- Submissions

Full Text
Gerontology & Geriatrics Studies
Certain Immunity Challenges of Covid-19; Neurotoxic Effects and Immunopsychiatry
James Paul Pandarakalam*
Department of Psychiatry, Hollins Park Hospital, UK
*Corresponding author: James Paul Pandarakalam, Department of Psychiatry, Hollins Park Hospital, Warrington, UK
Submission: May 03, 2021;Published: May 11, 2021

ISSN 2578-0093Volume6 Issue5
Abstract
Cytokine Release Syndrome (Cytokine storm) which is an autoimmune reaction or an autoimmune-like reaction accounts for the complications of COVID-19 and finding immunotherapeutic drugs capable of blocking the cytokine storm would prevent the morbidity and mortality of COVID-19. Certain groups of people are recognized to be more vulnerable to the complications of this infection than others. The Cytokine Release Syndrome (CRS) leads to several neurological conditions even meriting COVID-19 to be classified as a “neurotoxic illness.” The development and manifestations of neurological complications of COVID-19 may offer rational support to the novel concept of immunopsychiatry and immuno-neuropsychiatry that has the potential to evolve as a subspecialty of general psychiatry. CRS that occurs in severe cases of COVID-19 has rekindled an interest in autoimmune disorders and could offer distant conceptual models for the aetiological search of other medical conditions and even psychiatric conditions. The left-over autoantibodies are held responsible for the lingering symptoms of COVID-19. If schizophrenia is an autoimmune disorder, clozapine, the immunomodulator drug may be hypothetically halting the production of autoreactive antibodies responsible for the psychotic illness. In these desperate times, new treatment strategies may also involve repurposing the existing medications. Studies of COVID-19 might lead to new horizons in immunology.
Keywords:Immunity; Cytokine storm; Autoantibodies; COVID-19; Clozapine; Immuno-neuropsychiatry
Introduction
In December 2019, COVID-19 first emerged in Wuhan and swiftly disseminated throughout the entire globe causing high mortality. From genomic surveillance of clinical samples from patients with viral pneumonia in Wuhan, China, a novel coronavirus (termed 2019-nCoV) has been recognized [1]. A phylogenetic analysis of 2019-nCoV, sequenced from nine patients’ samples, showed that the virus belongs to the subgenus Sarbecovirus [2]. The same study identified that Covid-19 was more similar to two bat-derived coronavirus strains, bat-SL-CoVZC45 and bat-SL-CoVZXC21, than to known human-infecting coronaviruses, including the virus that caused the SARS outbreak of 2003. COVID-19, the viral disease does not discriminate people at a social or national level, but it seems to discriminate people at a genetic and immunity level. It is the historical observation that people who survive the ravages of epidemic disease are untouched when they are exposed to the same disease again and that they had become immune to the infection. Microorganisms can reproduce and evolve much more rapidly than their human hosts and human body invests heavily in cells dedicated to defense, which constitute the immune system. Most infections are efficiently cleared by an innate immune response. Most people who contract COVID-19 recover, but many have succumbed to death.
