- Submissions

Full Text
Global Journal of Endocrinological Metabolism
Clinical Correlates of Obesity: A Cross-Sectional Survey Among Adolescents in North-Western Nigeria
EseigbeP1*, Onuoha FM2, Moses LA3 and Ibrahim BY4
1Department of Family Medicine, Bingham University Teaching Hospital, Nigeria
2Department of Family Medicine, Federal Medical Centre, Nigeria
3Department of Family Medicine, National Hospital, Nigeria
4Department of Family Medicine, Ahmadu Bello University Teaching Hospital, Nigeria
*Corresponding author: Eseigbe Patricia, Department of Family Medicine, Nigeria
Submission: August 03, 2020; Published: September 08, 2020

ISSN 2637-8019Volume3 Issue1
Abstract
Background: Adolescents constitute a huge proportion of the world’s population. Adolescent obesity has globally increased to significant levels with serious public health consequences. The epidemic of obesity has major complications for health care now and in the future. Obesity is a major risk factor for a number of chronic medical conditions. As the obese adolescent grows into an obese adult, the morbidity and mortality associated with obesity occur with an increased risk.
Methods: This was a cross-sectional school –based study conducted in Kaduna, North-west Nigeria. The secondary schools were selected via simple random sampling and the participants were recruited using systematic sampling method. An interviewer-administered questionnaire was used to collect the participants’ demographics and their height, weight, and blood pressures were measured. Furthermore, their blood samples were taken for blood glucose and lipid profile estimation. Data analysis was done using SPSS version 20 and a p-value of <0.05 was regarded as statistically significant.
Result: A total of 150 adolescents participated in the study. The prevalence of obesity was 14.0% and overweight was 21.3%. Only 1.3% of the participants had diastolic hypertension, 12% had hyperglycemia and 8.7% had hyperlipidemia. There was a statistically significant association between obesity and hyperlipidemia (x2 = 9.23, p = 0.03).
Conclusion: Adolescent obesity poses greater risk for the development of some major chronic disorders. Prompt management of obesity in adolescence is key to curtailing the menace of obesity for the present and future health of the populace.
Keywords: Adolescents; Obesity; Blood pressure; Blood glucose; Blood lipids
Introduction
Adolescence is a crucial transit period of development from childhood to adulthood [1,2]. This period is characterized by rapid changes in body structure with physiologic and social functioning. Adolescents refer to persons between the ages of 10 and 19 years [2,3]. A huge proportion of the world’s population is adolescents. One in every five persons in the world is an adolescent [3]. Many chronic diseases in adulthood have their roots in adolescence [3]. Nearly two thirds of premature deaths and one third of the total disease burden in adulthood is associated with conditions or behaviors that began in adolescence [3].
Obesity is a state of excess adipose tissue mass or body fat [4,5]. Obesity in adolescents has risen to significant levels globally with serious public health consequences [6]. The WHO considers adolescent obesity to be a global epidemic [7,8]. Globally, obesity and its complications cause as many as 300,000 premature deaths each year, making it second to cigarette smoking as a cause of death [4]. Adolescent obesity is a strong predictor of adult obesity [4,9]. If an adolescent is obese, the likelihood that he/she would grow into an obese adult is 70% [10]. As an obese adolescent develops into an obese adult, the morbidity and mortality associated with obesity occur with an increased risk [6]. Obesity is a major risk factor for a number of chronic non-communicable conditions such as insulin resistance, hypertension, cardiovascular disease, type-2 diabetes mellitus, dyslipidemias, and some cancers [1,4,7-9]. These chronic conditions could lead to reduced quality of life and premature death [11].
Adolescent obesity is not a transient developmental phenomenon but can be a determinant of some cardiovascular disorders in adulthood [1,6]. The Harvard Growth Study found that boys who were overweight during adolescence were twice as likely to die from cardiovascular disease as those who had normal weight [1]. In addition, the Framingham prospective community study reported that relative body weight, weight change, and skinfold thickness were all directly related to blood pressure and the subsequent development of hypertension [12]. Other cardiovascular diseases include cardiomyopathies and heart failure due to increased workload of the heart [8].
Insulin resistance and the development of type-2 diabetes mellitus has also been associated with obesity [8,13,14]. Traditionally, type-2 diabetes had been a disease of adults, however, this now occurs in increased numbers among obese adolescents [6]. As body weight increases, insulin resistance and compensatory insulin secretion also increases to a point where the body’s ability to secrete the required insulin is impaired, with a resultant rise in blood glucose [8]. Obesity is also associated with dyslipidemia in the form of elevated triglyceride level, reduced high density lipoprotein (HDL) cholesterol, and an increase in atherogenic, low density lipoprotein (LDL) particles [1].
