- Submissions

Full Text
Intervention in Obesity & Diabetes
Management of Diabetes, Metabolic Syndrome and Cardiovascular Complications Using A Novel Diagnostic Platform
Obi Uzomba MD1 and Gundu HR Rao2*
1Medical Director, Diabetes Endocrinology & Metabolism Associates, USA
2Emeritus Professor, Laboratory Medicine, USA
*Corresponding author: Gundu HR Rao, Emeritus Professor, Laboratory Medicine, and Pathology, Director, Thrombosis Research, USA
Submission:November 12, 2018;Published: November 26, 2018

ISSN 2578-0263Volume2 Issue3
Abstract
Metabolic diseases such as hypertension, excess weight, obesity, type-2 diabetes and vascular diseases, have become epidemic worldwide. In the last three decades, obesity has increased by two-fold, and diabetes by four-fold globally. In spite of the decline in cardiovascular disease (CVD) related deaths in industrialized countries, CVD has remained the number one killer for over a century. With scientific discoveries, it is recommended, that once the risks for metabolic diseases are recognized, the only choice we have, is robust management of observed risks. Metabolic risks usually develop as cluster of risks, act in concert to promote the pathogenesis of atherothrombotic state and contribute significantly to the premature mortality and co-morbidities. There is considerable debate in the scientific community, as to what metabolic risks constitute, “metabolic syndrome”. Furthermore, there is no clear-cut guidelines or guidance for the most effective management of metabolic diseases, and resulting clinical complications, more so in preclinical or subclinical states. In view of these observations, we are trying to validate various diagnostic tools, for the development of a noninvasive integrated platform, for the early diagnosis of cardiometabolic risks and management of observed metabolic and cardiac risks. In this overview, we describe the use of a novel integrated platform called, TM-Flow, put together by Dr. Albert Maarek and his team at LD Technologies (www.ldteck.com), Miami, Florida. The integrated diagnostic platform, uses time-tested devices such as, oximeter, photo and volume plethysmography, blood pressure monitor with central aortic systolic pressure, , and a galvanic skin response monitor, to elicit a variety of hemodynamic body signals, representing physiopathology of the test subjects, and a proprietary software which analyzes, computes, and correlates with clinical diagnosis of the various risks and cluster of risks, indicating the progression of metabolic and cardiac diseases. In this review, we will introduce this novel diagnostic tool, and discuss the advantages or usefulness of this system in the early diagnosis, risk stratification, guide to therapy and post-therapy management of the progression or regression of the observed risks and risk-clusters.
Introduction
According to the Global Burden of Disease Study, and the NCD Risk Factor Task Force, metabolic diseases such as hypertension, childhood obesity, adolescent and adult obesity, prediabetes, type-2 diabetes, and vascular diseases, have reached epidemic proportions worldwide [1-17]. Management of modifiable risk factors, and a healthy life style, has contributed significantly to the decline in CVD related deaths in industrialized nations, yet there seems to be increases in diabetes related deaths, in low- and middleincome countries [12-17]. With the rapid progress in emerging technologies, and improved diagnosis, the list of metabolic risks, that promote metabolic disease and vascular disease are increasing by the day. Just to mention a few; oxidative stress, low grade chronic inflammation, altered blood flow dynamics, endothelial dysfunction (hardening of the arteries), prehypertension, insulin resistance, prediabetes, infant, childhood and adolescent obesity, subclinical atherosclerosis and many more. Having said this, it is important to note, not many of these metabolic risks are assessed are treated in day to day practice of preventive medicine.
