- Submissions

Full Text
Investigations in Gynecology Research & Womens Health
The Human Papillomavirus Vaccine: The Past, the Present, and the Future
Samantha Docos BS*, Sam Nguyen MD, Rehan Feroz DO and Michael Roche MD
Department of Gynecology, University Drive Hershey, USA
*Corresponding author: Samantha Docos BS, Professor of Gynecology, University Drive Hershey, PA 17033, USA, Tel: 717 283 7796; Email: swargner2@pennstatehealth.psu.edu
Submission: February 12, 2018;Published: April 06, 2018

ISSN: 2577-2015
Volume1 Issue5
History
The development of the Human Papillomavirus (HPV) vaccine is one of the great public health accomplishments of the 21st century. The roots of this success can be traced back thousands of years. Ancient Greeks and Romans made the first tentative steps in this journey by identifying genital warts as a sexually transmitted disease [1]. It would not be until 1907, however, with the novel field of microbiology entering full swing that Ciuffo would confirm the infectious nature of genital warts [2]. Viral particles were confirmed as the infectious vector in the 1940s [3]. HPV was first described as a dsDNA virus in 1965 [4]. Following this description, the large phylogeny of HPV was rapidly expanded to today where over 175 genotypes have been identified [5] with the pace of discovery accelerating; it wasn’t long before the connection with cervical cancer was drawn.
Since Hippocrates first noted the, at the time, incurable nature of cervical cancer, physicians had wrestled with this debilitating and deadly disease. Rigoni-Stern [6], a 19th century Italian scientist, noted cervical cancer predominated in married women, widows and prostitutes and rarely in virgins and nuns. With this correlation he proposed relationship of cervical cancer to sexual contact. Scientists in the 1950s initially suspected smegma as the culprit [7]. In the 1960s cases reports of genital warts progressing to cervical cancer were reported. This led to Zur Hausen [8] suggesting an association between HPV and cervical cancer in 1974. Less than a decade later, in 1982, HPV was isolated in cases of cervical cancer [9]. HPV was therefore the second virus (and first in humans) demonstrated to be a vector for malignancy [10]. Such a medical breakthrough led to recognition and acclaim that included the 2008 Nobel prize.
Pathogenesis
Viral structure
Human papillomaviruses, belonging to the family Papillomaviridae, are non-enveloped viruses with an icosahedral capsid containing double stranded DNA [11]. There are more than 200 HPV types that exist and they are divided into five genera (alpha, beta, gamma, mu, and nu) [12]. All of the high-risk HPV types belong to the alpha genera and infect mucosa. Beta and gamma HPV types infect cutaneous epithelium and Beta HPV types are part of the normal human virome present at birth [12,13]. The genome is composed of three main regions: the early coded region, late coded region, and non-coding control region. The early coded region contains E1, E2, E4-E7 [12] and encodes viral proteins that have regulatory functions in the infected epithelial cell [13]. The late coded region contains L1 and L2 [12] and encodes the two viral structural proteins that form the icosahedral capsid. Each type of HPV differs by at least 10% of its genotype in the highly conserved L1 region of the gene [13].
Cell entry and replication
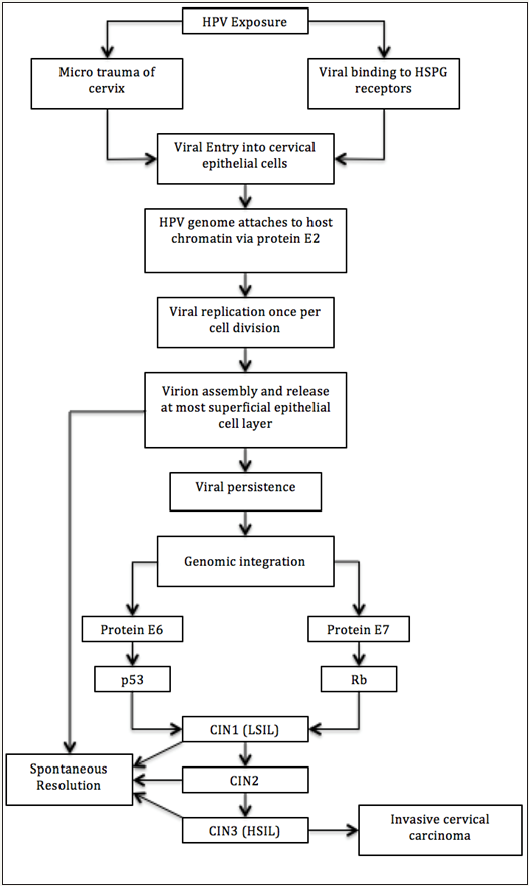
HPV can infect basal cervical epithelial cells one of two ways, via micro-trauma or binding to heparin sulfate proteoglycan (HSPG) receptors [13]. Micro-trauma occurs during sexual intercourse, vaginal delivery, or other sexually transmitted infections. The other way for HPV to infect cells is specific to high-risk HPV types, especially HPV-16. HPV-16 can bind to HSPG receptors directly on the cells of the squamous cellular junction between the endocervix and the ectocervix [13]. HPV can only replicate in diving cells that are undergoing maturation, senescence, and cellular death [12]. The cells which are actively dividing are the stem cells, located at the most basal layer of the epithelium. If HPV infects a parabasal cell that is not actively dividing then the infection has a greater chance of being only transient [14]. HPV attaches its genome to the host cell chromatin via the E2 protein from the early coded region. The virus replicates in all layers of the epithelium in a steady state fashion of one replication per cell division [14]. It is not until the most superficial layer that the virions are assembled and released [12] (Figure 1).