Other complications of obesity include metabolic syndrome, fatty liver and cirrhosis, gall-bladder disease, obstructive sleep apnea, skin diseases, and osteoarthritis. There are some psychosocial problems including depression that are also associated with obesity, which may be due to prejudice, discrimination, poor body image, and low self-esteem [4,5,7].
Materials and Methods
It was a cross-sectional study conducted in secondary schools in Kaduna metropolis when schools were in session between the months of February and March 2014. The sample size was calculated to be 150 using the formula for cross-sectional studies. The schools were selected using simple random sampling while the study participants were selected via a systematic sampling technique. After obtaining the permission from the principal of a school and the accent/consent from the selected participants’ parents, the data collection process was for two days. The first day was to further educate the participants on the study protocol and to instruct them not to eat breakfast the following day, which was the day for sample-collection. The participants were recruited in the morning immediately after the general assembly to avoid over-fasting. Their demographic data was collected using structured interviewer-administered questionnaires.
The participants’ heights (in meters) and weights (in kilograms) were obtained using standard measuring methods. Their body mass index (BMI) was calculated using the standard formula: BMI=weight (kg) / height2 (m2). The BMI was then charted with the scale on the BMI- for- age percentile. Normal BMI-for-age was between the 5th and 84th percentile, overweight was between the 85th and 94th and obesity was BMI- for- age at or above the 95th percentile [1,4].
Their blood pressure was measured with the participants sitting, using appropriately sized blood pressure cuff for the sphygmomanometer, and recorded in mmHg. The mercury sphygmomanometer used was Accoson, UK brand. The blood pressure was taken after the participants had rested for 6 minutes. The average of three blood pressure readings taken at an interval of 5 minutes was calculated as the blood pressure value. These values were charted against the blood pressure- for- age percentile. A systolic and/or diastolic BP percentile between the 90th and 95th percentile was pre-hypertension while percentile at or above the 95th mark was recorded as hypertension [1,15]. Their blood samples were collected for glucose and lipid levels. Fasting blood glucose in mg/dl was estimated using a Fine- test Autocoding™ glucometer manufactured by Info-pia Co. Ltd, Korea (2013). Values for fasting glucose less than 110mg/dl were normal and values at or greater than 110mg/dl were regarded as hyperglycaemia [1,16]. The fasting lipid profile in mg/dl was estimated using a rapid diagnostic test kit LipidPro™ machine manufactured by Info-pia Co. Ltd, Korea (2013). Values for LDL below 110mg/dl were normal and those at or greater than 110mg/dl were termed hyperlipidaemia [1,16].
Analysis of the data was done using SPSS version 21. Descriptive analysis and chi-square tests were done with a p-value of <0.05 regarded as statistically significant. Ethical approval to conduct this study was obtained from the Ethical Committee of the Ahmadu Bello University Teaching Hospital Kaduna and the Kaduna State Ministry of Health, and the permission to carry out the study from the Kaduna State Ministry of Education. Also, written permission was obtained from the principals of the selected schools. Informed written consent was obtained from the participants and their parents. The participants with abnormal clinical correlates were advised to seek medical attention at the State’s hospital, Barau Dikko Specialist Hospital, a tertiary institution located in the metropolis.
Results
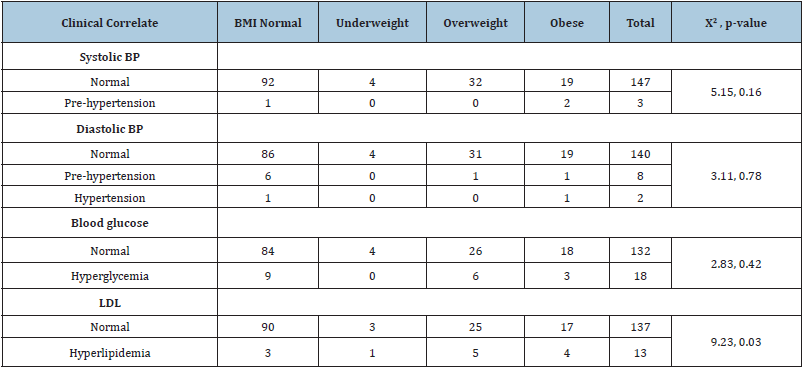
One hundred and fifty adolescents aged between 10 and 19 years participated in the study. Their mean age was 14.1+2.3 years. The distribution of the participants’ demographic data and clinical correlates are shown in Table 1 below. Majority (68, 45.3%) were in the mid-adolescence age group of 14-16 years with an overall female preponderance of 56.7%. The prevalence of overweight and obesity were 21.3% and 14.0% respectively. Only 2(1.3%) were hypertensive while 18(12.0%) and 13(8.7%) had hyperglycemia and hyperlipidemia, respectively. Table 2 shows the relationship between the clinical correlates and obesity. There was a significant relationship between BMI and hyperlipidemia (x2 = 9.23, p = 0.03).