Compromised immunity is a recognized morbidity factor for COVID-19. People with cardiovascular diseases, COPD, obesity, type 2 diabetes mellitus and other underlying health conditions are at increased risk of severe illness from COVID-19. In every medical crisis, there are scientific opportunities for progress. Never before have international efforts and collaborations been so important. It would be the responsibility of all medical specialties to solve the medical pandemic while civil authorities focus on the social aspects of the pandemic. Like the complications of Diabetes Mellitus infiltrating the whole field of medicine, complications of COVID-19 appear to encompass the whole system of medicine. SARS-CoV-2 is now being recognized as a neuro-invasive virus. Disparate neurological manifestations and long-term neuropsychiatric sequelae have been recounted in infections with numerous previously known coronaviruses. British scientists analyzing data from a widely used COVID-19 symptom-tracking app have found that there are six distinct types of the disease, each distinguished by a cluster of symptoms. The King’s College London team found that there are six types of COVID-19 that could be distinguished from each other by a cluster of symptoms and correlated with levels of severity of infection. They could be grouped as follows: a. ‘Flu-like’ with no fever: Headache, loss of smell, muscle pains, cough, sore throat, chest pain, no fever. b. ‘Flu-like’ with fever: Headache, loss of smell, cough, sore throat, hoarseness, fever, loss of appetite. Gastrointestinal: Headache, loss of smell, loss of appetite, diarrhoea, sore throat, chest pain, no cough. d. Severe level one, fatigue: Headache, loss of smell, cough, fever, hoarseness, chest pain, fatigue. e. Severe level two, confusion: Headache, loss of smell, loss of appetite, cough, fever, hoarseness, sore throat, chest pain, fatigue, confusion, muscle pain. f. Severe level three, abdominal and respiratory: Headache, loss of smell, loss of appetite, cough, fever, hoarseness, sore throat, chest pain, fatigue, confusion, muscle pain, shortness of breath, diarrhoea, abdominal pain.
When responding to infection, the immune system starts with fast innate immune mechanisms that halt infections at an early stage. They consist of four key elements: the proteins such as mannose-binding lectin, which noncovalently bind to the surfaces of pathogens; proteins that complement and bind covalently to pathogen surfaces; phagocytic cells, which kill pathogens by ingesting them; and cytotoxic cells and molecules that kill virus-infected cells [3]. The first line of defense is the protective epithelial surface of the body and the commensal microorganisms. The effector cells and molecules of the innate immune response deal with the pathogens that manage to penetrate the epithelial surfaces. Repeated exposure to the same pathogen will not modify or improve the innate defense mechanisms, as there is no antibody formation. If innate immunity is all about pathogen destruction, adaptive immunity involves pathogen recognition.
Adaptive Immunity Responses
Nonspecific reactions by leukocytes and humoral components are generated by invertebrates whereas vertebrates furthermore show specific responses through T cells and B cells that destroy cancers and synthesize immunoglobulins. A physiologic inflammatory response helps to control the focus of infection, whereas a dysregulated host response leads to macro- and micro-circulatory failure, thus inducing organ dysfunction, which determines the patient’s symptoms and the clinical course of disease [4,5]. The development of immunity to a pathogen through natural infection is a multi-step process that typically takes place over 1-2 weeks. The body responds to a viral infection immediately with a non-specific innate response in which macrophages, neutrophils, and dendritic cells slow the progress of virus and may even prevent it from causing symptoms. This non-specific response is followed by an adaptive response where the body makes antibodies that specifically bind to the virus. These antibodies are proteins called immunoglobulins. The body also makes T-cells that recognize and eliminate other cells infected with the virus. This is called cellular immunity. This combined adaptive response may clear the virus from the body, and if the response is strong enough, may prevent progression to severe illness or re-infection by the same virus. This process is often measured by the presence of antibodies in blood.
Infections that thwart innate immunity generally spread to specialized lymphoid tissues such as lymph nodes and spleen. The mechanism of adaptive immunity involves the B- and T-lymphocytes. Small circulating B- and T-lymphocytes with receptors that bind to pathogens or their macromolecular components are selected and activated in these secondary lymphoid organs. In addition to helping resident macrophages to phagocytose pathogens, these interactions of adaptive immunity provoke the macrophages to pour out inflammatory cytokines that activate neutrophils and Natural Killer (NK) cells to the site of the infection [3]. Thus, an adaptive immune response may terminate an infection and provide longterm protective immunity against a pathogen that triggered this immunity response. There are inherited leucocytes and acquired deficiencies in the immune system that can lead to debilitating chronic infections. A child may achieve passive immunity through consuming breast milk.
