- Submissions

Full Text
Global Journal of Endocrinological Metabolism
Atypical Presentation of Acute Lymphoid Leukemia as Hypercalcemia and Bilateral Renal Calculi
Madhu BJ1, Chithambaram NS*1, Harish G1, Vijaya Sarathi2
1 Department of Pediatrics, Vydehi Institute of Medical Sciences and Research Center, India
2 Department of Endocrinology, Vydehi Institute of Medical Sciences and Research Center, India
*Corresponding author: Chithambaram NS, S-15, Sudhamani Residency, 2d Kalyan Nagar, Chelekere, Bangalore, Pin: 560043; Email: chithams1@gmail.com
Submission: April 05, 2018; Published: August 31, 2018

ISSN 2637-8019Volume2 Issue5
Abstract
The presentations of acute leukemias in children are varied. Here, we report a child who presented with severe hypercalcemia with bilateral renal calculi who was later diagnosed to have acute leukemia. Intravenous Pamidronate is an effective and safe therapy for acute management of severe hypercalcemia in acute leukemias.
Keywords: Acute leukemia; hypercalcemia; Renal calculi; Pamidronate
Introduction
Hypercalcemia presenting as acute leukemia is a rare but an important complication in childhood leukemia. Hypercalcemia usually results in nonspecific constitutional symptoms, although it can also manifest as a life-threatening metabolic emergency [1]. Hypercalcemia alone or combined with osteolytic lesions can be the only presenting symptom of Acute Lymphoblastic Leukemia (ALL) in children. Here, we report a child who presented with severe hypercalcemia with bilateral renal calculi who was later diagnosed to have acute leukemia.
Case History
A 4yr 8-month-old boy was brought with the complaints of moderate to high grade fever on and off since last 9 months. Preceding this, the boy had a fracture of spine following fall from stairs for which he was treated conservatively. He had history of vomiting, 3-4 episodes per day, decreased appetite and weight loss in the last 9 months. He also had constipation, pain abdomen, frequent passing of urine, bone pains and generalized weakness. On examination, he was wasted and emaciated. He weighed 10 kgs with a height of 87cms, head circumference of 47cms and midarm circumference of 10cms. Systemic examination findings were within the normal limits. His WBC counts were 13,000/cmm, neutrophils-59.4%, lymphocytes-35.8%, RBC counts were 3.72 million cells/cmm, Hb-9.4g/dl. Peripheral smear showed normocytic hypochromic with neutrophilia. No blast cells were seen in peripheral smear. His renal functions and liver functions were normal. Serum Sodium -131mEq/l, Serum Potassium-4.1mEq/l, Serum Chloride-94mEq/l, and Serum Calcium was 19mg/dl (reconfirmed with a repeat test). Urine routine and microscopy examination were unremarkable. Other metabolic investigations like random plasma glucose was 125mg/dl, serum phosphorus 5.4mg/dl, serum uric acid 9.9mg/dl, serum magnesium- 1.58mg/dl. In view of severe hypercalcemia, he was treated with saline diuresis followed by intravenous Pamidronate infusion over 4 hours (1mg/kg). 24-hours later serum calcium level decreased from 19mg/dl to 12.7mg/dl. 48-h later serum calcium was 10.2mg/dl. Further evaluation revealed a parathormone levels of 6.8pg/ml. USG abdomen showed normal sized kidneys with loss of corticomedullary differentiation in both the kidneys and multiple renal calculi in both kidneys, largest measuring 4 mm and was reported as grade II medical renal disease with non-obstructing bilateral renal calculi. Skeletal survey showed multiple osteolytic lesions all over the body as shown in Figure 1.
Figure 1:X-ray showing osteolytic lesions at the upper ends of the both humerus.
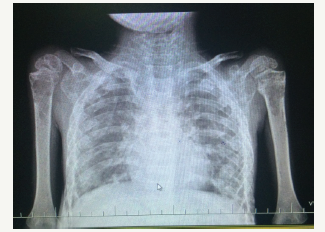
In view of the above findings, hematological malignancy was suspected, and bone marrow aspiration was done. This showed normal erythroid maturation, myeloid series markedly suppressed, and eosinophils are mildly increased. Lymphoid series show many atypical cells which large cells with high N/C ratio are, coarse chromatin, 0-1 nucleoli and scant cytoplasm, a few of these cells show nuclear convolutions which was reported as ALL. Bone marrow biopsy was inconclusive. Other tests like flowcytometry and chromosomal studies were advised to find out the type of ALL. However, the parents deferred any further evaluation and treatment despite counseling.