Figure 1: The evolution of HPV exposure to invasive cervical carcinoma or spontaneous resolution.
Cervical disease
Cervical disease can be classified in one of two ways. The first classification system contains three stages and is termed cervical intraepithelial neoplasia (CIN). The second classification system contains two stages and is termed squamous intraepithelial lesion (SIL) [13]. CIN 1, is mild dysplasia (“Weekly epidemiological record,” 2017) thought to represent a transient infection with a low probability of progressing to cervical cancer [12,13]. Approximately 80-90% of CIN1 cases regress spontaneously [13]. CIN2 is moderate to marked dysplasia (“Weekly epidemiological record,” 2017) and is considered premalignant but it is possible that it can be caused by either carcinogenic or non-carcinogenic HPV types [12]. CIN3 is severe dysplasia or carcinoma in situ (“Weekly epidemiological record,” 2017). This level of dysplasia is always caused by carcinogenic HPV types and has the same risk factors as cervical cancer [12]. Only 60% of CIN3 regress spontaneously [13]. As noted previously transient infections are generally a result of HPV infecting cells that are no longer dividing. This is consistent with what is seen between low grade CIN lesions and high grade ones. What is seen is that the low grade lesions express E6 and E7 in cells that have withdrawn from the cell cycle while high grade lesions express E6 and E7 in the proliferating cell compartment [14]. Also, CIN3 is also more likely than CIN1 or CIN2 to exhibit HPV genome integration [12].
Carcinogenesis
HPV is the most common sexually transmitted infection and the most common viral infection of the reproductive tract [12]. The overall lifetime risk of becoming infected with HPV via sexual transmission is 50-80% [12,13]. The highest incidence of infection occurs between the ages of 20-25 during an individual’s first sexual encounter [13]. Fortunately, a majority (70-90%) of infections is asymptomatic and resolve spontaneously within 1-2 years [12]. If the infection is not discovered and allowed to persist without treatment, the interval of time from HPV exposure to invasive carcinoma is about 20 years [11].
The transition from infection to cervical cancer occurs in a predictable order of events. It is thought that genomic integration is the key event leading to transformation from HPV infection to cancer [12]. One reason being that genomic integration can lead to genomic instability and thus tumor formation. E6 and E7 are responsible for inhibiting tumor suppressor pathway. Therefore, if they are more abundant, then the tumor suppression is released and carcinogenesis is more likely to occur. E6 is specifically responsible for targeting p53 via aubiquination-mediated pathway and degrading it. E7 is responsible for inhibiting the retinoblastoma protein, which leads to increased expression of S-phase protein, reentry into the cell cycle, and continued initiation of DNA synthesis [12]. Normally when unexpected cell proliferation occurs, the cell responds by undergoing apoptosis. Therefore the E7 activity would be expected to cause apoptosis. To counteract this E6 protein inhibits apoptosis specifically [13].
In addition to mechanisms created to enhance cellular proliferation and inhibit apoptosis, other mutations exist to help promote immortalization of malignant epithelial cells. One of the important steps to immortalization is abnormal growth of telomeres. In cervical cancer, E6 can activate telomerase leading to stabilization and even lengthening of telomeres. This allows the cells to continue to divide. Other mutations that help promote cancer include one set of mutations that allow the cells to break through the basement membrane and another that allows cells to move into the dermis. Together these mutations promote invasion [14].
