- Submissions

Full Text
Modern Approaches in Drug Designing
Investigation of the Opposite Capacities of Erythropoietin and U-74389G on Platelet Crit Levels
C Τsompos1*, C Panoulis2, K Τοutouzas3, A Triantafyllou4, G Ζografos5 and A Papalois6
*Corresponding author: Tsompos Constantinos, Department of Obstetrics & Gynaecology, Mesologi County Hospital, Nafpaktou street, Mesologi 30200, Etoloakarnania, Greece
Submission: August 09, 2017; Published: August 18, 2017

ISSN : 2576-9170Volume1 Issue1
Introduction
U-74389G is not famous for its increasing [1] capacity (p-value=0.0712). U-74389G as a novel antioxidant factor, implicates exactly only 255 published studies. The hypoxia reoxygenation (HR) type of experiments was noted in 4.31% of these studies. A tissue protective feature of U-74389G was obvious in these HR studies. The U-74389G chemically known as 21-[4-(2,6-di-1-pyrrolidinyl- 4-pyrimidinyl)-1-piperazinyl]-pregna-1,4,9(11)-triene-3,20-dione maleate salt is an antioxidant complex, which prevents the lipid peroxidation either iron-dependent, or arachidonic acid-induced one. Animal kidney, liver, brain microvascular endothelial cells monolayers and heart models were protected by U-74389G after HR injury. U-74389G also attenuates the leukocytes; downregulates the proinflammatory gene; treats the endotoxin shock; produces cytokine; enhances the mononuclear immunity; protects the endothelium and presents antishock property.
Erythropoietin (Epo) even if is not famous for its acute decreasing action (p-value=0.0615), it can be used as a reference drug in order a related capacity of U-74389G to become comprehensible. Although Epo is met in over 29,645 published biomedical studies, only a 10.48% of them negotiate the known type of HR experiments. Nevertheless, Epo as a cytokine, never goes out of the jurisdiction of the PCTl study. This experimental work tried to compare the kind of effects of the above drugs on a rat induced HR protocol. They were tested by calculating the serum PCTl alterations.
Materials and Methods
Animal preparation
The Vet licenses under 3693/12-11-2010 & 14/10-1-2012 numbers, the granting company and the experiment location are mentioned in preliminary references [1,2]. The human animal care of Albino female Wistar rats, the 7 days pre-experimental ad libitum diet, the non-stop intra-experimental anaesthesiology techniques, the acidometry, the electrocardiogram and the oxygen supply and post-experimental euthanasia are also described in preliminary references. Rats were 16-18 weeks old. They were randomly assigned to six 6 groups consisted in N=10. The stage of 45min hypoxia was common for all 6 groups. Afterwards, reoxygenation of 60min was followed in group A; reoxygenation of 120min in group B; immediate Epo intravenous (IV) administration and reoxygenation of 60min in group C; immediate Epo IV administration and reoxygenation of 120min in group D; immediate U-74389G IV administration and reoxygenation of 60min in group E; and immediate U-74389G IV administration and reoxygenation of 120min in group F. The dose height assessments for both drugs are described at preliminary studies as 10mg/Kg body mass.
Hypoxia was caused by laparotomic clamping the inferior aorta over renal arteries by forceps for 45min. The clamp removal was restoring the inferior aorta patency and reoxygenation. After exclusion of the blood flow, the protocol of HR was applied, as described above for each experimental group. The drugs were administered at the time of reperfusion; through inferior vena cava catheter. The PCT levels (PCTl) were determined at 60th min of reoxygenation (for A, C and E groups) and at 120th min of reoxygenation (for B, D and F groups). No relation was found between PCTl with animal’s mass (p-value=0.5401).
Statistical analysis
Table 1 presents the (%) declining influence of Epo regarding reoxygenation time. Also, Table 2 presents the (%) increasing influence of U-74389G regarding reoxygenation time. Chi-square tests were applied using the ratios which produced the (%) results per endpoint. The outcomes of chi-square tests are depicted at (Table 3). The statistical analysis was performed by Stata 6.0 software [Stata 6.0, Stata Corp LP, Texas, USA].
Table 1: The (%) declining influence of erythropoietin in connection with reoxygenation time.

Table 2: The (%) increasing influence of U-74389G in connection with reoxygenation time.
