- Submissions

Full Text
Modern Approaches in Drug Designing
COVID-19 Vaccines
Jagessar RC*
Department of Chemistry, University of Guyana, Turkeyen Campus, Georgetown, Guyana, South America
*Corresponding author: Jagessar RC, Department of Chemistry, University of Guyana, Turkeyen Campus, Georgetown, Guyana, South America
Submission: September 03, 2021;Published: September 14, 2021

ISSN: 2576-9170 Volume3 Issue4
Abstract
COVID-19 is a contagious disease induced by severe acute respiratory syndrome coronavirus 2, SARSCOV- 2. A possible mutant of SARS-COV-1. The disease has spread worldwide since, March 1, 2020, and then became a pandemic. All countries of the world have been affected. To date, there are 215,882,875 confirmed cases with 4,492,818 deaths. USA is the country with the greatest number of confirmed cases and most deaths, followed by our neighboring country Brazil. COVID-19 pandemic has been the impetus for the immediate use of antiviral drugs, such as chloroquine and Remdesivir, amongst others. The former was disapproved by FDA. However, the COVID-19 vaccine has been the only real solution to date, providing over 90% efficacy in all cases. Over 150 pharmaceutical companies are still pursuing research in order to find the best vaccine. Some of the vaccines currently being administered are Oxford Astrazenica, Sputnik-V, Moderna, Pfizer BioNTech, Sinopharm, Sinovac etc.
Keywords: COVID-19; Contagious disease; SARS-COV-2; USA; Oxford Astrazenica; Sputnik-V; Moderna
Introduction
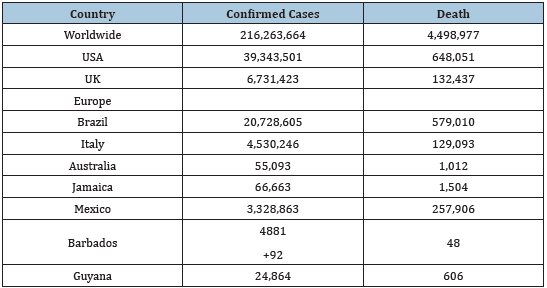
COVID-19) is a contagious disease induced by Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). The latter is said to be a mutant of SARS-COV-1 It was first identified in Wuhan, China, in December 2019 [1]. The disease has since spread worldwide, leading to an ongoing pandemic on the 3rd of March 2020 [2] To date, there are 215,882,875 confirmed cases with 4,492,818 deaths [3]; (Table 1).
Table 1: COVID-19 cases in selected countries.
Symptoms of COVID-19 include fever, cough, headache, fatigue, breathing difficulties, and loss of smell and taste. Symptoms may begin one to fourteen days after exposure to the virus. Severe symptoms include dyspnea, hypoxia, respiratory failure, shock, or multiorgan dysfunction. Older people are at a higher risk of developing severe symptoms [4-8].
COVID-19 disease transmits when people breathe in air contaminated by droplets and small airborne particles. This is highest when people are in proximity, but they can be inhaled over longer distances, particularly indoors. It is thought that the virus is airborne, requiring particulate matter for transmission. People remain contagious for up to 20 days and can spread the virus even if they do not develop any symptoms. Amongst the testing methods developed to diagnose the disease are the Real- Time Reverse Transcription Polymerase Chain Reaction (rRTPCR), Transcription-Mediated Amplification (TMA), or by Reverse Transcription Loop-Mediated Isothermal Amplification (RT-LAMP) from a nasopharyngeal swab [9,10].
Preventive measures include physical or social distancing, quarantining, ventilation of indoor spaces, covering coughs and sneezes, hand washing, and keeping unwashed hands away from the face. The use of face masks or coverings has been recommended in public settings to minimize the risk of transmissions [11,12].