Cytokine Release Syndrome
CRS accounts for many of the COVID-19 fatalities [6,7]. CRS is a disproportionate immune system panic response with serious consequences. In 1991, Alegre et al. [8] introduced the term cytokine release syndrome in experimental hyperinflammation models [8]. Ferrara et al. later coined the term cytokine storm in 1993 to describe the cytokine interleukin-1’s (IL-1) effect on graftversus- host disease after transplantation [9]. In 1996, Aikawa [10] described cytokine storms as the key pathway to multiple organ dysfunction syndrome after surgical infections and sepsis [10]. Metaphorically, CRS is comparable to using machine guns instead of a pistol to thwart a thief: not only is the house destroyed, but also neighbouring structures.
Cytokines are small proteins released by cells that function as immune response messengers or signaling molecules between cells. These proteins stimulate immune cells to act and then inhibit them appropriately, thereby keeping balance (Table 1). Additionally, cytokines can promote T-cell synthesis which can, in turn, lead to greater cytokine production. Cytokines also stimulate or inhibit inflammation as an immune response, and act as chemical attractants to bring infection-fighting cells to a site as needed. Cytokine is a general name, however; more specific names include lymphokines (cytokines made by lymphocytes), monokines (cytokines made by monocytes), chemokines (cytokines with chemotactic activities) and interleukins (cytokines made by one leucocyte that acts on other leucocytes) [11]. Interleukins mean between white cells.
Table 1: Summary of cytokines and immune cells.
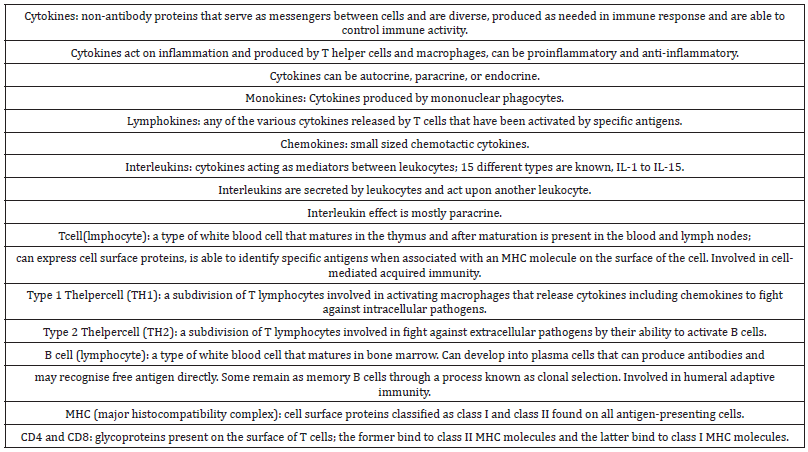
Leucocytes are the first to sense pathogen infiltration and alert immune cells, including T-cells and macrophages, to the infection site. Immune cells produce cytokines to help synchronize combat against a virus. Notably, when T-cells are activated, they release cytokines, which trigger additional T-cell production, which then releases even more cytokines. Cytokines activate immune cells at the infection site and prompt the arrival of more for action. One type of T-cell are cytotoxic T-cells, which roam the body and mercy-kill infected cells that chemically request to be killed, which stops increased virus production, such as that of COVID-19. Normally, cytotoxic T-cells only target infected cells and move on. Additionally, the immune system has a chemical indicator that tells overzealous immune cells to stand down once the threat has been neutralized. However, for unknown reasons, sometimes too many immune cells are mobilized to an infection site. Thus, the immune system becomes overwhelmed and malfunctions. This propagates a CRS, and this reaction inflames the tissue surrounding the infection. When the infection is in the lungs, this severe inflammation can cause permanent damage. CRS are indicated by elevated ferritin, hepatic enzymes, protein D-dimers and lactate dehydrogenase, which are assessed in haematological tests. In essence, our body’s immune response becomes so overwhelmed it stops differentiating between infected and healthy cells and attacks everything, including the immune system itself.
