- Submissions

Full Text
Investigations in Gynecology Research & Womens Health
eHealth for Care During Pregnancy: A Scan of Systematic Review Protocols Registered in PROSPERO
Stefhanie Conceição de Jesus1*, Graziele Telles Vieira1, Fiona Ann Lynn2, and Maria de Lourdes de Souza3
1Postgraduate Nursing Program, Federal University of Santa Catarina, Brazil
2School of Nursing and Midwifery, Queen’s University Belfast, Northern Ireland, United Kingdom
3Repensul Institute, Postgraduate Nursing Program, Federal University of Santa Catarina, Brazil
*Corresponding author:Stefhanie Conceição de Jesus, Postgraduate Nursing Program, Federal University of Santa Catarina, Florianopolis, Santa Catarina, R. Delfino Conti, S/N-Trindade, Florianópolis-Santa Catarina, 88040-370, Brazil
Submission:April 22, 2024;Published: May 16, 2024

ISSN: 2577-2015 Volume4 Issue5
Abstract
Objective: To identify and summarize the systematic review protocols assessing eHealth technologies to enhance care and practice during pregnancy.
Methods: A literature scan was conducted in PROSPERO, an international prospective register of systematic reviews. Search terms “e-health”, “ehealth” and “telemedicine” were applied. Retrieved protocols were screened against eligibility criteria. Additional searches of authors’ profiles were carried out to source publications of results. Characteristics of included protocols were presented in tabular form with an accompanying narrative summary.
Result: Thirteen systematic review protocols were identified as eligible for inclusion in the literature scan. Protocols were registered from 2016-2022. The eHealth technologies outlined in the protocols included digital interventions, mHealth, telehealth, and internet-based interventions. The technologies targeted outcomes related to general maternal health, diabetes management, maternal mental health, health education, and health behavior. Tools for assessing the risk of bias in included studies were widely cited. Reporting guidelines for the reviews and grading of recommendations were cited in the minority of protocols.
Conclusion: While eHealth technologies are an innovative area of maternity care research, there is limited high-level evidence summaries in the form of systematic reviews to help inform the development of evidence-based practice guidance. Further evidence syntheses assessing the effect, cost-effectiveness, and acceptability of eHealth for care in pregnancy are needed.
Keywords: eHealth; Digital technology; Pregnancy; Maternal health; Systematic review; Evidence-based
practice
Introduction
The period of pregnancy can present risks to the health of women when not accompanied by adequate antenatal care [1]. eHealth technologies may be useful tools for health care professionals and health workers to enhance practice and support care of pregnant women. They have the potential to support various aspects of health care, including health education, care, promotion and prevention, surveillance, and management [2,3], representing innovation in the context of maternity services.
The National Library of Medicine of the United States of America classifies eHealth as relating to Telemedicine, Tele-referral, Virtual Medicine, Tele Intensive Care, Mobile Health (mHealth), Telehealth. It defines the provision of health services through remote telecommunications, which includes interactive consultation and diagnostic services [4]. In its broadest sense, eHealth constitutes a technological innovation to improve health services in networking supported by information and communication tools [5].
In primary health care, three functions delineate eHealth as potential contributors. The first function is to “inform, monitor and track”, which uses the potential of technology to educate the population and monitor health parameters over time; the second is “interaction”, which uses eHealth technology to facilitate communication between agents in the health scenario; and the third is the “use of data”, in which health information is used for decision-making and intervention [5].
Research and development related to eHealth is relatively new in health sciences. Initiatives that encourage the development and research of eHealth technologies to improve maternal health are essential, especially to prevent maternal mortality [6,7]. Therefore, investing in this area of maternity care is part of the strategies to improve maternal and infant health. To achieve this, it is essential to carry out experimental research in this area of care.
Systematic reviews are highly regarded, especially in health research. This type of research provides a synthesis of evidence from the available literature to answer a scientific research question, investigated rigorously and transparently [8]. Systematic reviews require a protocol, as this contributes to the quality, transparency, and credibility of this type of evidence.
Systematic review protocols are documents that guide the review process. According to PRISMA-P, the protocols for systematic review must provide a detailed description of the methods, techniques, and procedures to be applied, from defining the question for the investigation to conducting, reporting, and disseminating results [9]. The protocol ensures transparency and replicability and minimizes biases in the review process [9].
Although publication of a systematic review protocol in a scientific journal is not a mandatory requirement for publication of a review, prospective registration is considered good practice. Many scientific journals encourage or, in some cases, require the submission and registration of the systematic review protocol in a database platform, such as PROSPERO. This is an international platform for prospective registration of systematic review protocols, with or without meta-analysis, in the field of human and animal health sciences [10,11]. This platform was launched in 2011 and is maintained by the Centre for Reviews and Dissemination (CRD) at the University of York, United Kingdom, as an open-access platform funded by the National Institute for Health Research (NIHR) [10,11].
The registration of systematic literature review protocols is not yet mandatory. However, it is important to strengthen the commitment of reviewers to include it as a milestone in the review process. A study by Sideri et al. [12] suggested that the registration of a protocol in PROSPERO was associated with an improvement in the quality of the systematic reviews, evaluated from the application of the AMSTAR tool. In addition, in the reviews whose protocols were registered in PROSPERO, greater citations were identified for the use of tools such as the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA), the use of the GRADE approach (Grading of Recommendations Assessment, Developing and Evaluation) to assess the quality of evidence, in addition to the inclusion of flow diagrams of the screening process and the provision of quantitative synthesis [12].