Eradication of the Virus
A first step to the eradication of the virus, involves having a knowledge of the structure of the virus. In order for scientists around the globe to prevent and find a cure for COVID-19, there is a need to know the structure of the virus. A virus particle, also known as a virion, is essentially nucleic acid (DNA or RNA), enclosed within a protein shell, coat, or capsid. Viruses are extremely small, approximately 20-400 nanometers in diameter and occur in varying sizes. Viruses also have a variety of shapes and sizes. Similar to bacteria, some viruses have spherical or rod shapes. Other viruses are icosahedral (polyhedron with 20 faces) or helical shaped. Viral shape is determined by the protein coat that encases and protects the viral genome. The protein coat that encases viral genetic material is known as a capsid. A capsid is composed of protein subunits called capsomeres. Capsids can have several shapes: polyhedral, rod or complex [13].
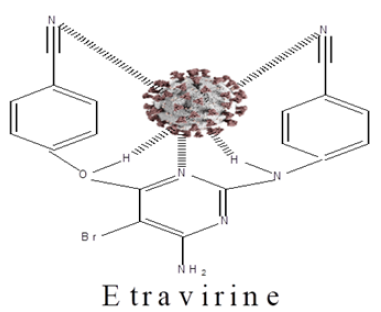
Capsids function to protect the viral genetic material from damage. In addition to the protein coat, some viruses have specialized structures. For example, the flu virus has a membrane-like envelope around its capsid. These viruses are known as enveloped viruses. Viruses may have double-stranded DNA, double-stranded RNA, single-stranded DNA or single-stranded RNA. The type of genetic material found in a particular virus depends on the nature and function of the specific virus. The viral genome can consist of a very small number of genes or up to hundreds of genes depending on the type of virus. Note that the genome is typically organized as a long molecule that is usually straight or circular [14]. In order for a drug to eradicate the virus, there must be complementary between the drug molecule and the SARS-COV-2 virus. The SARS-COV-2 virus is expected to form a complex with the drug molecule, as shown in (Figure 1). A vaccine mode of action is somewhat different as explain below (Figure 2).
Figure 1: Structure of the virus.
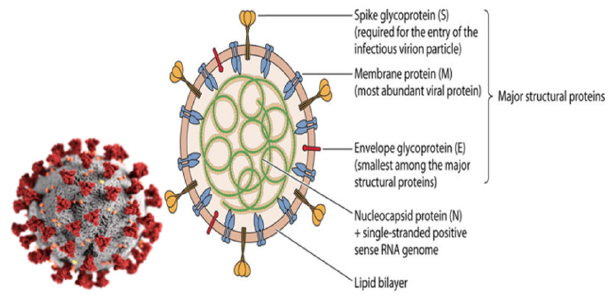
Figure 2: SARS-COV-2-Etravirine complex.
COVID-19 Vaccine
A vaccine is a biological preparation that provides active
acquired immunity to a particular pathogenic microorganism that
are the causative agents to particular infectious diseases. A vaccine
usually contains an agent that resembles the disease-causing
pathogens. It’s often made from weakened or killed forms of the
microorganisms, its toxins or one of its surface proteins. The agent
of the vaccine stimulates the immune system of humans to produce
antibodies that recognize the agent as a threat and destroy it. In
addition, the antibodies will further recognize and destroy any of
the pathogens associated with that agent that it may encounter
in the future. Vaccines can be prophylactic i.e., to prevent or
ameliorate the effects of a future infection by a natural or severe
pathogen or can be therapeutic i.e., to fight against a disease that
already occurred [15].
Vaccines are administered via the process of vaccination.
Vaccination is the most effective method of preventing infectious
diseases. Edward Jenner was the first to develop the concept of
vaccines and created the 1st vaccine to fight cowpox. Vaccines may be
monovalent (univalent) or multivalent (polyvalent). A monovalent
bacteria immunized against a single pathogen or single pathogen
or single microorganism. A multivalent vaccine is one that has two
or more vaccines that can be mixed in the same formulation [16].