In a subset of COVID-19 cases, research found that immune systems battling this virus are overwhelmed, as detailed above. In a nutshell, the immune cells’ uncontrolled reaction results in a CRS, and the disproportionate discharge of these chemicals damages multiple organs. CRS may result in more collateral damage to the body than even the invading pathogen. Blood vessels may rupture due to cytokine-induced inflammation, and the whole circulatory system may subsequently be affected. The number of deaths from COVID-19 has increased rapidly, and the cytokine interleukin-6 (IL-6) is one of the significant inflammatory factors in CRS, as it increases vascular permeability and affects heart function. Massive cell destruction, multiple organ failure, sepsis and even death can occur as a result. It is not clear whether COVID patients with CRS may already have pre-existing autoimmunity conditions that is comprised when they become critically ill or more likely, the immunological environment of serious COVID-19 infection, including TLR7 activation by SARS-CoV2 ssRNA, is sufficient to drive de novo autoreactivity against a variety of self-antigens [12,13].
A prolonged CRS eventually completely shuts down respiration, and there is nothing to prevent this [14]. Blocking CRS is the greatest challenge to saving patients with COVID-19. Therapeutic agents that convert the hyperinflammatory reaction to a proinflammatory modem comprise the ideal means of handling this crisis. Actemra (tocilizumab) by Roche, immunosuppressants and repurposed antivirals are currently used to combat CRS. Actemra blocks IL-6. Another drug, Kineret, blocks IL-1. A recent study reported promising preliminary results from an AstraZeneca cancer drug called Calquence (acalabrutinib) that stops cytokines at their root. Dexamethasone is the latest addition to the treatment regime. To pacify CRS, a dialysis-like device called CytoSorb has claimed some success by filtering out cytokines from a patient’s blood and returning it to the body [15]. Therapeutic studies should be extended to immunosuppressant drugs used in different medical specialties, including psychopharmacology [16].
Long-Covid
Some recovered patients are finding themselves periodically breathless, months after they have been found clear of the pathogen. Certain others are left with draining fatigue, skin rashes or diarrhoea. However, the most common feature is crippling fatigue. There are yet other symptoms that include: breathlessness, a persistent cough, joint pain, muscle aches, hearing and eyesight problems, headaches, loss of smell and taste as well as damage to the heart, lungs, kidneys, and gut [17,18]. One single centre study found that in patients who had recovered from COVID-19, 87.4% reported persistence of at least one symptom, particularly disabling fatigue, and dyspnoea [19]. Mental health problems have been reported including depression, anxiety and struggling to organize thinking clearly. These symptoms are not related to the severity of the illness.
Long-Covid is another immunity challenge posed by SARSCoV- 2. Autoreactive antibodies are thought to be responsible for long Covid symptoms that linger months after the apparent recovery from the viral infection. It has yet to be ascertained whether autoantibodies would dissipate over time or follow the pattern of other autoimmune diseases. In that case, it would be hard to find a permanent cure. If COVID-19 is leaving its permanent trails in human biology, that would also offer us hints to the pathogenesis of a few psychotic and neurological disorders. Patients acquired pneumonia from non-Covid sources can also have continual symptoms, suggesting that these lingering symptoms may not be exclusive to COVID-19.
Neurotoxic Effects-Immunopsychiatry
In the short duration of COVID-10 pandemic, it has already manifested several neurological and neuroradiological phenotypes and such a situation warrants a study into aetiology and mechanism of the link between SARS-CoV and the CNS [20]. Several laboratories have already studied SARS-CoV in a number of animal species, including primates, mice, rats, hamsters, and moss [21]. The affinity of coronavirus to CNS has been verified for almost all beta-coronaviruses, including SARS-CoV-1, MER-CoV, HcoV229E, HcoV-OC43, as well as others [22-24]. It is now well established that coronaviruses are not only restricted to the respiratory tract, but they have a neuroinvasive predisposition. They can also impinge on the CNS, leading to neurological maladies [25]. Thus, neurological phenotypes that affect both the central and peripheral nervous system, leading to anosmia, ageusia, necro-haemorrhagic encephalitis, Guillain-Barre syndrome have been documented [26- 28].