When it comes to diagnosis of metabolic risks, some are easy but many or complex issues. For instance, diagnosis or elevated blood pressure requires just a blood pressure (BP) monitor. However, if one were to use a continuous blood pressure monitor (ambulatory BP monitor), management of BP would be much more efficient, than the current practice of monitoring one- time measurements at the doctor’s office. Diagnosis of excess weight, obesity is also easy. Once can measure height, and weight of patients, calculate the body-mass index (BMI) or measure waist, hip circumferences, and calculate Waist/Hip ratio. Usual diagnostic methods for prediabetes and diabetes include, measurement of fasting glucose, impaired oral glucose tolerance test, glycosylated hemoglobin A1c (HBA1c), insulin sensitivity. In most clinics, worldwide, diabetes and hyperglycemia are managed by just measuring fasting glucose or at the most half yearly measurements of HBA1c. There are newer emerging technologies available, for predictive and preventive medicine. For instance, Japanese researchers recently presented their findings, about early prediction of future diabetes occurrence, in Berlin, Germany, in an international meeting at this year’s European Association for Study of Diabetes (EASD) conference. They measured the trajectories of fasting glucose, BMI, and insulin sensitivity in individuals. Their Findings showed that on average, several risk factors were more common among individuals, who went on to develop type 2 diabetes compared with those who didn’t. In particular, BMI, fasting glucose, and insulin resistance, were increased up to 10 years before diagnosis, and these differences widened over time. Yet another emerging technology is measurement of plasma free amino acids (PFAA) as predictors of future occurrence of diabetes [18-20]. Although these are useful discoveries, may not be suitable for population-based studies or at low- and medium- income countries.
Novel Diagnosis of Diabetes and Cardiometabolic Risks
The TM-Oxi system, takes measurements from a blood pressure (BP) device and a pulse oximeter. The SudoPath system, measures galvanic skin response to assess the SudoMotor pathway function. Spectral analysis of the photo plethysmograph (PTG) waveform, and electrochemical galvanic skin response, allow the TM-Oxi and SudoPath systems, to calculate several homeostatic markers, such as the PTG index (PTGi), PTG very low frequency index (PTGVLFi), and PTG ratio (PTGr). The focus of these studies was to evaluate these markers (PTGi, PTGVLFi, and PTGr) in CVD patients against a control group, and to calculate an independent cardiovascular risk factor score, diabetes and the diabetes-related neuropathy [21-23]. We have articulated the use of this methodology for the diagnosis of cardiometabolic risk, in many of our recent articles [21-26]. In the present study, we have used a new software developed by the LD Technologies, and the results of these studies are presented in Figure 1-4 as TM-Flow Reports. The TM-Flow is a Medical Devise Data system, that measures hemodynamic body signals, using photo and volume plethysmography, and galvanic skin response test. The system has integrated software, and calculates the mathematical analysis of the signals, and provides markers for diagnosis of cardiometabolic disease and associated cardiovascular and autonomic system dysfunctions.
Figure 1:Autonomic nervous system markers are presented: (sudomotor function), heart rate variability, para sympathetic and sympathetic tests.
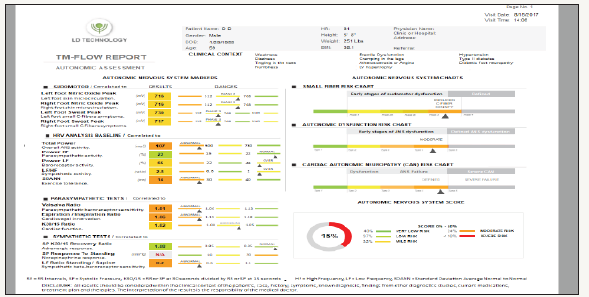
Figure 2:Vascular function markers are presented: Endothelial function (Stress Index), upper large artery (pulse pressure), and lower large artery (pulse wave velocity).

Figure 3:Lifestyle markers and suggestions: Diet markers (BMI), fitness markers (fitness level, mental stress, exercise tolerance, biologic oxygen saturation, heart rate).