On the other hand, good practices for PROSPERO’s use are required so that its registration can be a useful and adequate database. Users must perform a prior search to identify records in progress or completed about their topic of interest. Authors should be responsible for providing accurate data and updating revisions to the protocol whenever necessary, presenting appropriate justifications, as well as updating the status of the systematic review [13,14].
The PROSPERO initiative supports the scientific community with the publication and registration of protocols. It has also been a source for data extraction for literature scans and scoping reviews in health. For example, studies related to COVID-19 [15,16], perioperative acute pain [17], urology [18], and review methodologies [19].
By analyzing systematic review protocols registered in PROSPERO, researchers can gain an understanding of the basis for searching, appraising, and synthesizing evidence in a broad research area. In particular, the types of interventions, theoretical underpinnings, study designs, outcomes of interest, as well as potential knowledge gaps. This current study aimed to identify and analyze protocols of systematic reviews registered in PROSPERO related to eHealth technology interventions evaluated for care during pregnancy.
Method
A scan of the literature was conducted, using the PROSPERO platform as the main source of data to understand the basis for searching, appraising, and synthesizing evidence on eHealth technologies for care during pregnancy.
Data collection
To identify relevant protocols in PROSPERO, the search terms “e-health”, “ehealth” and “telemedicine” were entered separately. No limitation was placed on the date of registration. The search was carried out in February 2023.
Screening
The records of identified protocols were exported to EndNote web and duplicate protocols were removed. The titles of the remaining records were screened for relevance to eHealth technologies and pregnancy by one reviewer. A second reviewer independently screened a random sample of 10% of the records, as recommended by Garritty et al. [20]. Disagreements were resolved in a consensus meeting, with the participation of a third reviewer.
Eligibility criteria
Full records of registrations were then assessed independently by two reviewers to ensure relevance to the eligibility criteria. The inclusion criteria were (i) systematic review protocol registered in PROSPERO; (ii) indicated one or more eHealth technologies as an intervention for review; and, (iii) related to care during pregnancy. Exclusion criteria were records that did not fulfill all the inclusion criteria. Results of the search and screening process were presented in a flow diagram according to PRISMA [21].
Data extraction
Relevant data were extracted from included protocols using Microsoft Excel by one reviewer, and a second reviewer checked and verified the data entry. All disagreements were discussed with a third reviewer. Data were extracted related to the citation details (CRD registration number, year of registration in PROSPERO, country of origin, number of review team members and their organizational affiliations, funding sources, stage of review and number of versions), and methodological details (reference to reporting guidelines, risk of bias assessment tool, planned strategy for data synthesis, and methods for grading evidence). These items were categorized and descriptive statistical analysis performed with results presented as frequencies and percentages.
Data describing the types of study designs to be included in the review, condition or domain being studied, a description of the population of interest, intervention(s)/exposure(s) of interest, and primary and secondary outcome(s) of interest were summarized and presented in a characteristic of included studies table, as recommended by Page et al. [21]. An accompanying narrative synthesis was reported to summarize and discuss the methodological details and characteristics of the included systematic review protocols.
Additional information sources
Additional searches were carried out on the authors of included protocols to ascertain the status of the systematic reviews in August 2023. LinkedIn, orcid.org, institutional websites, and Google Scholar were used for this search to identify publications aligned with the systematic review protocols. Data extracted for these additional searches were: areas of expertise of the authors, publication identified of the systematic review, and/or protocol in a scientific journal.
Ethics statements
This study is part of a project entitled “e-Health technologies to improve care and support for women at risk of maternal mortality during the antepartum, intrapartum and postpartum period”, approved and funded by CONFAP-CNPq-The UK Academies Fellowships, for a Researcher Mobility Award for UK researchers in Brazil (2022TR002073).
Result
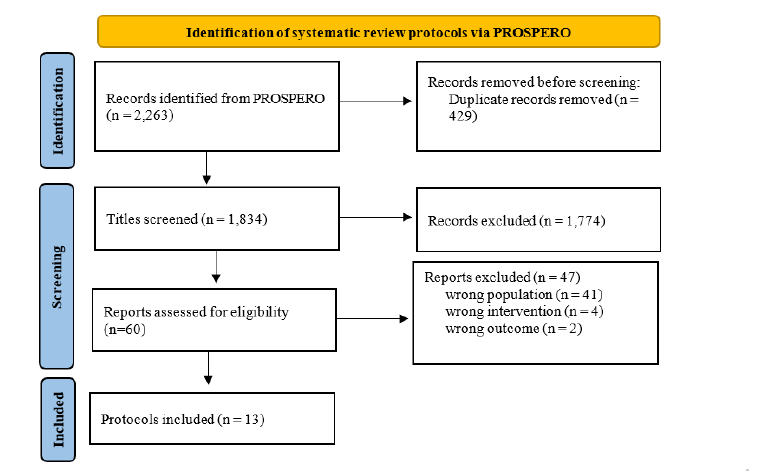
Thirteen systematic review protocols registered in PROSPERO were identified as eligible and included in the analysis. The process for identification and selection is shown in (Figure 1).
Figure 1:Flow diagram of the search process (Source: Adapted from Page et al. [21]).
(Table 1) presents descriptive statistics summarizing the citation details and methodological characteristics of the protocols. Records of eHealth-related protocols for care during pregnancy were registered in PROSPERO from 2016 to 2022. The majority of protocols were developed by review teams based in Asia (n=5, 38%), followed by North America (n=3, 23%) and Europe (n=3, 23%). The remaining two were from Australasia and a partnership between Australasia and Europe.
Table 1:Descriptive statistics of included protocols (n=13).