Butler et al note that the biological mechanism including autoimmunity could enlighten the aetiological research of psychotic disorders [29]. Such an assumption opens the door to the development of an immunity-based psychiatry. Immunopsychiatry is a fledgling field of research and it has great potential in the aetiological research of psychotic disorders for finding new treatment strategies [30,31]. Immuno-neuropsychiatry is yet another thought piece that needs further substantiation [32]. The autoimmune aetiology of a subset of schizophrenia is gaining ground [33-37]. Psychotic symptoms are conjectured to be the outcome of autoimmune antibodies against the brain and later shaped by neurological and psychosocial factors. Nicholson et al. [38] have earlier on hypothesised that anti-basal ganglia antibodies are responsible for obsessive compulsive disorder in some adult individuals [38]. OCD can coexist in 20% of bipolar disorders and such a comorbidity point towards, both the disorders being autoimmune disorders.
Neurotoxic effects of COVID-19 either directly through viral invasion or indirectly through CRS offer valuable insights into the pathogenesis of psychotic disorders. So, a discussion of such complications of COVID-19 is significant for health professionals interested in immunopsychiatry. As mentioned before, SARS-CoV-2 is associated with several neurological symptoms and syndromes, including headache, fatigue, anosmia, ageusia, anorexia, myalgias, asthenia, meningitis, encephalitis, Guillain-Barré Syndrome, altered consciousness, syncope, stroke and now they are getting amply written about [39-42]. Neurological effects of coronaviruses (including SARS-CoV-2) may be triggered by direct cytopathic effects of the virus, secondary effects of severe pulmonary infection, a CRS, or a combination of these [42]. SARS-CoV-2 may cause neurological disorders by directly infecting the brain or as a result of the strong activation of the immune system. Recent studies have detected the novel coronavirus in the brains of fatal cases of Covid-19. It has also been suggested that infection of olfactory neurons in the nose may enable the virus to spread from the respiratory tract to the brain. Olfactory and gustatory dysfunctions are now recognised clinical symptoms, and these symptoms indicate the viral affinity of the nervous system. Two-thirds of patients were observed to initially suffer from olfactory and gustatory symptoms, and the fatigue experienced by most patients is suggestive of neurotoxicity [43].
A full one-third of 214 COVID-19 patients experienced neurological symptoms such as headaches, dizziness, and cognitive impairment in a Cambridge study, and according to this study COVID-19 merits to be classified as a “neurotoxic illness.” Once the virus enters the brain, further infection is almost inevitablethere are ACE2 (Angiotensin-converting enzyme 2) membrane receptors on the surface of the brain cells through which the virus easily penetrates the tissue, causing inflammation. Both ischaemic and haemorrhagic strokes have been reported in COVID-19 cases. In milder cases, encephalitis can cause flu-like symptoms; in more severe cases, it might bring seizures, paralysis, and confusion. Almost a third of SARS and MERS patients hospitalized experienced confusion while treated in the hospital [44]. Early evidence from the COVID-19 pandemic suggests that delirium could be a similar common problem. The impacts of COVID-19 on the brain and the effects of potentially altered brain function on mental health issues are both unclear.
It is now well proven that SARS is clinically neurotoxic, causing mental health and neurological disorders including neuromuscular and joint findings [45]. COVID-19 has also been linked with encephalitis [46] and Guillain-Barré syndrome [47-49], where the immune system attacks the nerves-an overreaction of the immune cells in an attempt to thwart the viral pathogen. This is akin to a CRS. It is uncertain if the encephalitis encountered in COVID-19 is due to a direct invasion of the virus into the nervous system or the destruction is due to the overactivity of the immune system. Again, the mechanisms of Guillain-Barré appear to be associated with the normally protective antibodies of immune system, which are also thought to occasionally go ‘haywire’ by attacking the nerves and their coating.