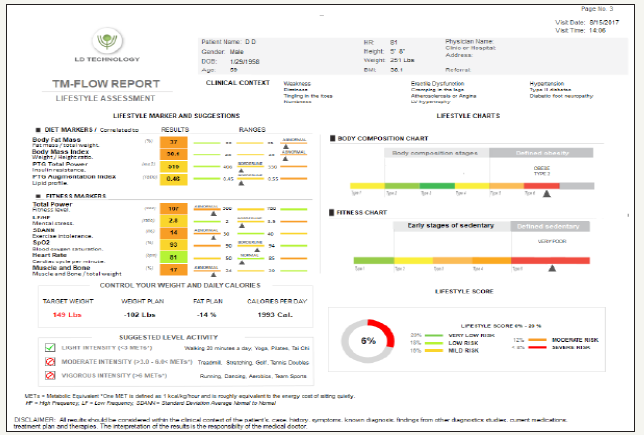
Figure 4:Cardiometabolic score: suggests that the treatment management is not effective.

In a typical TM Flow Report, which includes microvascular and autonomic assessment, Sudo- Motor tests include: left foot nitric oxide peak and right foot nitric oxide peak, representing response of skin microcirculation; left foot sweat response and right foot sweat response, representing C- fiber density. Parasympathetic tests include: markers of the parasympathetic baroreceptor sensitivity, makers of cardiovagal innervation, and cardiac function, while standing. Sympathetic reflex tests include: adrenergic response during standing, norepinephrine response during standing, and markers of the sympathetic beta-baroreceptor sensitivity. Blood pressure responses include: brachial systolic pressure when heart contracts, BP when heart relaxes delta minus diastolic pressure, and markers of the pressure of the aorta.
Markers of artery stiffness include: peripheral small artery tone, aortic stiffness, and lower extremity stiffness (pulse wave velocity). Lower artery markers include: right and left leg artery stiffness (Brachial, left ankle and right ankle: systolic and diastolic). Vascular and endothelial homeostatic markers include: marker for insulin resistance, marker for glucose control, marker for fibrinogen, marker for inflammation and marker for LDL- cholesterol. Body composition markers include: dry lean mass, fat mass as percent of the total weight. Autonomous nervous system (ANS) markers include: marker for the ANS activity, parasympathetic activity at rest, marker of mental stress, marker for the VO2 max.
From the clinical point of view, early stages of sudomotor dysfunction includes, C-fiber inflammation, micro circulation disorder and reduced C-fiber density. ANS and Vascular Factors Score includes; endothelial chart, body composition chart and fitness chart. Whereas, TM- Flow cardiometabolic score report, summarizes the major clinical findings. For instance, if the mild inflammation is detected, appropriate interventions are recommended. In this section of the report, comments include; vascular and ANS risk factors, and hemodynamic risks. In each section of the flow chart, the first column reports the raw data, followed by diagnosis in the next column, in terms of “abnormal, borderline, or normal,” color-coded green to red, to indicate, low to high risk. Each section also contains, a final risk score in percent of overall risk for each category or cluster of risks. Having described the old version (TM-Oxi), as well as the new version, TM-Flow data systems, we will present a typical case history. After making the diagnosis the patient was treated with appropriated interventions, including anti-glycaemic therapy. Although the therapy lowered the blood glucose level and made the patient normo-glycaemic, clinical complications related to diabetes were not significantly altered, suggesting the need for further therapeutic interventions to reduce, reverse, or prevent the development of acute vascular events or end organ failure.
Preliminary Diagnosis
59-year-old male, with Type 2 DM diagnosed more than 20 year ago. Upon initial consultation, his medications included Novolin 70/30 and glyburide 10mg daily. He is intolerant to metformin due to worsening GI side effects. He was not on any specific healthy eating plan and did not exercise regularly. HBA1c at consultation was 8.9%. He complained of both nocturnal and daytime hypoglycaemic episodes as low as 40s-50s occurring 2-3 times a week, requiring medical intervention. His complications are microvascular with autonomic neuropathy, background retinopathy, mild resolving microalbuminuria, glomerular filtration rate is more than 60ml/ min. Comorbidities include hypertension, hyperlipidaemia, obesity. Clinical symptoms included: general weakness, dizziness, sensations of rest tachycardia, bowel habit changes, tingling and numbness of the lower extremities. TM -Flow chart reports are presented in Figures 1-4.
