscopy [11]. The information shown in the results section are the mean from three experiments carried out by triplicate.
Results and Discussion
Effect of incubation time and pH
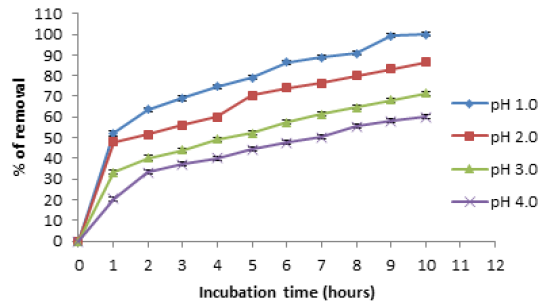
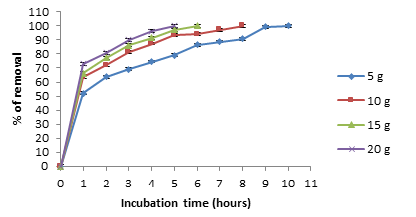
The optimum time and pH for Cr (VI) removal for pineapple biomass, was 10 hours and pH 1.0, at constant values of biosorbent dosage (5g/100mL), with an initial metal concentration of 100mg/L, and a temperature of 28 °C (Figure 1). It was used a pH meter Corning Pinnacle 530 model and we use nitric acid 1M to maintain the pH. The literature [12,13], report an optimum time of 2 and 4 hours for the removal of Cr (VI) by Macadamia nutshell powder [12]and Amarantus caudatus [13], with 2.5g/L and 5g/100mL, of natural biomass respectively, 24 hours for the avocado shell, with 102mg/L and 1g/L of Chromium (VI) [14], 5 minutes for almond green hull, with a concentration of 10mg/L of the metal and 4g of biomass [15], and 9 hours for sawdust of pine tree [16], with an initial metal concentration of 50mg/L. Changes in the cell permeability of unknown origin, could partly explain the differences founded in the incubation time, providing greater or lesser exposure of the functional groups of the cell wall of the biomass analyzed [1,3, 6-8]. Adsorption efficiency of Cr (VI) was observe a maximum at pH 1.0 with the biomass analyzed, and this is like to most reports [1,13-16], but some report other pH´s, like for avocado shell [14], and almond green hull [15], which report an optimum pH of 2.0. This was due to the dominant species (CrO42- and Cr2O72-) of Cr ions in solution, which were expected to interact more strongly with the ligands carrying positive charges [1,7,8].
Figure 1:Effect of incubation time and pH on Chromium (VI) removal by the biomass of A. comosus shell. 100mg/L Cr (VI), 100rpm, 28 °C. 5g of biomass.
Effect of the temperature
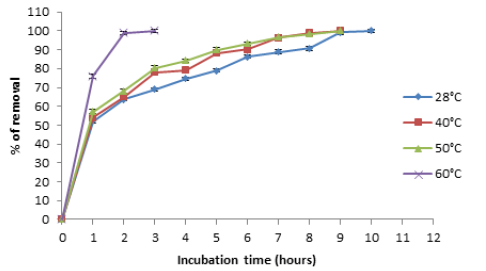
Temperature was found to be a critical parameter in the bio adsorption of Cr (VI) (Figure 2). To maintain constant the temperature in all experiments, we use a shaking bath Yamato BT- 25 model. The total removal was observed at 60 °C and 28 °C with 3 hours and 24 hours of incubation. This results are coincident for tamarind shell with 95% of removal at 58 °C and 3 hours [16], for the adsorption of cadmium (II) from aqueous solution on natural and oxidized corncob (40 °C and 5 days) [17], but these are different for the mandarin waste [18], Caladium bicolor (wild cocoyam) biomass [19], and Saccharomyces cerevisiae [20]. The increase in temperature increases the rate of removal of Cr (VI) and decrease the contact time required for complete removal of the metal, to increase the redox reaction rate [16].
Figure 2:Effect of the temperature on Chromium (VI) removal by the biomass of A. comosus shell. 100mg/L Cr (VI), 100rpm, pH 1.0. 5g of biomass.