Further intense exploration is warranted regarding the possibility of COVID-19 causing immune-mediated neurotoxic effects on the brain-just like SARS. From 1918 to 1919, a Spanish flu pandemic emerged, which was traced to a spike in Parkinson’s disease after the virus caused degeneration of nerve cells. Hearing loss has been reported in several cases of recovered COVID-19 patients. Even in asymptomatic cases, COVID-19 infection could have deleterious effects on cochlear hair cell functions [50]. Typically, virus-induced hearing loss is sensorineural, although conductive and mixed hearing losses can be seen following infection with certain viruses. The mechanism of the viral hearing loss is unclear, although drugs used in symptomatic cases have been indicated as responsible.
Even among young patients who had no previous risks of cardiac risk factors, cardiovascular accidents have been reported. An increased incidence of clot formation has been observed in patients with COVID-19, and the uptick in clotting phenomenon occurring in COVID-19 may explain the high incidence of ischaemic strokes, a connection that was also noted during the 2002-2003 outbreak of SARS [51]. It has been speculated that in the future years, the inflammatory systemic process and/or the inflammatory process of the brain could initiate long-term mechanisms that generally lead to an upsurge of neurological and neurodegenerative disorders [20]. CoV positive individuals should be investigated routinely for neurological symptoms, including headache, impaired consciousness, paresthesias, and other neurological signs. Treatment of infection-related complications is an important factor in better prognosis of severe COVID-19 patients.
The viral precipitation of autoimmune disorder is a wellrecognized phenomenon. The novel viral infection rekindles a fresh interest in the viral and autoimmune aetiology of a subset of schizophrenia. Studies of the neurotoxic effects of COVID-19 due to an autoimmune reaction may lead to the arena of immunoneuropsychiatry. The impact of CRS on various physiological systems including nervous system give a clue to the mechanism of aberrant immunological reactions leading to the production of psychotic symptomatology. Multiple complications of COVID-19, including neurological morbidities are attributable to CRS. Even though such an exaggerated immune activity has been identified in other situations, there is a dearth of effective medications to combat the autoimmune development. CRS puts the clinician in an ambivalent state in the sense that immunosuppressants would make the patient more vulnerable to a continued infectious process-yet they have been used due to a lack of choices. Strategies to prevent and control CRS, the pivotal process responsible for all the fatalities are crucial. It is clearly an autoimmune reaction and finding immunotherapeutic drugs capable of halting the CRS would prevent the morbidity and mortality of COVID-19. Such drugs may also have some bearing in the future treatments of psychotic disorders that have an autoimmune aetiology.
Clozapine- an Immunomodulator
Patients prescribed clozapine are found to be highly vulnerable to the influenza virus. In the same way, at the beginning of the current pandemic, clozapine users were thought to be at a higher risk of catching COVID-19, even though there are no data to suggest or confirm such an assertion. Clozapine has an immunosuppressant effect [52]. Hospital admissions due to community acquired pneumonia are higher among clozapine-treated patients [53,54]. It may be possible that such a predisposition makes clozapine users highly vulnerable to pneumonia, in the event that they catch COVID-19. These are still early days to evaluate the risks of COVID-19 among clozapine users, if this factor poses any additional risks at all.
From what is known about clozapine pharmacology, it has been hypothesised that clozapine possibly impairs immunological mechanisms, which would increase the risk of pneumonia in infected patients [55]. It has also been suggested that once fever and/or pneumonia develops, the clozapine dose should be cut in half to decrease the risk of clozapine intoxication, and if the signs of clozapine intoxication still persist in spite of the reduced dosage, it is recommended that use of the drug be completely stopped [56]. It has also been advocated that reducing the dose of clozapine would be desirable rather than abruptly withdrawing it because of the risk of rebound psychosis that could be confused with COVID linked psychiatric symptoms [57]. Clozapine works on immunomodulation and neuromodulation and its immunomodulatory effects may outweigh the neuromodulation. It may have the potential to control the autoreactive antibodies. Clozapine becomes an immunosuppressant at high doses, but higher doses have the clinical advantage of enhancing its neuromodulation. Higher doses risk inviting various side effects.