Discussion
We report a rare case of ALL presenting with hypercalcemia. In contrast to adults, hypercalcemia is very rare in children with cancer (0.4%). Incidence of hypercalcemia in children with acute leukemia or lymphoma at diagnosis is < 0.3% [2]. A higher incidence (4.8%) of hypercalcemia occurs in children with early pre- B-cell ALL [3]. An association of t(17;19) with hypercalcemia has also been reported among children with ALL [4-6]. However, we could not get the exact typing and genetic studies in our patient. In contrast to children with ordinary ALL, gastrointestinal symptoms (nausea, vomiting, abdominal pain) and skeletal symptoms (bone pain, gait disturbance) are the predominant presenting complaints in ALL children with hypercalcemia [1]. Most of the described cases of hypercalcemia in ALL children have been characterized by absence of blasts in peripheral blood film and are associated with multiple osteolytic lesions [1,7]. Our patient also had similar features. The unique finding in our patient was the presence of bilateral renal calculi. The long-standing hypercalcemia and hypercalciuria had led to formation of renal calculi. Although, urolithiasis has been reported as a complication of ALL therapy, to the best of our knowledge, this is the first case of pediatric ALL to have multiple renal calculi before initiation of chemotherapy [8].
Severe Hypercalcemia with serum calcium level as high as 20.8 mg/dl have been reported in children with ALL [4]. Hypercalcemia in ALL is of multifactorial origin. It may result from markedly increased osteoclasic resorption in the areas surrounding malignant cells within the marrow space (local osteolytic hypercalcemia ) or production of humoral factors like PTHrP by tumour cells which increase bone resorption and retention of calcium through kidney (humoral hypercalcemia of malignancy) or very rarely in association with true PTH dependent hypercalcemia [9]. PTHrP should be measured in conditions where hyper calcaemia is an uncommon association as it may help confirm a paraneoplastic aetiology. Chemotherapy normalizes hypercalcemia in most patients with ALL. Additional measures like saline diuresis, calcitonin, corticosteroids and bisphosphonates may be used in moderate to severe cases of hypercalcemia. Pamidronate at a dose of 1mg/kg is a safe and effective treatment for severe, refractory hypercalcemia of malignancy in children [10].
Conclusion
This rare case highlights the presence of severe hypercalcemia with multiple renal calculi at the initial presentation of acute leukemia in the absence of blasts in the peripheral blood film. Intravenous Pamidronate is an effective and safe therapy for acute management of severe hypercalcemia in acute leukemias.
References
- Trehan A, Cheetham T, Bailey S (2009) Hypercalcemia in acute lymphoblastic leukemia: an overview. J Pediatr Hematol Oncol 31(6): 424-427.
- McKay C, Furman WL (1993) Hypercalcemia complicating childhood malignancies. Cancer 72(1): 256-260.
- Hibi S, Funaki H, Ochiai KR, Ikushima S, Todo S, et al. (1997) Hypercalcemia in children presenting with acute lymphoblastic leukemia. Int J Hematol 66(3): 353-357.
- Hunger SP, Devaraj PE, Foroni L, Secker LMW, Cleary ML (1994) Two types of genomic rearrangements create alternative E2A-HLF fusion proteins in t (17;19)-ALL. Blood 83(10): 2970-2977.
- Inukai T, Hirose K, Inaba T, Kurosawa H, Hama A, et al. (2007) Hypercalcemia in childhood acute lymphoblastic leukemia: frequent implication of parathyroid hormone-related peptide and E2A-HLF from translocation 17; 19. Leukemia 21(2): 288-296
- Minson KA, Prasad P, Vear S, Borinstein S, Ho R, et al. (2013) t(17;19) In children with acute lymphocytic leukemia: a report of 3 cases and a review of the literature. Case Rep Hematol 2013: 563291.
- Soni PN (1993) Hypercalcemia and multiple osteolytic lesions in childhood acute lymphoblastic leukaemia. Postgrad Med J 69(812): 483-485.
- Howard SC, Kaplan SD, Razzouk BI, Rivera GK, Sandlund JT, et al. (2003) Urolithiasis in pediatric patients with acute lymphoblastic leukemia. Leukemia 17(3): 541-546.
- Andrew F, Stewart MD (2005) Hypercalcemia associated with cancer. N Engl J Med 352: 373-379.
- Young G, Shende A (1998) Use of pamidronate in the management of acute cancer-related hypercalcemia in children. Med Pediatr Oncol 30(2): 117-121.
© 2018 Chithambaram NS. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