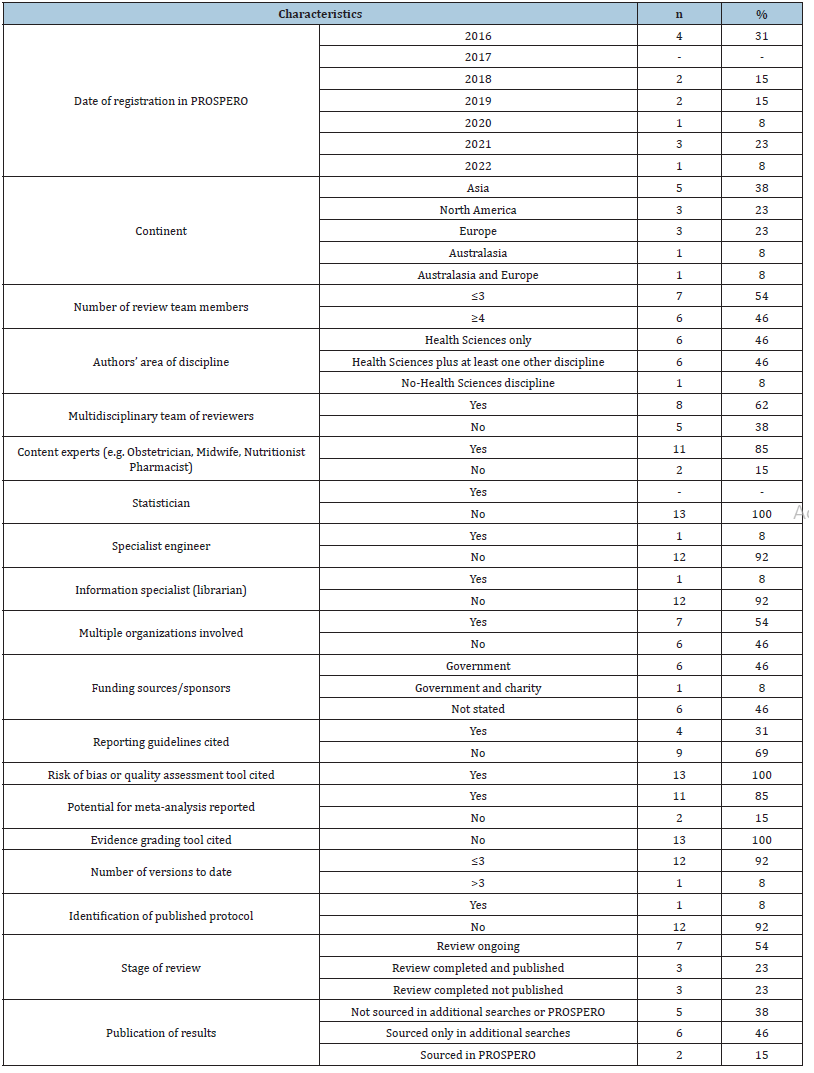
Six protocols (46%) were authored by four or more authors. In addition, six protocols (46%) were planned by professionals in the area of Health Sciences, although partnerships from other disciplines were identified, such as Biological Sciences, Human Sciences, Social Sciences, and Engineering. The review teams were composed of multidisciplinary teams in the majority. Most of the protocols were planned by content specialists such as health professionals, and only one protocol had included an engineering specialist. An information specialist was listed as a co-author in only one review team. We identified in seven protocols that the review teams were from multiple organizations that were largely higher education institutions. Most of the protocols (n-7, 54%) reported a source of funding, with six funded by government institutions, and one by a government institution and charitable organization.
In four (31%) protocols citation of reporting guidelines was identified, and in all protocols a risk of bias tool was cited to assess the quality of the included studies. We observed that in eleven (85%) protocols a meta-analysis would be carried out if feasible and dependent on the characteristics of the included primary research studies identified as eligible for inclusion. None cited the GRADE approach, or similar, for grading recommendations of findings.
Most protocols (n=12, 92%) had less than three versions listed, and only one protocol listed six versions. Six protocols were listed as completed reviews (three with a publication sourced, and three without a publication sourced), and seven as ongoing (54%). Of the latter, two records had not been updated in PROSPERO, as subsequent publications of findings from the systematic reviews were identified in the additional searches. On the PROSPERO platform, the subsequent publication of the systematic review protocol in an academic journal was not described in any record. In the additional searches, a publication of one systematic review protocol in a scientific journal was identified. The publications of the systematic reviews that stemmed from the protocols. Were obtained only through the additional searches and had not been listed in PROSPERO, which provides authors with a specific section for this purpose.
(Table 2) presents the characteristics of the included protocols in terms of the type of study designs to be included, the condition and population being studied, interventions of interest, with an example of the technology cited in the protocol, and outcomes of interest.
Table 2:Characteristics of included protocols (n=13).
Note: CRD: registration identifier number in PROSPERO.