Clinical Diagnosis and Implications
Cardiac autonomic neuropathy in type-2 diabetes confers, a very high risk for silent coronary ischemia. Peripheral autonomic neuropathy affects, lower extremity and induces microcirculation dysfunction
Diagnosis
Insulin resistance syndrome, hyperglycaemia, endothelial dysfunction with inflammatory markers and hyper coagulable state. Findings confer atherosclerotic risk, with residual risk, stratifying this patient as very high risk for multiple adverse cardiovascular events (MACE).
Diagnosis
Insulin resistance syndrome with type-2 diabetes. Hyperlipidaemia associated complications, patient classified as, very high risk for MACE. The risk score better individualizes his lipid set range and goals. Adiposity based chronic disease-stage 2, per moderate to severe complications, classified as Type-2 Obesity per BMI. Clinical and practice significance of these findings suggest employing, complication centric protocol for diagnosis of overweight/obesity, and for better stratification and optimal level of management of the observed risks. Poor fitness level with exercise intolerance, accompanied by lower oxygen saturation, significance is diagnosis of cardiac autonomic neuropathy, with risk for silent coronary events. This justifies ischemic non-obstructive and obstructive and functional work up, to better stratify for individualized level of physical activity, for functional safety and cardiometabolic improvement. In view of the history of Type 2 diabetes, with comorbid metabolic syndrome and associated complications, optimal therapy requires, range of lifestyle changes with medications dedicated to pathophysiologic approach, to modify the complex dysfunctional cardiometabolic abnormalities, aiming at the core metabolic defects. Recent cardiovascular outcome (CVOT) trials, have reflected on such approaches for diabetes management [27,28]. Thus, a paradigm shift in the current diabetes management is advocated, for optimal therapy. Researchers of these trials concluded, that “Beside the high relevance of CV endpoints, microvascular endpoints, should not be neglected. It was agreed, that novel treatments should also be tested against treatment approaches, which have already shown to have proven CV benefit. It was acknowledged, that it will be an increasing challenge to demonstrate an additional CV benefit. Suggestions included, the need for mechanistic studies to further understanding mechanisms responsible for CV and/or microvascular benefits, as well as potential side effects. Studies should also help to clarify, which group of diabetic patients benefits from specific treatment strategies or class of treatment, and which diabetic patient groups do not.”
Discussion
One of the major contributors for the observed diabetes complications, is the late diagnosis of diabetes conditions [26]. It has been quite well established, that full-blown diabetes occurs almost a decade after the prediabetes stage develops [29,30]. Now the researchers from Shinshu University in Japan, have tracked over 27,00 non-diabetic and diabetic adults, and found that increased fasting glucose, higher body mass index, and impaired insulin sensitivity, were detectable up to 10 years before the diagnosis of diabetes, as well as prediabetes. Results of these studies were reported, in this year’s European Association for the Study of Diabetes (EASD) Annual Meeting in Berlin, Germany (Diabetology. Oct 4, 2018). In view of these observations, it can be easily concluded, that by the time an individual is diagnosed as diabetic, he or she will have had hyperglycaemia-mediated vascular damage for over two decades. Therefore, if at all we need to stop, reduce, reverse diabetes, we need to start our efforts way early in the game. Furthermore, even after the diagnosis, by and large clinicians pay a great emphasis in manging hyperglycaemia, more than the chronic complications associated with this disease. These strategies will not be change to able to stop, reduce, or reverse, the growing epidemic of obesity and diabetes world-wide. Type-2 diabetes is a considerably more complex disease, than is expressed by hyperglycaemia only. Insulin resistance, dyslipidaemia, oxidative stress, inflammatory activation, endothelial dysfunction, and hypercoagulability are all components in this metabolic disease. Having said that, a diagnostic platform that can monitor all this diabetes related chronic complications does not exist. In this article, we are describing an integrated platform, that can diagnose individual cardiometabolic risks as well as cluster of such risks, related to macro and micro-vascular complications.