Table 1: Distribution of the demographic data and clinical correlates of the participants.

Table 2: Association between clinical correlates and adolescent obesity (n=150).
Discussion
Data from several developing countries show an increasing prevalence of obesity in adolescent [17-19].
This study found a prevalence of 14% for adolescent obesity and 21.3% for over-weight. A study by Oduwole et al in Lagos, Nigeria conducted in 2012 reported a prevalence rate of 9.4% for adolescent obesity [20]. The health significance of adolescent obesity sterns from its association with other co-morbidities. Studies have indicated that obesity is linked with non-communicable disorders such as hypertension, type-2 diabetes mellitus, and dyslipidemia [11,17,21]. There also seem to be a positive relationship between obesity and hypertension [12,22]. Obesity significantly contributes to morbidity and mortality from cardiovascular disorders [6]. In this study, only 1.3% of the participants had hypertension, and this was diastolic. Also, of note was the prevalence rate of pre-hypertension that was 7.3%. A higher proportion (4.8%) of obese adolescents had diastolic hypertension when compared to the non-obese adolescents (0.8%), though this was not statistically significant. A similar study conducted in Southwest Nigeria reported that obesity significantly increased the risk of both systolic and diastolic blood pressures in the hypertensive range especially in older adolescents [20]. It also reported a hypertensive prevalence of 16% among the adolescents. In a Canadian study, the prevalence of hypertension in a pediatric population was increased at the 85th percentile of BMI-for-age [23]. Another study in the USA found that the risk of hypertension increased as the degree of obesity increased with a three-fold greater risk of hypertension among the severely obese children compared to others [24]. The true burden of obesity-related hypertension and the threshold for BMI to predict a high blood pressure is unknown as data on this is scarce [20,23].
Although studies have shown that being obese increases the risk of developing type 2 diabetes mellitus, it is said to predominantly manifest in adulthood [13,14]. There has been an increase in the prevalence of type 2 diabetes mellitus in adolescents globally over the past two-three decades [25]. This rise in adolescent type 2 diabetes mellitus has coincided with the adolescent obesity epidemic. From this study, 12% of the participants had hyperglycemia with a higher proportion (14.3%) of the obese adolescents having hyperglycemia compared to the non-obese with 11.6%. In the United States, one in three newly diagnosed diabetes cases among adolescents is attributed to type 2 diabetes mellitus [25]. Consequently, the American Diabetes Association has recommended that the screening for type 2 diabetes should begin during adolescence (age 10) or at the onset of puberty in over-weight or obese adolescents [25]. Studies have reported an association between obesity and increased plasma triglycerides, high levels of low-density lipoproteins and cholesterol with lower levels of high-density lipoproteins, which results in an increased cardiovascular risk [19,26,27]. This study reported an 8.7% prevalence of high LDL levels among the participants. There was also a higher percentage (19.1%) of obese adolescents with hyperlipidemia compared with 6.98% among the non-obese adolescents. A significant association (p = 0.03) was reported between BMI and hyperlipidemia in this study. A study in Taiwan recorded a prevalence rate of 13.8% for dyslipidemia among over-weight adolescents with a two-fold increase risk of dyslipidemia when compared with the normal weight participants [28]. Similarly, a study conducted among Korean adolescents showed that dyslipidemia increased with an increasing BMI [29]. About one-fifth of the adolescents studied in Korea were reported to have at least one abnormal lipid profile [30].
Conclusion
The epidemic of adolescent obesity has major implications for health now and in the future. This study has indicated the higher risk of developing hypertension and dyslipidemia among obese adolescents compared to their non-obese counterparts. This further highlights the significant health issues associated with adolescent obesity. Findings from this study would assist health practitioners in their approach to the management of adolescent obesity. There is the need to promptly and adequately address the scourge of adolescent obesity with its co-morbidities.
References
- Gahagan S (2011) Overweight and obesity. In: Kliegman RM, Behrman RE, Jenson HB, Staton BF (Eds.), (19th edn), Nelson Textbook of Pediatrics: Saunders Elsevier Inc., USA, pp. 179- 188.
- Federal ministry of health (2007) National policy on the health and development of adolescents and young people in Nigeria, Nigeria.
- World Health Organization (2009) Child and adolescent health. World health organization regional office for Africa, Africa.