A COVID-19 vaccine is one that has been developed to provide
immunity and ultimate eradication of the SARS-COV-2 virus in
the human system. SAR-COV-2 is a virus that causes coronavirus
diseases that ultimately leads to death in humans. It is said to be
a mutant of SARS-COV-1. Once SARS-COV-2-genetic sequence data
was elucidated and shared by GISAID, over 150 Pharmaceutical
companies worked towards the development of a vaccine. These
are in different phases of development. Prior to the COVID-19
pandemic, no vaccine was developed in a rapidly fast period of time
to be effective. In addition, no vaccine existed for the treatment of
Coronaviruses in humans, but in animal. The SARS-COV-2 vaccine is
the fastest vaccine developed to date [17].
There are several vaccine technologies developed to eradicate
COVID-19 disease currently. These include RNA vaccines,
adenovirus vector vaccines, inactivated virus vaccines, subunit
vaccines. RNA vaccines, uses RNA to stimulate an immune response.
When the vaccine is injected into humans, the vaccine RNA acts as
a messenger RNA (mRNA) to cause cells to build the SARS-COV-2
spike protein. This allows the body to identify and destroy the
SARS-COV-2 virus. RNA vaccines nucleoside modified messenger
RNA. RNA vaccines developed to date are Pfizer-BIONTech and
Moderna [18].
Adenovirus vector vaccines. These are non-replicating viral
vector vaccines that uses an adenovirus shell containing DNA that
encodes a SARS-COV-2-protein. They don’t produce new virus
particles but produce only the antigens/antibodies that produce
the immune response. Vaccines of this type include the Oxford-
Astra Zeneca COVID-19-vaccine, the Sputnik V-COVID-19 vaccines.
Inactivated virus vaccines consist of virus particles that have been
grown in culture and then are killed using heat or formaldehyde,
deactivating their disease producing capacity. However, still
stimulating an immune reaction. Examples of such vaccines include
Chinese Coronavirus, BBIBP-CorV, WIBP-CorV, the Indian COVAXIN,
Russian Covivac, the Kazakhstani vaccine QAZVac, Iranian COVIVAN
Barekat. Vaccines of this type in clinical trials include Valneva
COVID-19.
Subunit vaccines present one or more antigens, without
introducing whole pathogen particles. The antigens are protein
subunits. Approved vaccines of this type are peptides vaccines.
EpiVacCorona and ZF2001. Other subunit vaccines that are in the
pipeline are Novavax COVID-19 vaccine, Soberanc 02 (a conjugate
vaccine) and the Sanoti-GSK vaccine [19]. Other types of COVID-19
vaccines that are on clinical trials are virus like particles vaccines,
multiple DNA plasmid vaccines, two lentivirus vector vaccines,
a conjugate vaccine and a vesicular stomatitis virus vaccine,
displaying the SARS -COV-2 spike protein. Scientists have also used
other vaccines that are non-specific to the SARS-COV-2 virus [20].
Status on the Use of COVID-19 Vaccines
There has been 39.7% of the 1st dose of vaccine administered (3,118,469,602). To date: 2,091,593, 226 (26.7%) of the second dose administered. This number will increase globally. It’s anticipated that COVID-19 vaccines will show a high percentage of efficacy against the variant strains: Alpha (B.1.1.7), Alpha (B.1.1.7), Beta (B.1.351) and gamma (P.1) [21,22].
Conclusion
COVID-19 vaccination is on the increase worldwide. It is said that persons vaccinated are the ones less likely to die from the virus. Over 150 pharmaceutical companies are working worldwide to eradicate the disease via the emergence of the best vaccine. To date, COVID-19 vaccines administered are Oxford Astrazenica, Sputnik-V, Moderna, Pfizer BioNTech, Sinopharm, Sinovac. The preference for each varies from one country to another. It’s anticipated that with the use of these vaccines and others, the death toll from COVID-19 will fall. In addition, these vaccines will take into consideration the viral strains.
References
- (2021) Infection of the new coronavirus told the WHO team that his parents had shopped there. The Wall Street Journal.