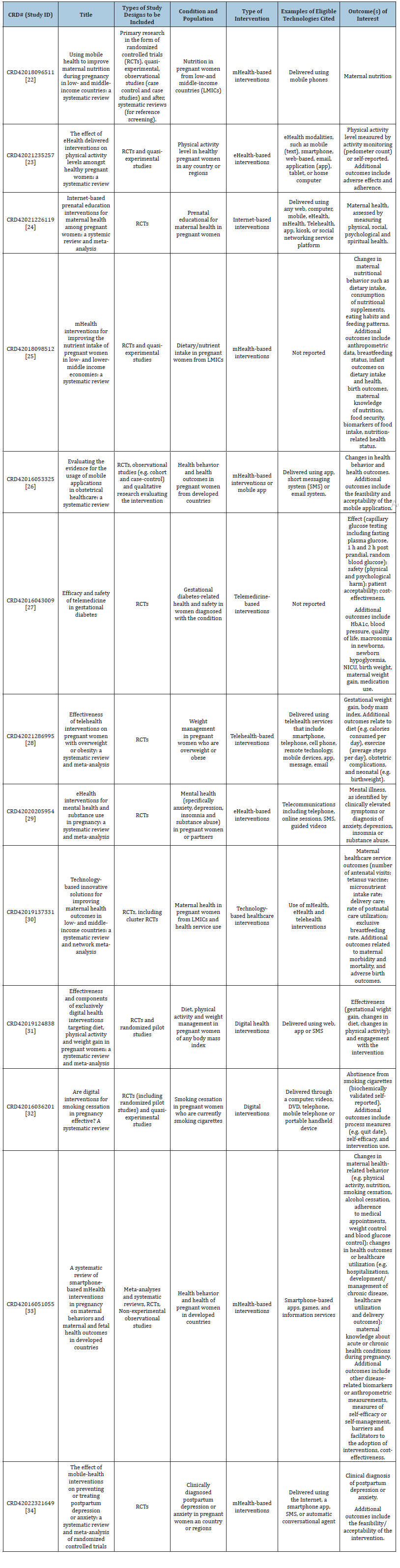
For the study designs, all protocols set out to review RCTs, with seven of the protocols [24-34] citing RCTs alone (including randomized pilot studies and cluster RCTs) for the type of study design to be included. The remaining six protocols [22,23,25,26,32,33] cited additional types of studies to be reviewed, such as quasi-experimental or observational studies, including cohort and case-control studies. Systematic reviews were included in two protocols [22,33], with the latter planning to use this type of study only for reference screening. Only one protocol [26] included studies with a quantitative and qualitative approach, as their review question included synthesizing evidence on the feasibility and acceptability of mobile applications.
While this literature scan aimed to identify eHealth technologies for pregnancy care, it was not restricted to pregnant women and their infants. However, only one protocol included partners of pregnant women within the population of interest and outcomes related to their mental health [29]. Populations with specific clinical diagnoses or additional care needs were identified, such as gestational diabetes, overweight or obesity, cigarette consumption, mental illness and substance abuse. Eight protocols [22-26,30,31,33] did not identify populations with specific clinical diagnoses or additional care needs, rather focused on general pregnant populations. Related to the setting, three protocols focused on evidence from low-middle income countries [22,25,30], two to developed countries only [26,33], with the remaining protocols specifying no limitations on the region [23,24,27,29,31,32,34].
We identified a variety of conditions being studied in the protocols. These included general health, mental health, nutrition, physical activity levels, prenatal educational interventions, gestational diabetes management, weight management, health behavior, and smoking cessation. The eHealth technologies were cited with different typographies in the protocols such as eHealthbased interventions, mHealth-based interventions, internet-based interventions, telemedicine-based interventions, telehealth-based interventions, and digital health interventions. Different examples of eHealth technologies were identified in the protocols, including short messaging system, email system, application (app), videos, games, DVD, or automatic conversational agent for example.
According to primary and secondary outcomes listed in the protocols, five technologies were aimed at maternal behavior changes [25,26,29,32,33]. The effectiveness or the effect of eHealth on pregnancy care were reported in the protocols for the primary and/or secondary outcomes. Two protocols [26,34] referred to the assessment of feasibility and acceptability, one of which included quantitative and qualitative studies [26]. Evidence of selfefficacy or self-management measures, barriers, and facilitators to intervention adoption, and cost-effectiveness measures were reported in one protocol [33]. This protocol included a broader range of study designs for eligibility, and was the only one that included systematic reviews and meta-analyses in the report.
Discussion
eHealth technologies have the potential to support care for pregnant women [35]. Although innovation in these types of technologies has expanded in recent years, the use of eHealth for this specific population and area of care remains uncertain. This is especially related to the availability of high-level evidence to help inform the development of evidence-based practice guidance. Through the analysis of systematic review protocols registered in PROSPERO, we identified research protocols that serve as a basis for searching, evaluating, and synthesizing the development of evidence in this area of research.
To our knowledge, this is the first study analyzing systematic review protocols registered in PROSPERO on eHealth for pregnancy care. We identified and analyzed 13 systematic review protocols, registered from 2016 to 2022, from countries classified by the World Bank as high-income [36]. A network analysis examining the allocation of research across different disciplines and economic income levels of countries. Found indicate that high-income countries shared common trends in the research productivity distribution over time [37]. It has also been considered that led by high-income countries have limited transferability to Low-and Middle-Income Countries (LMICs) [38].
The protocols analyzed were mostly composed of four or more authors. Systematic reviews have fundamental stages in which they are carried out independently between authors [21,39], which should include paired and independent screening, selection, and extraction by two or more authors [40]. Conflicts can thus be identified in subjective decisions and consensus reached through discussion with the team of reviewers [39]. Therefore, the number of review team members was considered appropriate.
Five review teams were not considered to be multidisciplinary. Protocols produced by health science professionals prevailed. While this was expected for the study area of pregnancy care. A greater degree of input and collaboration may have been expected with colleagues from the areas of engineering and computer sciences. Co-authorship from this discipline was identified in only one protocol. Systematic reviews related to eHealth intervention would benefit from the necessary expertise to guarantee full assessment of the applicability, quality, and safety. A range of experts may be required, such as content experts who know about pregnancy and related care (e.g., midwives, obstetric nurses, obstetricians, general practitioners); information specialists, who understand systematic search techniques (e.g., subject librarians); technological experts or engineers to support the technical and technological review of interventions; systematic review experts to support the methodological development of the review; and a statistician if the authors plan to perform meta-analyses.