Malignancy
Epidemiology
HPV accounts for more than 5% of all human cancers, [13] more than 50% of infectious cancers in women and 5% of infectious cancers in men [12]. In terms of the specific cancers it causes, HPV is estimated to be responsible for 91-100% of cervical cancers, 88-93% of anal cancers, 70-78% of vaginal cancers, 50-63% of penile cancers, and 15-69% of vulvar cancers. It is more difficult to determine for oropharyngeal cancer due to tobacco and alcohol use but it is estimated to be 26-72% [15]. In 2012 there were 630,000 new cases of HPV related cancers in women and 530,000 of them (84%) were cervical cancer. This caused approximately 266,000 deaths worldwide which is 8% of cancer death in women for that year [11].
Most cancer registries worldwide report that the incidence of cervical cancer rises between the ages of 20-40 then plateaus or continues to increase smoothly beyond that age. It is important to note that the age specific incidence reported in cancer registries is highly influenced by the screening programs in that country [14].
Association between HPV and Cervical cancer
Squamous cell carcinoma found positive for HPV DNA is caused by about 15 high risk HPV types (most notably type 16,18,45,31,33,52, and 58) approximately 90-95% of the time [11]. In terms of cervical cancer specifically, HPV 16 accounts for approximately 50-55% of cases and HPV 18 causes about 10-15% of cases with the remaining cases being caused by other high risk HPV types [12,13]. Multiple studies have consistently found that HPV DNA is found in cervical cancer specimens 90-100% of the time [14]. Findings such as these have lead to HPV becoming the first ever identified “necessary cause” of a human cancer [14]. This association between HPV and cervical cancer is one of the strongest associations ever seen in a human cancer. For example, the strength of association (relative risk/odds ratio) between different risk factors and the human cancer they cause is 500 for HPV DNA 18 and cervical adenocarcinoma in the Philippines, Costa Rica, and Bangkok, but only 50 for HBsAg and liver cancer in Greece, HCV and liver cancer in Italy, and only 10 for cigarette smoking and lung cancer [14].
Screening
The strength of association found between HPV and cervical cancer has led to efforts to improve screening programs and develop HPV vaccines. The United States Preventative Task Force (USPSTF) recommends that all women begin cervical cancer screening when they turn 21 [12]. From age 21 to 30 the recommendation is to undergo a pap smear to look at cytology every 3 years. From the age of 30 to 65 the recommendation is to continue with the cytology every 3 years or that can be replaced with co-testing with cytology via pap smear and HPV DNA detection every 5 years [12]. In addition to extending the screening interval, HPV DNA testing can also be useful when cytology comes back as atypical squamous cells of undetermined significance [12,14]. It is important to note that HPV testing has lower specificity in younger women whereas the specificity increases in women greater than 30 years old and tends to be similar to cytology [14].
Screening is important to try and prevent the progression of HPV infection and cellular dysplasia to carcinoma. When pre invasive disease of the cervix is detected then ablative methods such as burning or freezing, also known as cryotherapy, or surgical removal via a loop electrosurgical excision procedure (LEEP) or cone biopsy should be performed [11].
Vaccine Development
It was not long after the identification of HPV as the instigator for cervical cancer that efforts turned towards developing a vaccine. Animal studies in the 1980s and 90s demonstrated excellent protection in various host species [16]. Following this success Merck and GSK undertook nearly simultaneous development of HPV vaccines. Merck focused on a quadrivalent vaccine while GSK developed a bivalent version. Both vaccines were developed using virus-like particles that mimic the L1 epitope [17]. This epitope induced a high antibody response making it an ideal candidate for vaccine trials [18]. An additional advantage of these vaccine designs is that by utilizing virus-like particles they are noninfectious.
Initial phase II trials were highly encouraging, demonstrating 100% protection from cervical dysplasia due to protection against subtypes [19-23]. With these results in hand, multiple international phase III trials were started. When results were reported, these studies also demonstrated greater than 90% efficacy in all subgroups analyzed [24-26]. Following publication of this data, Merck was the first to obtain licensure from the FDA in an expedited manner in 2006, and GSK followed closely behind.