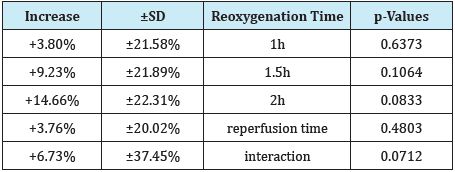
Table 3: The U-74389G/erythropoietin efficacies ratios on PCTl after chi-square tests application.

Results
The successive application of chi-square tests revealed that U-74389G accentuated the PCTl by -.2312044-fold [-.2667818 - -.200372] than Epo at 1h, by -.6719365-fold [-.6726743 - -.6711995] at 1.5h, by -1.330756-fold [-1.848376 - -.9580512] at 2h, by 5.620077-fold [5.601274 - 5.638943] without drugs and by -.9771515-fold [-.9784991 - -.9758057] whether all variables have been considered (p-value=0.0000).
Discussion
The unique available study investigating the increasing effect of U-74389G on PCTl was the preliminary one [1]. Although the most famous activities of neuroprotection and membrane-stabilization properties, it accumulates in the cell membrane, protecting vascular endothelium from peroxidative damage but hardly penetrates the blood-brain barrier. It elicits a beneficial effect in ototoxicity and Duchenne muscular dystrophy. It increases γGT, SOD, and GSH levels in oxygen-exposed cells. It treats septic states and acts as immunosuppressant in flap survival. It prevents the learning impairments, it delays the early synaptic transmission decay during hypoxia improving energetic state of neurons. It shows antiproliferative properties on brain cancer cells and is considered as a new promising anti inflammatory drug for the treatment of reperfusion syndrome in IR injuries.
The same authors confirmed [2] the short-term declining effect of Epo preparations in non iron deficient rats. Rajajee et al. [3] considered acute liver failure patients typically coagulopathic and at risk for intracranial hemorrhage. Fealy et al. [4] associated the decreased platelet count (hazards ratio, 1.19; p = 0.03) with a reduced likelihood of circuit clotting repeated events in critically ill adults requiring continuous renal replacement therapy. Muzaffar et al. [5] could differentiate among normo coagulant, hypo coagulant, hyper coagulant states by thromboelastography unlike conventional coagulation assay which estimated the platelet count at 153.96±99.16 (×103/mm3). Htet et al. [6] estimated almost 20% of critically ill patients having moderate-to-severe coagulopathy (platelets<50k/uL). Shaver et al. [7] associated the elevated micro particle (MP) concentrations in the airspace during ARDS with activation of coagulation, reduced risk of ARDS and contribute to acute lung injury in critically ill patients. Etchill et al. [8] associated the high platelets-to-packed RBCs ratio (>1:2) with decreased 48hour mortality 54.4% (p=0.032) in trauma patients. Mundt et al. [9] showed that differences in platelet counts are not formaldehyde exposure dependent since it induces damage to hematopoietic cells. Francischetti et al. [10] found that post-cannulation platelet counts among other variables also have discriminative power within 30days in surviving patients on ECMO. Jones et al. [11] claimed that packed platelets break down and undergo chemical changes during storage (known as the storage lesion) that lead to an inflammatory response once the blood components are transfused to patients deteriorating their outcome. Bruchim et al. [12] significantly (P <0.05) associated the haemostatic derangements of lower tPCA at 12hours post presentation with the 60% mortality in dogs with naturally occurring heatstroke. Pannu et al. [13] reversed rapidly and safely severe thrombocytopenia (≤20,000/mm3) secondary to dengue virus (DEV) infection by administration of a single dose of 50μg/kg (25 IU/kg) anti-D IV. Patients in the intervention group were achieving 9.85-fold