The TM-Flow Data System, is a Non-Invasive Cardiometabolic Point of Care Integrated System, put together with well-tested FDA approved medical devices like, pulse oximeter, blood pressure monitors, and galvanic skin response monitors, which can access variability in functionalities of body functions and responses, to electrical and mechanical stimulations, collect electrical outputs, compute, analyze, and diagnose cardiometabolic risks and diseases with cardiovascular and autonomic systems. The system is novel, less expensive, mobile and easy to use, compared to the number of diagnostic tools needed to obtain the type of information that TMFlow generates. The data generated using this system, is validated by clinical studies using gold standard assays, using other standard established clinical methodologies.
Conclusion
In 2008, the U.S Food and Drug Administrations (FDA), issued a new guidance for the development of antidiabetes drugs for type- 2 diabetes. Since that time, nine cardiovascular outcomes trials (CVOTs) have reported their findings and another 13 are under way. Since diabetics have increased risk for CV events, these FDA recommendations are timely and useful. However, diabetics also have many other clinical complications other than those related to CVD, as a result of hemodynamic alterations. In the majority of clinics worldwide, management of diabetes is more or less restricted, to the management of blood glucose levels or to bring down the glycosylated hemoglobin (HBA1c) levels, down to meet various regulatory guidelines or guidance statements. Many clinical trials have reported, that lowering the glycaemia alone will not reverse, reduce or prevent, the vascular pathology or the development of clinical complications. We and others are of the opinion, if simple cost effective noninvasive diagnostic platforms are available, then various metabolic and cardiovascular risks associated with the severity of hyperglycemia can be better managed. In this case report, we have used a novel diagnostic platform called. TMFlow and shown, the capabilities of a well-integrated system, in detecting clinical features, which could have been easily missed in regular clinical visits. In addition, this case-study demonstrates, that normalizing blood glucose values or the HBA1c values alone, will not be sufficient in the optimal management of this complex chronic metabolic disease.
Acknowledgement
Authors acknowledge their thanks for the technical advice provided by Dr. Albert Maarek, CEO, LD-Technologies, Miami, Florida, USA, and look forward to working with him and his team.
References
- Forouzanfar M, Liu P, Roth GA, Ng M, Biryukov S, et al. (2017) Global burden of hypertension and systolic blood pressure of at least 110 to 115mmHg, 1990-2015. JAMA 317(2): 165-182.
- Van LF, Mertens IL, De Block CE (2006) Mechanisms linking obesity with cardiovascular disease. Nature 444(7121): 875-880.
- GBD 2015 Risk Factors Collaborators (2015) Global, regional, and national comparative risk assessment of 79 behavioral, environmental and occupational, and metabolic risk or clusters of risks, 1990-2015: A systematic analysis for the global burden of diseases study 2016. Lancet 390(10100): 1345-1422.
- Zheng X, Jin C, Liu Y, Zhang J, Zhu Y, et al. (2015) Arterial stiffness as a predictor of clinical hypertension. J Clin Hyperten (Greenwich) 17(8): 582-291.
- Ng M, Fleming T, Robinson M, Thomson B, Graetz N, et al. (2013) Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013. Lancet 384(9945): 766-781.
- Lu Y, Hajifathalian K, Ezzati M, Woodward M, Rimm EB, et al. (2014) Metabolic mediators of the effect of body-mass index, overweight and obesity on coronary heart disease and stroke: a systematic analysis of 97 prospective cohorts with 1.8 million participants. Lancet 383(9921): 970-983.
- Krug EG (2016) Trends in diabetes: Sounding the alarm. Lancet 387(10027): 1485-1486.
- Smith CM, Bhattacharya J, Goldhaber JD (2012) Diabetes, its treatment, and catastrophic medical spending in 35 developing countries. Diabetes Care 35(2): 319-326.
- https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(16)00618-8/fulltext
- Alexander L, Anderson HR, Bachman VF, Biryukov S, Brauer M, et al. (2015) Global, regional, and national comparative risk assessment of 79 behavioral, environment and occupational, and metabolic risk or clusters of risk in 188 countries, 1990-2013: A systematic analysis for the global burden of disease study 2013. Lancet 386(10010): 227-2323.