Vaccine Implementation
Global vaccine coverage
By October of 2014, 64 countries nationally, four countries sub national, and 12 overseas territories had introduced the HPV vaccine into their national immunization program. A majority of the settings were high income and upper middle income. They found that from 2006 to 2014 about 47 million women received the full course of vaccine and that about 59 million women received at least one dose. This represents 39.7% and 50.1% of the targeted females respectively and 1.4% and 1.7% of the total female population [27]. Unfortunately the populations that still do not have access to the HPV vaccine are the populations that carry most of the burden of cervical cancer worldwide. Access to the HPV vaccine in those low and low-middle income countries is almost non-existent. The HPV vaccine has hardly introduced outside of high income counties. The middle-income countries that it was introduced into were the pacific countries, South Africa, Libya, Seychelles, Malaysia, some Kazakhstan regions, Macedonia, and Bulgaria. The only three GAVIeligible countries were Bhutan, Lesotho, and Rwanda [28]. Efforts should be focused on introducing the vaccine into these low and middle income countries since screening is scarce and vaccination is the only other feasible option for prevention. GAVI is the Vaccine Alliance, which supports projects in developing countries to provide vaccines. Unfortunately only low income countries and 40% of lower middle income countries meet eligibility for GAVI [27]. When trying to fill the gaps, school based programs may be the answer since they have been reported to achieve the highest coverage [27]. Other than accessibility to the vaccine, there are other factors affecting vaccine uptake that will take time to change [29]. For example, social norms and values related to sexual activity and trust in vaccination programs and health care providers [30]. According to the World Health Organization (WHO), by March 31, 2017, 71 countries (37%) had introduced HPV vaccine into their national immunization program for girls and 11 countries (6%) also for boys which are an increase from October 2014 [11].
Implementation has been only moderately more effective in the United States. The annual National Immunization Survey for Teens was performed on 20,475 adolescents (9661 females and 10814 males) aged 13-17 years old [31]. It showed that from 2015 to 2016 there was an increase in the percentage of individuals receiving at least one dose of the HPV vaccine from 56.1% to 60.4%. The estimated coverage varies from state to state. For example there are 11 states with < or equal to 55% coverage and 9 states plus Washington DC that have > or equal to 72% coverage. All other states fall somewhere in between. Interestingly, the initiation of HPV vaccine was 16% lower among adolescents living outside of central cities compared to those living in central cities [31,32].
American cancer society recommendations
According to the American Cancer Society, about 28,500 cases of cancer could be prevented each year by HPV vaccination in the United States [15]. Therefore, clinicians should all strongly recommend for their patients to be vaccinated against HPV at the age of 11-12, trying their best to complete the series by their 13th birthday [15]. This recommendation is due to providing the greatest effectiveness. The ACS agreed with 2015 ACIP (advisory committee on immunization practices) recommendation which was:
“Females and males: routine vaccination with 3-dose series at age 11 to 12 (series can be started beginning at age 9). Vaccination recommended for females aged 13-26 and for males aged 13- 21 who have not been vaccinated previously or who have not completed the 3 dose series.
Males age 22-26 may be vaccinated. Vaccination recommended through age 26 y for men who have sex with men and for person who are immune-compromised, including those with HIV infection” [15].
In October 2016, the ACIP updated its recommendation, which the ACS adopted. The updated recommendation is as follows:
“For persons initiating vaccination before the 15th birthday, the recommended immunization schedule is 2 doses of HPV vaccine. The second dose should be administered 6 to 12 months after the first dose (0, 6-12-month schedule)” [33].
World Health Organization recommendation
The World Health Organization released an updated position paper on the HPV vaccination on 12 May 2017. The WHO recommends that for successful implementation there are a variety is approaches and steps that can be taken. These include “focus on education about reducing behaviors that increase the risk of acquiring HPV, training of health workers and information to women about screening, diagnosis, and treatment of precancerous lesions and cancer. The strategy should also include increased access to quality screening and treatment services and to treatment of invasive cancers and palliative care” [11]. The primary population should be girls age 9-14 years old prior to becoming sexually active and the secondary target population should be females 15 years or older and males [11]. This secondary population should only be targeted if it is feasible, affordable, cost-effective, and does not interfere with providing the vaccine to the primary target population. The WHO recommended vaccination schedule:
“A 2 dose schedule with adequate spacing between the first and second dose in those aged 9-14 years. A 2 dose schedule with a 6 month interval between doses for individuals receiving first dose before 15. Those aged 15 or greater at the time of the second dose are also covered by 2 doses. A 3 dose schedule (0,1-2, 6 months) should be used for individuals 15 or greater and also for those younger than 15 known to be immune-compromised and/or HIV infected” [11]. Other important information to be aware of is that the HPV vaccine can be administered at the same time as other non-live and live vaccines by using separate syringes and injection sites [11]. Also, the vaccine is safe to provide to individuals who are immune-compromised and/or have an HIV infection. The data in pregnant women is limited so that vaccine should be avoided in pregnant women [11]. They also recommend studying the effectiveness of a 1-dose schedule and the safety of administering the vaccine in children less than 9 years old [11].