more frequently the platelet count of ≥50,000/mm3 than control group (P = 0.0019) at the end of 48h. Mohammed et al. [14] significantly increased PLTs intracellular ascorbic acid levels by 2.66-fold (Lo Vitamin C) and by 13.08-fold (Hi Vitamin C, P<0.05) after Vitamin C supplementation. Also, Vitamin C at the higher dose (3mmol/L) also induced the release of several eicosanoids including thromboxane B2 and prostaglandin E2, as well as products of arachidonic acid metabolism via the lipoxygenases pathway such as 11-/12-/15-hydroxyicosatetraenoic acid (P <0.05). Marly Voquer et al. [15] ascribed the stronger clot in the FIBTEM assay of rotational thromboelastometry at a stronger contribution of platelets in feline blood of people than cats. Aubron et al. [16] independently associated platelet transfusion with 5.5-fold more hospital-acquired infections in the ICU (p < 0.01), with 2.56%-fold more infections (p < 0.001). Navas Carrillo et al. [17] described the formation of luminal thrombi secondary to platelet activation and the release of thrombogenic elements within the atherosclerotic lesions in acute coronary syndromes. Appiah Kubi et al. [18] detected 36 oncogenic platelet-derived growth factor receptor (PDGFR) ETV6 (TEL)-PDGFRB and FIP1L1- PDGFRA fusions genes. 33 were as a result of chromosomal translocation, FIP1L1-PDGFRA and EBF1-PDGFRB were the result of chromosomal deletion and CDK5RAP2- PDGFRΑ was the result of chromosomal insertion in hematological malignancies. Asanuma et al. [19] estimated the mean values of PCT significantly lower and the TPO levels significantly higher in hemodialysis patients (HD) patients undergoing treatment with rHuEPO at 9000 IU/week than those in healthy controls. Haddad et al. [20] examined the effect of intra peritoneal injections of 40mg/kg of the U-74389G every 12 hours, on acute otitis media in guinea pigs. Streptococcus pneumonia organisms were inoculated into the right tympanic cavity; with the left ear served as a control one. According to above, Table 3 shows that U-74389G accentuated by -.9771515- fold [-.9784991 - -.9758057] the PCTl than Epo (p-value=0.0000); a trend accentuated along time, in Epo non-deficient rats. A metaanalysis of these ratios from the same experiment, for 4 other seric variables, provides comparable results (Table 4).
Table 4: A U-74389G / erythropoietin efficacies ratios meta-analysis on 4 hematologic variables (3 variables with balancing efficacies and 1 variable with opposite efficacies) [21].
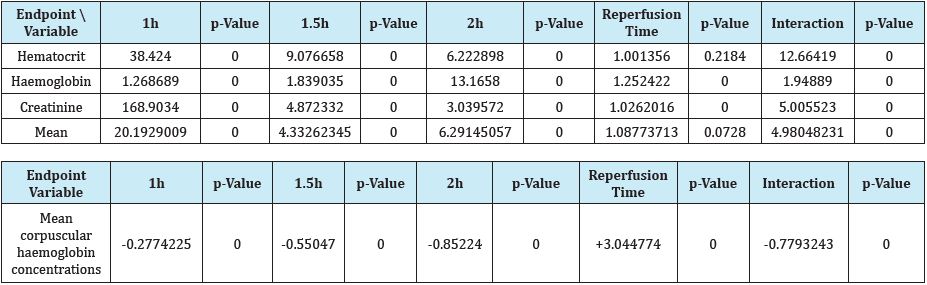
Conclusion
The anti-oxidant capacities of U-74389G accentuated by -.9771515-fold [-.9784991 - -.9758057] the PCTl than Epo (p-value=0.0000) in rats. Also, this trend is accentuated along the short term time frame of the experiment.
References
- Tsompos C, Panoulis C, Toutouzas K, Zografos G, Papalois A (2015) The acute effect of the antioxidant drug “U-74389G” on platelet crit levels during hypoxia reoxygenation injury in rats. Acta Phlebologica 16(2): 77-81.
- Τsompos C, Panoulis C, Τοutouzas K, G Ζografos, Papalois A (2013) The effect of erythropoietin on platelet crit during ischemia reperfusion injury in rats. Annals of the Romanian Society for cell Biology 18(2): 39- 42.
- Rajajee V, Fontana RJ, Courey AJ, Patil PG (2017) Protocol based invasive intracranial pressure monitoring in acute liver failure: feasibility, safety and impact on management. Crit Care 21(1): 178.
- Fealy N, Aitken L, du Toit E, Lo S, Baldwin I (2017) Faster Blood Flow Rate Does Not Improve Circuit Life in Continuous Renal Replacement Therapy: A Randomized Controlled Trial. Crit Care Med.