Differing clinical and/or methodological expertise will add depth to discussions, contributing to the robustness of the developed protocol and consequently the planned systematic review. In most protocols, different authors linked to different HEIs were identified. Collaborative research can promote a diversity of perspectives, complementary expertise, resource sharing, validity, and reliability, improve impact, and better solve possible scientific challenges [41]. Thus, scientific and institutional collaborations are important in promoting innovation, quality, and relevance of scientific discoveries.
The production of science and technology is known to be important for the social, economic, and technological development of a country. Research funding contributes to subsidizing these advances [42]. In this study, most systematic review protocols recorded sources of funding from governmental bodies. This result is important in view, above all, of the study population for which it is intended: pregnant women, suggesting it is a valued area of research for public policy.
Although the PRISMA-P is aligned with PROSPERO, only four protocols cited the use of this reporting guideline in the registration. PRISMA-P consists of an item checklist intended to facilitate the preparation and reporting of a robust protocol for systematic review [9]. Clear and comprehensive protocols enhance the comprehension and evaluation of review methods and enable the identification of any alterations to the methods and selective reporting in concluded reviews [9]. Therefore, reporting guidelines are designed to facilitate the comprehensive reporting of protocols as well as manuscripts, ultimately contributing to the enhancement of the quality of available publications.
Tools to assess the risk of bias were cited in the systematic review protocols evaluated. The risk of bias reflects the methodological quality of the scientific studies included in the review [43]. In referencing the utilization of validated methodological tools, the authors showcase their understanding of review methods and the need to uphold a high level of methodological quality in their work.
Regarding meta-analysis, eleven protocols cited a potential achievement, if feasible and dependent on the characteristics of the included primary research studies identified. Meta-analyses provide quantitative analyses of individual studies by calculating statistical mean, probabilities, and effect levels [44]. However, they should only be performed if data are presented appropriately [44]. If it is feasible, reviewers should plan how to accomplish it.
GRADE, or a similar approach, was not mentioned in the evaluated protocols. It is an approach that assesses safety and quality, and grades the quality of evidence in a system that considers four levels of: high, moderate, low, and very low [43]. GRADE is a widely accepted and used tool, yet it was not considered in the planned protocols, nor was any other instrument cited to grade the certainty of the evidence. This demonstrates that there was a lack of consideration on the part of the reviewers regarding the strength of their results and its ability to inform policy and practice.
Health decisions should be based on the best scientific evidence. By omitting the tool to grade the level of evidence, from the data of the studies included in a systematic review, patient safety may be put at risk. Decision-making could be influenced by biased studies of low methodological quality [43]. Therefore, researchers should use validated tools in systematic reviews carried out to foster studies with reliable results applicable to clinical practice.
A fundamental and guiding requirement in the PROSPERO platform, in addition to the methodological data of the protocols, is the current status of a systematic review. In this study, the largest number of protocols were ongoing, with two protocols having completed and published the systematic review in a scientific journal. Reviewers are required to commit to updating the progress of the systematic review so that the protocol record reflects the current status, especially when the study is completed [13]. In this way, reviewers will be contributing to scientific development and for other studies to be planned efficiently.
One possible explanation for the low status update of the systematic review protocols in PROSPERO is the abandonment of the systematic review [13]. In this situation, other review teams interested in the topic may avoid duplication of effort [14]. However, this prevents the advancement of science. It may be appropriate for review teams interested in the topic to contact authors to confirm the status of the review and the potential opportunity for resuming an inactive review.
eHealth technologies are health interventions, and therefore require evidence of reliability and validity for their implementation in clinical practice, as they should not compromise patient safety. When methodologically planned and properly recorded, Randomized Controlled Trials (RCTs) or systematic reviews of these studies provide the most reliable quantitative results about the effects of health interventions for informing practice [45].
Systematic reviews are not restricted to RCT study designs, although they are the most commonly reviewed type of study in systematic reviews of interventions of effect. Systematic reviews may consider other study designs, including those with qualitative approaches [45]. In this scan of the literature, only one protocol considered the inclusion of quantitative and qualitative studies, as one of the main objectives was to evaluate the acceptability of mHealth interventions to service users. It is important that authors can transparently integrate quantitative and qualitative data in their studies and conduct an adequate synthesis of the integrated results. Thus, recommending eHealth technology should only be made if these technologies are planned, developed, and validated using high-quality study designs that also evaluate the perspectives of different stakeholder groups for the technology.
The stakeholder’s participation is important to develop effective health solutions focused on real needs. This goes beyond adapting to service users’ expectations, but directly influences the ease, usability, and, consequently, the success of implementing these technologies. Additionally, actively including patients in the development process promotes transparency, builds trust, and ensures solutions are culturally sensitive and ethical [43]. Therefore, from the service users’ perspectives, it is possible to implement more integrated, adaptable, and care-aligned eHealth technologies in the care of pregnant women.
Healthcare providers offer a clinical and healthcare management perspective, which is essential to ensure that technologies are aligned with health practices and protocols, and in compliance with regulatory standards [46]. It is important to evaluate from health professionals’ perspectives the acceptability to use and deliver eHealth technologies. The integration of eHealth into practice may also raise concerns among stakeholders about its viability. According to Feroz et al. [46], stakeholders expressed concerns about malfunction and safety aspects, possible machine inaccuracies, increased clinical workload, and resistance to learning new technologies [46]. This highlights the need to train users and conduct ongoing assessments of them and the technologies used.