- Zimmer C (2021) An oily, 100-nanometer-wide bubble of genes has killed more than two million people and reshaped the world. Scientists don't quite know what to make of it. The Secret Life of a Coronavirus.
- (2020) Coronavirus disease 2019 (COVID-19). Centers for Disease Control and Prevention.
- Saniasiaya J, Islam MA, Baharudin A (2021) Prevalence of olfactory dysfunction in coronavirus disease 2019 (COVID-19): A meta-analysis of 27,492 Patients. The Laryngoscope 131(4): 865-878.
- Saniasiaya J, Islam MA, Baharudin A (2020) Prevalence and characteristics of taste disorders in cases of COVID-19: A meta-analysis of 29,349 Patients. Otolaryngology Head and Neck Surgery 165(1): 33-42.
- Agyeman AA, Chin KL, Landersdorfer CB, Liew D, Ofori-Asenso R (2020) Smell and taste dysfunction in patients with covid-19: a systematic review and meta-analysis. Mayo Clin Proc 95 (8): 1621-1631.
- Oran DP, Topol EJ (2021) The proportion of SARS-CoV-2 infections that are asymptomatic: A systematic review. Annals of Internal Medicine 174 (5): 655-662.
- (2020) Interim clinical guidance for management of patients with confirmed coronavirus disease (COVID-19). US Centers for Disease Control and Prevention (CDC).
- Li C, Zhao C, Bao J, Tang B, Wang Y, et al. (2020) Laboratory diagnosis of coronavirus disease-2019 (COVID-19). Clinica Chimica Acta. International Journal of Clinical Chemistry 510: 35-46.
- Ai T, Yang Z, Hou H, Zhan C, Chen C, et al. (2020) Correlation of chest CT and RT-PCR testing for coronavirus disease 2019 (COVID-19) in China: A Report of 1014 Cases. Radiology 296 (2): E32-E40.
- (2020) Recommendation regarding the use of cloth face coverings, especially in areas of significant community-based transmission. US Centers for Disease Control and Prevention (CDC).
- (2020) Scientific Brief: SARS-CoV-2 and Potential Airborne Transmission. COVID-19 Published Science and Research. U.S. Centers for Disease Control and Prevention (CDC).
- Melief CJ, van Hall T, Arens R, Ossendorp F, van der Burg SH (2015) Therapeutic cancer vaccines. The Journal of Clinical Investigation 125 (9): 3401-3412.
- Bol KF, Aarntzen EH, Pots JM, Olde Nordkamp MA, van de Rakt MW, et al. (2016) Prophylactic vaccines are potent activators of monocyte-derived dendritic cells and drive effective anti-tumor responses in melanoma patients at the cost of toxicity. Cancer Immunology, Immunotherapy 65(3): 327-339.
- Brotherton J (2015) HPV prophylactic vaccines: lessons learned from 10 years’ experience. Future Virology 10(8): 999-1009.
- Frazer IH (2014) Development and implementation of papillomavirus prophylactic vaccines. Journal of Immunology 192 (9): 4007-4011.
- Ledford H (2020) What the immune response to the coronavirus says about the prospects for a vaccine. Nature 585 (7823): 20-21.
- Li YD, Chi WY, Su JH, Ferrall L, Hung CF, et al. (2020) Coronavirus vaccine development: from SARS and MERS to COVID-19. Journal of Biomedical Science 27 (1): 104.
- Subbarao K (2021) The success of SARS-CoV-2 vaccines and challenges ahead. Cell Host & Microbe 29(7): 1111-1123.
- Padilla TB (2021) No one is safe unless everyone is safe- why we need a global response to COVID-19. Business World.
- Vergano Dan (2021) COVID-19 vaccines work way better than we had ever expected. scientists are still figuring out why. Buzz Feed News.
- (2021) COVID-19 vaccine tracker. Regulatory Affairs Professional Society.
© 2021 Jagessar RC. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