The center for disease control recommendation
The Center for Disease Control (CDC) recommendations are consist with the ACS and WHO recommendations. They recommend routine vaccination with the HPV vaccine between ages 11 and 12 with 9 years old being the earliest that it can be given. The Advisory Committee on Immunization Practices (ACIP) also recommends vaccinating females age 13 to 26 and males age 13 to 21 if they are not already immunized. For men who have sex with men, transgender, and immune-compromised the recommendation is to vaccinate until the age of 26. In terms of dosing, two doses are required for individuals starting the vaccine series before they turn fifteen with at least five months between the two doses. If there are less than 5 months between the two doses then the individual needs to receive three doses. Three doses are also required for individuals starting the series after their fifteenth birthday as well as those that are immune-compromised. The three doses should be administered at 0, 1-2, and 6 months [34].
References
- Burns DA (1992) “Warts and all”- the history and folklore of warts: a review. J R Soc Med 85(1): 37-40.
- Zur Hausen H (2009) Papillomaviruses in the causation of human cancers- a brief historical account. Virology 384(2): 260-265.
- Strauss MJ, Shaw EW, Bunting H, Melnick JL (1949) Crystalline Virus- Like Particles from Skin Papillomas Characterized by Intranuclear Inclusion Bodies. Proc Soc Exp Biol Med 72(1): 46-50.
- Crawford LV (1965) A study of human papilloma virus DNA. J Mol Biol 13(2): 362-372.
- Bernard HU, Burk RD, Chen Z, van Doorslaer K, Hausen H zur, et al. (2010) Classification of papillomaviruses (PVs) based on 189 PV types and proposal of taxonomic amendments. Virology 401(1): 70-79.
- Scotto J, Bailar JC (1969) Rigoni-Stern and medical statistics. A nineteenth-century approach to cancer research. J Hist Med Allied Sci 24(1): 65-75.
- Van Howe RS, Hodges FM (2006) The carcinogenicity of smegma: Debunking a myth. Journal J Eur Acad Dermatol Venereol 20(9): 1046- 1054.
- Hausen H Zur (1976) Condylomata Acuminata and Human Genital Cancer. Cancer Res 36(2): 794.
- Gissmann L, Diehl V, Schultz-Coulon HJ, zur Hausen H (1982) Molecular cloning and characterization of human papilloma virus DNA derived from a laryngeal papilloma. J Virol 44(1): 393-400.
- Coffin JM, Hughes SH, Varmus HE (1997) The Place of Retroviruses in Biology. Retroviruses.
- (2017) Weekly epidemiological record Relevé épidémiologique hebdomadaire. 92:241-268.
- Mui UN, Haley C, Tyring SK (2017) Viral Oncology: Molecular Biology and Pathogenesis. J Clin Med 6(12). pii: 111.
- Graham SV (2017) The human papillomavirus replication cycle, and its links to cancer progression: a comprehensive review. Clin Sci (Lond) 131: 2201-2221.
- Bosch FX, Lorincz A, Muñoz N, Meijer CJLM, Shah KV (2002) The causal relation between human papillomavirus and cervical cancer. J Clin Pathol 55(4): 244-265.
- Saslow D, Andrews KS, Manassaram-Baptiste D, Loomer L, Lam KE et al. (2016) Human papillomavirus vaccination guideline update: American Cancer Society guideline endorsement. CA Cancer J Clin 66(5): 375-385.
- Schiller JT, Lowy DR (1996) Papillomavirus-like particles and HPV vaccine development. Semin Cancer Biol 7(6): 373-382.
- Kirnbauer R, Booy F, Cheng N, Lowy DR, Schiller JT (1992) Papillomavirus L1 major capsid protein self-assembles into virus-like particles that are highly immunogenic. Proc Natl Acad Sci U S A. 89(24): 12180-12184.