- Mark HB, Merck Research Laboratories (2006) The merck manual of diagnosis and therapy. In: Robert SP, Thomas VJ, Justin LK, Michael B (Eds.), (18th edn), Whitehouse Station, NJ: Merck Research Laboratories, USA, pp. 56- 61.
- Centers for Disease Control and Prevention (2012), USA.
- Raj M, Kumar RK (2010) Obesity in children and adolescents. Indian J Med Res 132(5): 598-607.
- Milos P (2006) Adolescent Obesity, USA.
- Gregory JA, Donald DH (2011) Obesity In: Rakel RE, Rakel DP (Eds.), (8th edn), Textbook of Family Medicine: Elsevier Saunders, China pp. 802- 817.
- Merwevander MT (2012) Obesity in childhood and adolescence. South African Medical Journal 102(5): 289.
- Thomas H (2010) Childhood and Adolescent Obesity. Boulder Medical Center, USA.
- Etukumana EA, Puepet FH, Obadofin M (2013) Prevalence of overweight and obesity among rural adults in north central Nigeria. Nigerian Journal of Family Practice 3(2): 41-46.
- Onuoha F, Azudialu B, Alabi A, Oguojiofor N (2014) The correlation between quetelet index and blood pressure as seen in federal medical center, Owerri, Nigeria. Nigerian Journal of Family Practice 5(2B): 7-12.
- Metz DS (2011) Etiology, treatment and prevention of obesity in childhood and adolescence: A decade in review. J Res Adolesc 21(1): 129-152.
- Ogunbode AM, Ladipo M, Ajayi IO, Fatiregun AA (2011) Obesity: An emerging disease. Niger J Clin Pract 14(4): 390-394.
- Baylor College of Medicine (2012) Pediatric Blood Pressure Reference, USA.
- US National Library of Medicine (2013) Coronary risk profile, USA.
- World Health Organization (2012) Obesity and Overweight. WHO Fact sheet, USA.
- Rossouw HS, Grant CC, Viljoen M (2012) Overweight and obesity in children and adolescents: The South African problem. S Afr J Sci 108(5-6): 31-37.
- Misra A, Shrivastava U (2013) Obesity and dyslipidemia in South Asians. Nutrients 5(7): 2708-2733.
- Oduwole AA, Ladapo TA, Fajolu IB, Ekure EN, Adeniyi OF (2012) Obesity and elevated blood pressure among adolescents in lagos, Nigeria: A cross-sectional study. BMC Public Health 12: 616.
- Novac O, Matasaru S, Tataru S, Felea D, Cosmescu A, et al. (2009) Assessment of complications of excess weight in school-age children and adolescents. Rev Med Chir Soc Med Nat lasi 113(3): 740-744.
- Kotsis V, Stabouli S, Papakatsika S, Rizos Z, Parati G (2010) Mechanisms of obesity-induced hypertension. Hypertension Research 33(5): 386-393.
- Koebnick C, Black MH, Wu J, Martinez MP, Smith N, et al. (2013) High blood pressure in overweight and obese youth: Implications for screening. J Clin Hypertens 15(11): 793-805.
- Lo JC, Chandra M, Sinaiko A, Daniels SR, Prineas RJ, et al. (2014) Severe obesity in children: Prevalence, persistence and relation to hypertension. Int J Pediatr Endocrinol 2014(1): 3.
- Xu H, Verre MC (2018) Type 2 diabetes mellitus in children. Am Fam Physician 98(9): 590-594.
- Misra A, Khurana L (2008) Obesity and the metabolic syndrome in developing countries. J Clin Endocrinol Metab 93(11): 9-30.
- Klop B, Elte JWF, Cabezes MC (2013) Dyslipidemia in obesity: Mechanisms and potential targets. Nutrients 5(4): 1218-1240.
- Kuo P, Syu J, Tzou IL, Chen P, Su H, et al. (2014) Prevalence and trend of dyslipidaemia from 1996 to 2006 among normal and overweight adolescents in Taiwan. BMJ Open 4(2): e003800.
- Kim SH, Ahn BC, Joung H, Park MJ (2012) Lipid profiles and prevalence of dyslipidemia in Korean adolescents. Endocrinol Metab 27(3): 208-216.
- Yang S, Hwang JS, Park HK, Lee HS, Kim HS, et al. (2012) Serum lipid concentrations, prevalence of dyslipidemia, and percentage eligible for pharmacological treatment of Korean children and adolescents; Data from the Korean national health and nutrition examination survey IV (2007-2009). PLoS One 7(12): e49253.
© 2020 AEseigbe Patricia. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