- Merlotti C, Morabito A, Pontiroli AE (2014) Prevention of type-2 diabetes; a systematic review and meta-analysis of different intervention strategies. Diab Obes Metab 16(8): 719-727.
- Uusitupa M, Peltonen M, Lindström J, Aunola S, Parikka P (2009) Ten-year mortality and cardiovascular morbidity in the finnish diabetes prevention study-secondary analysis of the randomized trial. PLoS One 4(5): e5656.
- Li G, Zhang P, Wang J, Gregg EW, Yang W, et al. (2008) The long-term effect of lifestyle interventions to prevent diabetes in China da qing diabetes prevention study: a 2-year follow-up study. Lancet 371(9626): 1783-1789.
- Diabetes Prevention Program Research Group (2002) Reduction in the incidence of type-2 diabetes with lifestyle intervention or metformin. N Engl J Med 346: 393-403.
- Puska P (2002) Successful prevention of non-communicable disease: 25-year experience with North Karelia project in Finland. Publ. Health Med 4(1): 5-7.
- Di Cesare M, Bennet JE, Best N, Stevens GA, Danaei G, et al. (2013) The contributions of risk factor trends in cardiometabolic mortality in 26 industrialized countries. Int J Epidemiol 42(3): 838-848.
- Yusuf S, Hawken S, Ounpuu S, Dans T, Avezum A, et al. (2004) Effect of potentially modifiable risk factors associated with myocardial infraction in 52 countries (the INTEHEART study): Case-control study. Lancet 364(9438): 937-952.
- X Bi, Henry CJ (2017) Plasma-free amino acid profiles are predictors of cancer and diabetes development. Nutr Diab 7(3): e249.
- Felig P, Marliss E, Cahill GF (1969) Plasma amino acid levels and insulin secretion in obesity. N Engl J Med 282(15): 811-816.
- Wang TJ, Larson MG, Vasan RS, Cheng S, Rhee EP, et al. (2011) Metabolite profiles and the risk of developing diabetes. Nat Med 17(4): 448-453.
- Gandhi PG, Rao GHR (2014) The Spectral analysis of photoplethysmography to evaluate an independent cardiovascular risk factor. Int J Gen Med 7: 539-547.
- Gandhi PG, Rao GHR (2015) Detection of neuropathy using a SudoMotor test in type-2 diabetes. Degenerative Neurological and Neuromuscular Disease 5: 1-7.
- Maarek AA, Gandhi PG, Rao GHR (2015) Identifying autonomic neuropathy and endothelial dysfunction in type II diabetic patients. EC Neuropathy 2.2(2015): 63-78.
- Rao GHR (2018) Diagnosis of early risks, management of risks, and reduction of vascular disease. J Clin Card & Diagn 1(1): 1-11.
- Rao GHR (2018) Integrative approach to the management of cardiometabolic diseases. J Cardiol and Cardiovasc Sci 2(3): 37-42.
- Rao GHR, Bharathi M (2016) Mother and child: First step for prevention of cardiometabolic disease. J Cardiol (Photon Journal) Photon. 109: 179-186.
- Schnell O, Standl E, Cartrinoiu D, Genovese S, Lalic N, et al. (2018) Trial (CVOT) summit of the diabetes and cardiovascular disease (D & CVD) EASD study group. Cardiovasc Diabetol 17(1):30.
- Cefalu WT, Kaul S, Gerstein HC, Holman RR, Zinman B, et al. (2018) Cardiovascular outcomes trials in Type-2 diabetes: Where do we go from here: Reflections from a diabetes care editors’ expert forum. Diab Care 41(1): 14-31.
- Bansal N (2015) Prediabetes diagnosis and treatment: A review World J Diab 6(2): 296-303.
- Bloomgarden ZT Inzuccchi SE, Karniele E, Le RD (2008) The proposed terminology “A(1c)-derived average glucose is inherently imprecise and should not be adopted. Diabetologia 51(7): 1111-1114.
© 2018 Gundu HR Rao. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