Effect of the initial metal concentration
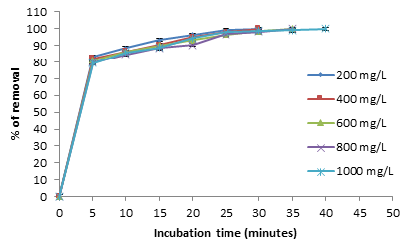
At low metal concentrations (200mg/L) a 28 °C, we observe the best results for removal, with the biomass analyzed, because the removal of the metal was 100% and 48%, at 24 hours for 200 and 1000mg/L, respectively (Figure 3). The results are coincident for A caudatus [13], with a total removal of the metal (200mg/L) at 10 hours, for almond green hull with 50mg/L of the metal and different amounts of sorbent (13,15,18 and 20g) at pH of 6.0 after 40 minutes of incubation [15], for modified Russian knapweed flower powder with increasing initial concentration of Cr (VI) solution from 2 to 20mg/L, chromium removal reduced, with a removal efficiency for 2 and 20mg/L were 99.85% and 39.41%, respectively [21], and in Vetiveria zizanioides, varying the concentration of Cr (VI) from 1 to 5mg/L in synthetic solution, pH 1.0, with stirring rate of 150rpm at room temperature and contact time of 4 and 5h for grass and roots, respectively. It can be concluded from that removal efficiency decreases with increase in initial concentration of Cr (VI) in synthetic solution [22]. This was due to the increase in the number of ions competing for the available functional groups on the surface of biomass [1,4]. On the other hand, at 60 °C, the biomass studied, shows the same results for removal, adsorbing 100% between 30 and 35 minutes with 200 and 1000mg/L of metal, respectively (Figure 4) with respect to other biomasses, this results are like for Penicillium sp., IA-01 [23], but there are other studies, which report a major biosorption with low concentrations: Citrus reticulata and Tamarindus indica shell [24,16]. In addition, we observe the development of a blue-green and white precipitate, which changes more rapidly at higher temperatures (date not shown).
Figure 3:Effect of the initial concentration of the metal on Chromium (VI) removal by the biomass of A. comosus shell. 100rpm, pH 1.0. 5g of biomass. 28 °C.
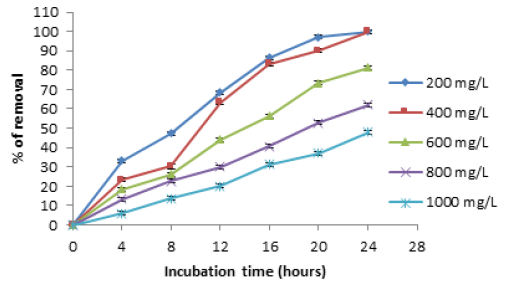
Figure 4:Effect of the initial concentration of the metal on Chromium (VI) removal by the biomass of A. comosus shell. 100rpm, pH 1.0. 5g of biomass. 60 °C.
Effect of the biosorbent dose
The influence of biomass concentration on the removal capacity of Cr (VI) is depict in Figure 5. If we increase, the amount of biomass also increases the removal of the metal in solution, with more biosorption sites of the same, because the amount of added biosorbent determines the number of binding sites available for metal biosorption [25-27]. For Almond green hull, with a concentration of 10mg/L of the metal with 0.5, 1, 1.5, 2, 2.5, 3, 3.5, 4, 4.5, and 5g of biomass [15]. Too, the removal efficiency using oleaster (Elaeagnus) seed and cherry (Prunus avium) stone biochar, increased with the increase of the biochar mass for a certain Cr(VI) concentration [22], for modified Russian knapweed flower powder at the initial chromium concentrations of 2, 10 and 15mg/L at pH 2, and the results showed that chromium removal at an equilibrium condition increased with increasing adsorbent dose [22].
Figure 5:Effect of biomass concentration of A. comosus on the removal of 100mg/L Cr (VI), 28 °C, pH 1.0, 100rpm.
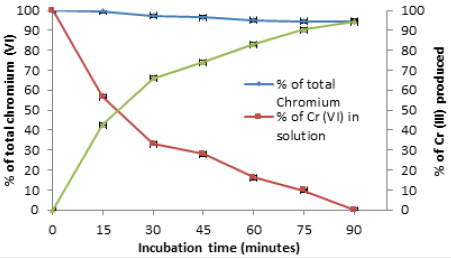
Time course of Chromium (VI) decrease and Chromium (III) production
We analyzed the ability of A. comosus biomass to reduce 1.0g/L of Cr (VI) and Cr (III) production in solution. The biomass exhibited a great efficiency to diminish metal level with the concomitant production of Cr (III) in the solution, as at 90min of incubation, we observe a drop in metal initial concentration to almost undetectable levels, without change significant in total Chromium concentration, which remained constant over time, in solution control, probably due to the vitamin C content in the shell (Figure 6) [28].
Figure 6:Time-course of Cr (VI) decrease and Cr (III) production in solution with 1.0g/L Cr (VI). 28 °C, pH 1.0, 100rpm.