Ziebland et al. [47] argue that eHealth applications can have unforeseen or unintended effects on care work. They suggest greater depth and contextualization in the study of eHealth applications and their implications for practice, developing a more precise understanding and more effective implementation of these technologies. This may include organizational, cultural, and social factors that influence whether or not eHealth technologies are accepted. In the research by Frenert et al. [48], from the perspective of nurses, they recognized that the implementation of eHealth applications in practice adds additional tasks to the professional and is invisible work at an organizational level. This represents a barrier to eHealth adoption that needs to be investigated and recognized in the eHealth implementation.
The protocols reviewed herein were aimed at investigating the use of eHealth interventions in a variety of settings: high income countries, as well as LMICs all income country group. It is known maternal morbidity and mortality are higher in LMICs [7]. The causes which have been associated with the quality of care, and access to care [7]. eHealth interventions present an opportunity to communicate and engage with pregnant women and provide easier access to care and support
This study identified several conditions of clinical importance outlined in the protocols. These conditions hold significance for both maternal and fetal outcomes, encompassing aspects such as maternal nutrition, physical activity levels, prenatal educational interventions, gestational diabetes, weight management, mental health, maternal well-being, and smoking cessation.
In terms of eHealth, different typographies were identified when evaluating the protocols extracted on the PROSPERO platform including eHealth-based interventions, mHealth-based interventions, internet-based interventions, telemedicine-based interventions, telehealth-based interventions, and digital health interventions. Turning to literature on defining these typographies suggests that eHealth addresses a broader term of communication and information technologies in the healthcare sector [5], while mHealth is related to the use of mobile technologies in healthcare [49]. Telemedicine is an application of remote medical care through means of telecommunications [50], while telehealth encompasses a broader aspect of telemedicine, also including the educational, administrative, and support sectors [50,51]. Internet-based interventions incorporate health applications delivered over the Internet [52], while digital health interventions are a general category that encompass all digital health-related technologies [53]. Our results suggest that there is interchangeability in the application of terminologies, although in practice they differ in the contexts and aspects of how they are applied in health.
Considering that eHealth technologies identified in the analyzed protocols were directed to pregnant users, except one that also included the partner, the outcomes listed in the protocols related to maternal behavior changes were expected. Much of maternal care during pregnancy related to self-care [54]. Certainly, much of this type of care is not carried out in the presence of a healthcare professional but rather in one’s home, workplace, or other locations. Therefore, effective technologies should be designed to support pregnant women in maternal care across various daily situations. These technologies must be adequately planned and aligned with the specific needs of pregnancy. With only one protocol reporting outcomes related to self-efficacy or self-management measures, there is potential for further research and development in targeted eHealth interventions for maternal health during pregnancy.
The integration of new technologies into the healthcare sector necessitates the presentation of a robust level of evidence, ensuring their safe application and the enhancement of care quality. Assessments of effect constitute an essential component of such analyses [55]. However, the inclusion of cost-effectiveness measures is also important, as they are integral to the evaluation of technologies and represent an indication of their value to maternity services [53].
eHealth technologies present a promising opportunity for healthcare professionals to engage and communicate with women, support health care decision-making, promote good health and well-being, and prevent adverse outcomes for the mother and baby. eHealth technologies are flexible for use in universal health care provision or for targeting groups of women for a particular condition. However, policymakers should be mindful when reviewing evidence of effect sourced from systematic reviews of these interventions by taking into account how accessible the technologies are in their own settings, the level of digital literacy required of users, and their cost. Few of the protocols set out to assess these issues related to transferability, implementation, and sustainability.
Given that our results are based on a limited number of 13 systematic review protocols, the results of such analyses should be cautious regarding generalizability. Mainly because we were interested in any aspect of pregnancy care and not limited to a subgroup of the population or locality. It can be assumed that the small number of protocols identified was due to the single source searched. In addition, eHealth in pregnancy care is a relatively new area of health research expanding globally.
Although it was not the objective of our study, these findings contribute in some way to the creators and coordinators of PROSPERO creating electronic alert systems to monitor protocols with reminders to authors to update information. It is essential that reviewers, in addition to the initial registration of the protocol, record information about the review, whether it was completed, abandoned, or under development, including partial data. Furthermore, they contribute to proposing editorial guidelines to encourage the prospective registration of systematic review protocols.
Our findings also provide specific insights for researchers and policymakers, highlighting specific areas in need of further research and evidence. This can inspire future research and contributions to advancing knowledge at the intersection of eHealth and pregnancy care. Furthermore, these discoveries can significantly contribute to the development of science by revealing weaknesses in protocol reports and records. This insight serves as a guide for researchers who wish to refine their methodologies and promote transparency and accuracy in communicating research results.
Conclusion
We identified systematic review protocols for eHealth, mHealth, telehealth, telemedicine, digital interventions, and internet-based interventions for care during pregnancy. These eHealth technologies were applied across various aspects of maternity care including maternal nutrition, physical activity level, prenatal educational interventions, gestational diabetes, weight management, mental health, maternal health, and smoking cessation. Some reviewers were interested in eHealth technologies aimed at behavioral changes in women, and they were all directed to improve health outcomes for pregnant women. However, there was a limited number of systematic review protocols and the reporting of methodological quality was limited. Researchers should develop and publish further evidence syntheses to assess the effectiveness, cost-effectiveness, acceptability, and feasibility of eHealth interventions in pregnancy care. This call for further research emphasizes the importance of advancing the evidence base and methodological quality in the realm of eHealth for pregnancy care.