- Bachmann MF, Hoffmann Rohrer U, Kündig TM, Bürki K, Hengartner H, et al.(1993) The Influence of Antigen Organization on B Cell Responsiveness. Source Sci New Ser 262(5138): 1448-1451.
- Villa LL, Costa RLR, Petta CA, Andrade RP, Ault KA, et al. (2005) Prophylactic quadrivalent human papillomavirus (types 6, 11, 16, and 18) L1 virus-like particle vaccine in young women: a randomised double-blind placebo-controlled multicentre phase II efficacy trial. Lancet Oncol 6(5): 271-278.
- Harper DM, Franco EL, Wheeler CM, Moscicki AB, Romanowski B, et al. (2006) Sustained efficacy up to 4.5 years of a bivalent L1 virus-like particle vaccine against human papillomavirus types 16 and 18: followup from a randomised control trial. Lancet 367(9518): 1247-1255.
- Harper DM, Franco EL, Wheeler C, Ferris DG, Jenkins D, et al. (2004) Efficacy of a bivalent L1 virus-like particle vaccine in prevention of infection with human papillomavirus types 16 and 18 in young women: A randomised controlled trial. Lancet 364(9447): 1757-1765.
- Mao C, Koutsky LA, Ault KA, Wheeler CM, Brown DR et al. (2006) Efficacy of human papillomavirus-16 vaccine to prevent cervical intraepithelial neoplasia: a randomized controlled trial. Obstet Gynecol 107(1): 18-27.
- Koutsky LA, Ault KA, Wheeler CM, Brown DR, Barr E, et al. (2002) A controlled trial of a human papillomavirus type 16 vaccine. N Engl J Med 347(21): 1645-1651.
- Paavonen J, Jenkins D, Bosch FX, Naud P, Salmerón J, et al. (2007) Efficacy of a prophylactic adjuvanted bivalent L1 virus-like-particle vaccine against infection with human papillomavirus types 16 and 18 in young women: an interim analysis of a phase III double-blind, randomised controlled trial. Lancet 369(9580): 2161-2170.
- Joura EA, Leodolter S, Hernandez-Avila M, Wheeler CM, Perez G, et al. (2007) Efficacy of a quadrivalent prophylactic human papillomavirus (types 6,11,16,and18) L1 virus-like-particle vaccine against high-grade vulval and vaginal lesions: a combined analysis of three randomised clinical trials. Lancet 369(9574): 1693-1702.
- The FUTURE II Study Group (2007) Quadrivalent vaccine against human papillomavirus to prevent high-grade cervical lesions. N Engl J Med 356(19): 1915-1927.
- Bruni L, Diaz M, Barrionuevo-Rosas L, Herrero R, Bray F, et al. (2016) Global estimates of human papillomavirus vaccination coverage by region and income level: a pooled analysis. Lancet Glob Health 4(7): e453-463.
- Perlman S, Wamai RG, Bain PA, Welty T, Welty E, et al. (2014) Knowledge and awareness of HPV vaccine and acceptability to vaccinate in sub- Saharan Africa: a systematic review. PLoS One 9(3): e90912.
- Marlow LAV (2011) HPV vaccination among ethnic minorities in the UK: Knowledge, acceptability and attitudes. Br J Cancer 105(4): 486-492.
- Wigle J, Coast E, Watson-Jones D (2013) Human papillomavirus (HPV) vaccine implementation in low and middle-income countries (LMICs): Health system experiences and prospects. Vaccine 31(37): 3811-3817.
- Walker TY, Elam-Evans LD, Singleton JA, Yankey D, Markowitz LE, et al. (2017) National, Regional, State, and Selected Local Area Vaccination Coverage Among Adolescents Aged 13-17 Years-United States, 2016. MMWR Morb Mortal Wkly Rep 66(33): 874-882.
- Daley EM, Vamos CA, Thompson EL, Gregory D Zimet, Zeev Rosberger, et al. (2017) The feminization of HPV: How science, politics, economics and gender norms shaped U.S. HPV vaccine implementation. Papillomavirus Res 3: 142-148.
- Smith RA, Andrews KS, Brooks D, Fedewa SA, Manassaram Baptiste D, et al. (2017) Cancer screening in the United States, 2017: A review of current American Cancer Society guidelines and current issues in cancer screening. CA Cancer J Clin 67(2): 100-121.
- HPV Vaccine Recommendations | Human Papillomavirus | CDC.
© 2018 Samantha Docos BS. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