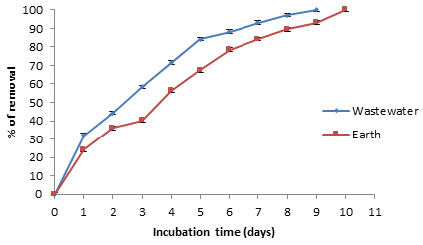
Removal of Chromium (VI) in industrial wastes with A. comosus biomass
Finally, we analyzed the possible usefulness of this biomass for removal this metal from industrial wastes. The biomass (5g) was incubate with non-sterilized contaminated earth and water containing 100mg Cr (VI)/g, resuspended in trideionized water to a final volume of 100mL. It was observing that in 9 and 10 days of incubation, the Cr (VI) concentration of water and earth samples, decrease fully in water and 97.2% in earth (Figure 7), without change significant in total Chromium content during the experiments. In the experiment control (without biomass), the metal concentration of the water and earth samples decreased by about of 6% and 18% (date not shown); which can be caused by indigenous microflora and (or) reducing components present in the samples [1,4,8&29]. The chromium removal abilities of this biomass are equal or better than other biomasses reported, for example A. caudatus [13], Melaleuca diosmifolia leaf [28], T. indica, M. paradisiaca, C. limonium, and C. sinensis shells [30], maize leaf [31], C. melo shell [9], sawdust of pine tree [16], M. indica, M. paradisiaca, C. paradise, C. melo y C. máxima shells [24].
Figure 7:Removal of Cr (VI) in industrial wastes incubated with 5g of A. comosus biomass. 28 °C, 100rpm, 100mL of contaminated earth and water, (100mg Cr (VI)/g earth and 100mg/L of water) (adjusted).
Conclusion
In this work, the biomass analyzed, showed complete capacity of biosorption of 100mg/L of Cr (VI) in solution at 10 hours at pH 1.0 with 5g of natural biomass, and at 60 °C, in 3 hours the metal was fully adsorbed, besides this biomass, removal the metal in wastewater with 5 of biomass in water and earth (both with 100mg/g of earth, and 100mg/L of water) decrease 100% and 97.2%, respectively. These results suggest the potential applicability of this natural biomass for the remediation of Cr (VI) from polluted water and earth.
Acknowledgement
All the authors are grateful for the cooperation of the Research and Linking Center “El Balandran”, which is an extension of the Multidisciplinary Academic Unit of the UASLP, for the elaboration of some experiments in its facilities. In the same way the authors thank the Faculty of Chemical Sciences of the UASLP for the use of their facilities for the elaboration of some experiments. The authors declare that there is no conflict of interest and that the project was not financed by any instance, it was carried out with their own means.
References
- Ahemad M (2014) Bacterial mechanisms for Cr (VI) resistance and reduction: an overview and recent advances. Folia Microbiologica 59(4): 321-332.
- Chowdhury S, Jafar MA, Al Atta O, Husain T (2016) Heavy metals in drinking water: Occurrences, implications and future needs in developing countries. Sci Total Environ 569-570: 476-488.
- Pérez Bou L, Salgado Bernal I, Larrea Duarte C, Martínez Sardiñas A, Cruz Arias ME, et al. (2018) Biosorción microbiana de metales pesados: características del proceso. Revista Cubana de Ciencias Biologicas 6(1): 1-12.
- Pradhan D, Sukla LB, Sawyer M, Rahman PK (2017) Recent bioreduction of hexavalent chromium in wastewater treatment: A review. Journal Industrial and Engineering Chemistry 55: 1-20.
- https://www.epa.gov/
- Barrera Díaz CE, Lugo Lugo V, Bilyeu B (2012) A review of chemical, electrochemical and biological methods for aqueous Cr (VI) reduction. J Hazard Mater 223-224: 1- 12.
- Tejada Tovar C, Villabona Ortiz A, Garcés Jaraba L (2015) Adsorción de metales pesados en aguas residuales usando materiales de origen biológico. Tecno Lógicas 18(34): 109-123.
- Gutiérrez Corona JF, Romo Rodríguez P, Santos Escobar F, Espino Saldaña AE, Hernández Escoto H (2016) Microbial interactions with chromium: basic biological process and applications in environmental biotechnology. World J Microbiol Biotechnol 32(12): 191.
- Acosta Rodríguez I, Cárdenas González JF, Moctezuma Zárate MG, Martínez Juárez VM (2013) Removal of hexavalent chromium from solutions and contaminated sites by different natural biomasses. In: Applied bioremediation-active and passive approaches. Yogesh B Patil and Prakash Rao (Eds.), INTECH, Croatia, pp. 207-224.
- Greenberg AE, Clesceri LS, Eaton AD (1992) Standard methods for the examination of water and wastewater. American Public Health Association, (18th edn), Washington, USA.
- Pantaler RP, Pulyaeva IV (1985) A spectrophotometric study of complexation between chromium and chromazurol S. Journal of Analytical Chemistry 40: 1634-1639.
- Londolani C, Maremeni C, Modise SJ, Mtunzi FM, Klink MJ et al. (2018) Adsorptive removal of hexavalent Chromium by Diphenylcarbazidegrafted Macadamia nutshell powder. Bioinorganic Chemistry and Applications p. 14.