Funding Sources
This work was supported by the Brazilian research agency Fundação de Amparo à Pesquisa e Inovação do Estado de Santa Catarina (FAPESC-Foundation for Research and Innovation Support of the State of Santa Catarina), Public Call 12/2022-FAPESC ABROAD-International Cooperation in Science, Technology and Innovation and Bilateral Agreements.
References
- De Paula M, Hofelmann DA (2023) Quality assessment of prenatal and puerperium care. Einstein (São Paulo) 21: eAO0094.
- Gonçalves-Bradley DC, Maria ARJ, Ricci-Cabello I, Villanueva G, Fonhus MS, et al. (2020) Mobile technologies to support healthcare provider to healthcare provider communication and management of care. Cochrane Database Syst Rev 8(8): CD012927.
- Van Den Heuvel JFM, Groenhof TK, Veerbeek JHW, Solinge WWV, Lely AT, et al. (2018) eHealth as the next-generation perinatal care: an overview of the literature. J Med Internet Res 20(6): e202.
- National Library of Medicine.
- Van Der Kleij RMJJ, Kasteleyn MJ, Meijer E, Bonten TN, Houwink EJF, et al. (2019) SERIES: eHealth in primary care. Part 1: concepts, conditions and challenges. Eur J Gen Pract 25(4): 179-189.
- Angarita AM, Cochrane E, Bianco A, Berghella V (2023) Prevention of postpartum hemorrhage in vaginal deliveries. Eur J Obstet Gynecol Reprod Biol 280: 112-119.
- World Health Organization (2019) Trends in maternal mortality 2000 to 2017: estimates by WHO, UNICEF, UNFPA, World Bank Group and the United Nations Population Division, p. 12.
- Patel JJ, Hill A, Lee ZY, Heyland DK, Stoppe C (2022) Critical appraisal of a systematic review: a concise review. Crit Care Med 1(50): 1371-1379.
- Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A et al. (2015) Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev 4(1): 1.
- Page MJ, Shamseer L, Tricco AC (2018) Registration of systematic reviews in PROSPERO: 30,000 records and counting. Syst Rev 7(1): 32.
- Schiavo JH (2019) PROSPERO: An international register of systematic review protocols. Med Ref Serv Q 38(2): 171-180.
- Sideri S, Papageorgiou SN, Eliades T (2018) Registration in the international prospective register of systematic reviews (PROSPERO) of systematic review protocols was associated with increased review quality. J Clin Epidemiol 100: 103-110.
- Andrade R, Pereira R, Weir A, Ardern CL, Espregueira-Mendes J (2019) Zombie reviews taking over the PROSPERO systematic review registry. It’s time to fight back!. Br J Sports Med 53(15): 919-921.
- Rombey T, Doni K, Hoffmann F, Pieper D, Allers K (2020) More systematic reviews were registered in PROSPERO each year, but few records’ status was up-to-date. J Clin Epidemiol 117: 60-67.
- Beresford L, Walker R, Stewart L (2022) Extent and nature of duplication in PROSPERO using COVID-19-related registrations: a retrospective investigation and survey. BMJ Open 12(12): e061862.
- Souza ML, Conceição Junior AR, Bentes CML, Teixeira EP, Bentes MN, et al. (2021) COVID-19: emerging themes from the systematic review records performed in the PROSPERO. Int Jour Develp Res 11(7): 48503-48506.
- Carev M, Civljak M, Puljak L, Dosenovic S (2023) Characteristics, completion and publication of PROSPERO records in regional anesthesia for acute perioperative pain. Jour Comp Effect Res 12(3): e220129.
- Khaleel S, Sathianathen N, Balaji P, Dahm P (2019) Quality of urological systematic reviews registered in PROSPERO. BJU Intern 124(2): 195-196.
- Hu K, Zhao L, Zhou Q, Mei F, Gao Q, et al. (2021) Inconsistencies in study eligibility criteria are common between non-Cochrane systematic reviews and their protocols registered in PROSPERO. Res Synth Methods 12(3): 394-405.
- Garritty CM, Norris SL, Moher D (2017) Developing WHO rapid advice guidelines in the setting of a public health emergency. J Clin Epidemiol 82: 47-60.
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TM, et al. (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372: 71.
- Feroz A, Ali KB, Nuruddin R (2018) Using mobile health to improve maternal nutrition during pregnancy in low-and middle-income countries: a systematic review. PROSPERO CRD42018096511.
- Atkinson C, Levy S (2021) The effect of eHealth delivered interventions on physical activity levels amongst healthy pregnant women: A systematic review. PROSPERO CRD42021235257.
- Kim HK, Chae J (2021) Internet-Based prenatal interventions for maternal health among pregnant women: a systemic review and meta-analysis. Children and Youth Services Review 127.
- Saronga N, Rollo ME, Burrows T, Collins CE, Ashamn AM (2019) mHealth interventions for improving the nutrient intake of pregnant women in low- and lower-middle income economies: a systematic review. Matern Child Nutr 15(2): e12777.
- Overdijkink S, Velu A, Kok M, Schonewille-Rosman A, Steegers-Theunissen R (2016) Evaluating the evidence for the usage of mobile applications in obstetrical healthcare: A systematic review. PROSPERO CRD42016053325.
- Lee S, Leblalta B, Kebali MH (2016) Efficacy and safety of telemedicine in gestational diabetes. PROSPERO CRD42016043009.
- He Y, Huang C, He Q, Liao S, Luo B (2024) Effects of mHealth-based lifestyle interventions on gestational diabetes mellitus in pregnant women with overweight and obesity: systematic review and meta-analysis. JMIR Mhealth Uhealth 12: e49373.