- Muzaffar SN, Baronia AK, Azim A, Verma A, Gurjar M, et al. (2017) Thromboelastography for Evaluation of Coagulopathy in Nonbleeding Patients with Sepsis at Intensive Care Unit Admission. Indian J Crit Care Med 21(5): 268-273.
- Htet N, Vaughn J, Adigopula S, Hennessey E, Mihm F (2017) Needleguided ultrasound technique for axillary artery catheter placement in critically ill patients: A case series and technique description. J Crit Care 41: 194-197.
- Shaver CM, Woods J, Clune JK, Grove BS, Wickersham NE, et al. (2017) Circulating microparticle levels are reduced in patients with ARDS. Crit Care 21(1): 120.
- Etchill EW, Myers SP, McDaniel LM, Rosengart MR, Raval JS, et al. (2017) Should All Massively Transfused Patients Be Treated Equally? An Analysis of Massive Transfusion Ratios in the Nontrauma Setting. Crit Care Med 45(8): 1311-1316.
- Mundt KA, Gallagher AE, Dell LD, Natelson EA, Boffetta P, et al. (2017) Does occupational exposure to formaldehyde cause hematotoxicity and leukemia-specific chromosome changes in cultured myeloid progenitor cells? Crit Rev Toxicol 47(7): 592-602.
- Francischetti IMB, Szymanski J, Rodriguez D, Heo M, Wolgast LR (2017) Laboratory and clinical predictors of 30-day survival for patients on Extracorporeal Membrane Oxygenation (ECMO): 8-Year experience at Albert Einstein College of Medicine, Montefiore Medical Center. J Crit Care 40: 136-144.
- Jones AR, Frazier SK (2017) Consequences of Transfusing Blood Components in Patients With Trauma: A Conceptual Model. Crit Care Nurse 37(2): 18-30.
- Bruchim Y, Kelmer E, Cohen A, Codner C, Segev G, et al. (2017) Haemostatic abnormalities in dogs with naturally occurring heatstroke. J Vet Emerg Crit Care (San Antonio) 27(3): 315-324.
- Pannu AK, Bhalla A, Singhal M, Suri V, Shafiq N, et al. (2017) Safety and Efficacy of a Single Dose of Anti-D (WinRho®) in Severe Thrombocytopenia Secondary to Dengue Virus Infection. Indian J Crit Care Med 21(2): 80-84.
- Mohammed BM, Sanford KW, Fisher BJ, Martin EJ, Contaifer D, et al. (2017) Impact of high dose vitamin C on platelet function. World J Crit Care Med 6(1): 37-47.
- Marly Voquer C, Riond B, Jud Schefer R, Kutter AP (2017) Reference values for rotational thromboelastometry (ROTEM) in clinically healthy cats. J Vet Emerg Crit Care (San Antonio). 27(2): 185-192.
- Aubron C, Flint AW, Bailey M, Pilcher D, Cheng AC, et al. (2017) Is platelet transfusion associated with hospital-acquired infections in critically ill patients? Crit Care 21(1): 2.
- Navas Carrillo D, Marín F, Valdés M, Orenes Piñero E (2017) Deciphering acute coronary syndrome biomarkers: High-resolution proteomics in platelets, thrombi and microparticles. Crit Rev Clin Lab Sci 54(1): 49-58.
- Appiah Kubi K, Lan T, Wang Y, Qian H, Wu M, et al. (2017) Plateletderived growth factor receptors (PDGFRs) fusion genes involvement in hematological malignancies. Crit Rev Oncol Hematol 109: 20-34.
- Asanuma M, Seino K, Mizuno T, Nasu M, Yamauchi F, et al. (2010) Plasma thrombopoietin level and platelet indices in hemodialysis patients receiving recombinant human erythropoietin. Int J Lab Hematol 32(3): 312-319.
- Haddad J, Egusa K, Takoudes TG (1998) Effects of 21-aminosteroid U-74389G on acute otitis media in a guinea pig model. Otolaryngol Head Neck Surg 118(1): 44-48.
- Tsompos C, Panoulis C, Toutouzas K, Triantafyllou A, Zografos G, et al. (2017) Comparison of the acute chromic capacities of erythropoietin and U-74389G concerning mean corpuscular hemoglobin concentration levels. Discovery 53(260): 443-447.
© 2017 C Τsompos, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