- Rodríguez A, Pacheco NC, Cárdenas JF, Tovar J, Martínez VM, et al. (2017) Bioadsorción de Cromo (VI) en solución acuosa por la biomasa de Amaranto (Amaranthus caudatus). Avances en Ciencias e Ingeniería 8(2): 11-20.
- Netzahuatl Muñoz AR, Pineda Camacho G, Barragán Huerta BE, Cristiani Urbina E (2010) Evaluación de la cascara de aguacate para la remoción de cromo hexavalente y cromo total de soluciones acuosas. Revista CENIC 41(4): 1-10.
- Sahranavard M, Ahmadpour A, Reza Doosti M (2011) Biosorption of hexavalent Chromium ions from aqueous solutions using Almond green hull as a low-cost biosorbent. European Journal of Scientific Research 58(3): 392-400.
- Acosta Rodríguez I, Ruíz Trujillo KC, Cárdenas González JF, Moctezuma Zárate MG, Martínez Juárez VM (2015) Chromium (VI) removal by Sawdust of pine tree. International Journal of Latest Research in Science and Technology 4(2): 124-128.
- Agarwal GS, Kumar H, Chaudari S (2006) Biosorption of aqueous chromium (VI) by Tamarindus indica seeds. Bioresour Technol 97(7): 949-956.
- Leyva R, Bernal LA, Acosta I (2005) Adsorption of cadmium (II) from aqueous solution on natural and oxidized corncob. Separation and Purification Technology 45(1): 41-49.
- Zubair A, Bhatti HN, Hanif MA, Shafqat F (2008) Kinetic and equilibrium modeling for Cr(III) and Cr(VI) removal from aqueous solutions by Citrus reticulate waste biomass. Water, Air, Soil and Pollution 191(1-4): 305-318.
- Michael Horsfall Jnr, Spiff AI (2005) Effects of temperature on the sorption of Pb2+ and Cd2+ from aqueous solution by Caladium bicolor (wild cocoyam) biomass. Electronical Journal of Biotechnology 8(2): 162-169.
- Ozer A, Ozer D (2003) Comparative study of the biosorption of Pb (II), Ni (II) and Cr (VI) ions onto Saccharomyces cerevisiae: Determination of biosorption heats. J Hazard Mater 100(1-3): 219-229.
- Ghaneian MT, Dehvari M, Jamshidi B (2013) A batch study of hexavalent chromium removal from synthetic wastewater using modified Russian knapweed flower powder. International Journal of Environmental Health Engineering 2(5): 38-46.
- Tyagi U, Khandegar V (2018) Biosorption potential of Vetiveria zizanioides for the removal of Chromium (VI) from synthetic wastewater. Journal of Hazarous Toxic Radioactive Waste 22(4).
- Acosta Rodríguez I, Arévalo Rangel DL, Cárdenas González JF, Moctezuma Zárate MG, Martínez Juárez VM (2015) Hexavalent Chromium (VI) Removal by Penicillium sp. IA-01. In: Nafumi Shiomi (Ed.), Advances in Bioremediation of wastewater and polluted soil. INTECH, Croatia, pp. 165-198.
- Acosta Rodríguez I, Cárdenas González JF, Torre Bouscoulet ME, González Estrada A, Guerrero Martínez SE, et al. (2015) Biosorption of chromium (VI) by different natural biomasses. Journal of Multidisciplinary Engineering Science and Technology 2(7): 1736-1741.
- Kahraman HT, Pehlivan E (2017) Cr6+ removal using oleaster (Elaeagnus) seed and cherry (Prunus avium) stone biochar. Powder Technology 306: 61-67.
- Rangabhashiyam S, Selvaraju N (2015) Adsorptive remediation of hexavalent chromium from synthetic wastewater by a natural and ZnCl2 activated Sterculia guttata shell. Journal of Molecular Liquids 207: 39- 49.
- Kuppusamy S, Thavamanic P, Megharajb M, Venkateswarlue K, Bok Lee Y et al. (2016) Potential of Melaleuca diosmifolia leaf as a low-cost adsorbent for hexavalent chromium removal from contaminated water bodies. Process Safety and Environmental Protection 100: 173-182.
- Volesky B, Holan ZR (1995) Biosorption of heavy metals. Biotechnol Prog 11(3): 235-250.
- Kelly Vargas K, Cerro López M, Reyna Téllez S, Bandala ER, Sánchez Salas JL (2012) Biosorption of heavy metals in polluted water, using different waste fruit cortex. Physics and Chemistry of the Earth, Parts A/B/C 37- 39: 26-29.
- Adesola NA, Oyebamiji J, Adebowale S (2006) Biosorption of lead ions from aqueous solution by maize leaf. International Journal of Physical Sciences 1(1): 23-26.
© 2019 Cárdenas González JF. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