- Silang K, Sanguino H, Sohal PR, Rioux C, Tomfohr-Madsen L, et al. (2021) eHealth interventions for mental health and substance use in pregnancy: a systematic review and meta-analysis. Int J Environ Res Public Health 18(19): 9952.
- Rahman O, Yamaji N, Suzuki D, Sasayamaet k, Yoneoka D, et al. (2022) Technology-based innovative healthcare solutions for improving maternal and child health outcomes in low-and middle-income countries: A systematic review and network meta-analysis. 8(1): e40954.
- Rhodes A, Smith A, Chadwick P, Llewellyn CH, Croker H (2020) Effectiveness and components of exclusively digital health interventions targeting diet, physical activity and weight gain in pregnant women: a systematic review and meta-analysis 8(7): e18255.
- Griffiths S, Parsons J, Naughton F, Fulton E, Tombor I, et al. (2016) Are digital interventions for smoking cessation in pregnancy effective? A systematic review. PROSPERO CRD42016036201 5: 207.
- Hussain T, Smith P, Yee LM (2020) Mobile phone-based behavioral interventions in pregnancy to promote maternal and fetal health in high-income countries: systematic review. JMIR Mhealth Uhealth 8(5): e15111.
- Lewkowitz AK, Whelan Ar, Ayala Nk, Hardi A, Stoll C, et al. (2024) The effect of digital health interventions on postpartum depression or anxiety: a systematic review and meta-analysis of randomized controlled trials. Am J Obstet Gynecol 230(1): 12-43.
- Denicola N, Grossman D, Marko K, Sonalkar S, Tobah YSB, et al. (2020) Telehealth interventions to improve obstetric and gynecologic health outcomes a systematic review. Obstet Gynecol 135(2): 371-382.
- World Bank Open Data. Free and open access to global development data. The World Bank.
- Jaffe K, Ter Horst E, Gunn Laura H, Zambrano JD, Molina G (2020) A network analysis of research productivity by country, discipline, and wealth. PLoS One 15(5): e0232458.
- Rubagumya F, Hopman WM, Gyawali B, Mukherji D, Hammad N et al. (2022) Participation of lower and upper middle-income countries in clinical trials led by high-income countries. JAMA Netw Open 5(8): e2227252.
- Siddaway AP, Wood AM, Hedges LV (2019) How to do a systematic review: a best practice guide for conducting and reporting narrative reviews, meta-analyses, and meta-syntheses. Annu Rev Psychol 70: 747-770.
- Higgins J, Thomas J, Chandler J, Cumpston M, Li T, et al. (2023) Cochrane handbook for systematic reviews of interventions version 6.4.
- Rørstad K, Aksnes DW, Piro FN (2021) Generational differences in international research collaboration: A bibliometric study of Norwegian University staff. PLoS One 16(11): e0260239.
- Schwartzman S (2022) Pesquisa e Pós-Graduação no Brasil: duas faces da mesma moeda? Estudos Avançados 36(104): 227-254.
- Schunemann HJ, Cuello C, Akl EA, Mustafa RA, Meerpohl JJ, et al. (2019) GRADE guidelines: 18. How ROBINS-I and other tools to assess risk of bias in nonrandomized studies should be used to rate the certainty of a body of evidence. J Clin Epidemiol 111: 105-114.
- Scheidt S, Vavken P, Jacobs C, Koob S, Cucchi D et al. (2019) Systematic reviews and meta-analyses. Z Orthop Unfall 157(4): 392-399.
- Munn Z, Stern C, Aromataris E, Lockwood C, Jordan Z (2018) What kind of systematic review should I conduct? A proposed typology and guidance for systematic reviewers in the medical and health sciences. BMC Med Res Methodol 18(1): 5.
- Feroz AS, De Vera K, Bragagnolo ND, Saleem S, Bhutta Z et al. (2022) Understanding the needs of a mobile phone–based telemonitoring program for pregnant women at high risk for pre-eclampsia: interpretive qualitative description study. JMIR Form Res 6(2): e32428.
- Ziebland S, Hyde E, Powell J (2021) Power, paradox and pessimism: On the unintended consequences of digital health technologies in primary care. Social Science & Medicine 289: 114419.
- Frenert S, Petersson L, Erlingsdottir G (2023) “More” work for nurses: the ironies of eHealth. BMC Health Serv Res 23(1): 411.
- Istepanian RSH (2022) Mobile health (m-health) in retrospect: the known unknowns. Int J Environ Res Public Health 19(7): 3747.
- Gajarawala SN, Pelkoski JN (2021) Telehealth Benefits and Barriers. J Nurse Pract 17(2): 218-221.
- Beheshti L, Kalankesh LR, Doshmangir L, Farahbakhsh M (2022) Telehealth in primary health care: a scoping review of the literature. Perspect Health Inf Manag 19(1): 1n.
- Mu TY, Li YH, Xu RX, Chen J, Wang YY, et al. (2021) Internet-based interventions for postpartum depression: A systematic review and meta-analysis. Nursing Open 8(3): 1125-1134.
- Gentili A, Failla G, Melnyk A, Puleo V, Di Tanna GL et al. (2022) The cost-effectiveness of digital health interventions: A systematic review of the literature. Front Public Health 10: 787135.
- Schmidt-Hantke J, Jacobi C (2023) Investigating perspectives on e-health interventions to enhance maternal mental well-being: Results of a stakeholder interview. PLOS Digit Health 2(8): e0000326.
- Burches E, Burches M (2020) Efficacy, effectiveness and efficiency in the health care: the need for an agreement to clarify its meaning. Int Arch Public Health Community Med 4(1): 1-3.
© 2024 Stefhanie Conceição de Jesus. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