Less is known about its immunological effects. The immunological anomalies associated with clozapine treatment are pointed out to support the immunomodulatory properties of clozapine. They include the following: A. elevated serum immunoglobulin levels [58], B. decreased mitogenic response of peripheral blood lymphocytes to phytohaemagglutinin and pokeweed mitogen [59,60], C. the presence of morphologically abnormal large lymphocytes in the blood and bone marrow [52], D. increased serum IL-2 receptor levels [61], E. decreased IL-2 levels [62], F. IFN-g production [49], G. high serum level of IL-6 [63]. H. an increase in T suppressor lymphocytes in drug-naive schizophrenic patients and an increase in T helper lymphocytes in drug-treated patients [64]. The conjecture is that clozapine may be quietening the subtle autoimmune reaction that may be happening in schizophrenia. Canas CA has hypothesised that the auto reactivity antibody formation goes through two phases of immunity in the COVID-19 cases [65]. Firstly, there is an immune suppression and secondly there is impaired immune reconstitution. As a result of immune suppression, body loses its self-tolerance to self-antigens and this in turn lead to failed immune reorganisation.
Therefore, what is needed is a gentle immune modulator drug that can be of immunotherapeutic benefit. Clozapine is useful in regaining the immune cadence at lower doses. Of note, many low dose clozapine users are found to be more resilient to COVID-19 and it is an unanswered puzzle. In an isolated case, it was found that a patient on clozapine with a history of flue related pneumonia was spared from COVID-19 even when her mother with whom she lived got infected with COVID-19. The immunosuppressant and immunotherapeutic effect of clozapine at higher doses and lower doses respectively are yet only clinical conjectures that need scientific validation. Clozapine has to be liberated from its stigma as an anti-schizophrenia drug only. It could be used an add-on therapy in other situations. Scientific facts evolve as thought pieces which later get transformed as falsifiable, modifiable, or confirmable hypothesis.
The supposition of Canas CA is particularly relevant to explain the rebound psychosis associated with abrupt withdrawal of high dose clozapine; an apparent immunosuppression is suddenly terminated that result in a failed immune modulation. Similar phenomena are observed in clinical practice, where a form of transient immunosuppression and immunodeficiency occurs before a sudden reactivation of the immune components, which is associated with the triggering of autoimmunity or autoinflammation phenomena [65]. Examples quoted by Canas CA are withdrawal of chemotherapy for cancer [66], withdrawal of glucocorticoids [67], withdrawal of anti-TNF medication in patients with tuberculosis [68], or initiation of treatment for HIV infection [69], postpartum [70] etc.
If schizophrenia is an autoimmune disorder, there should be taking place subtle autoimmune reaction leading to the psychotic symptom formation and clozapine when used may be arresting the autoreactive antibody production. So far, there has not been any experimental drug trials involving this drug to control the autoimmune reactions in other critical crisis like COVID-19. Clozapine as an add on therapy is worth experimenting in desperate cases of cytokine storm due to Covid-19. Unfortunately, clozapine has the stigma of a psychiatric drug and that may be an impediment in non-psychiatric medical trials. Such testing will have to be initially limited to psychiatric patients who are suffering from complications of COVID-19. Currently the popular drug is hydrocortisone which is a glucocorticoid and works as an anti-inflammatory and by immune suppression in critically ill COVID patients.
Conclusion
Tests, novel treatments, and vaccines are the three tools needed to combat COVID-19. Now that so many people have been infected with COVID-19, over time, the virus may struggle to spread due to the left-over antibodies and herd immunity. If the virus confers immunity, herd immunity which is a natural by-product of an epidemic, may just become a reality. These are still early days to establish whether COVID-19 will confer either a short-term or longterm immunity onto people who have recovered from this infection. Herd immunity is not something that can be made to happen. In the absence of a vaccine, herd immunity assumes that the vast majority of people who recover from the coronavirus will then be immune, which has not yet been conclusively determined. If this viral infection only offers short-term immunity, herd immunity will not have much significance, and even a safe and effective vaccine will not be able to eradicate the illness; only effective medications will be able to save this critical situation. Balancing personal immunity is highly significant to combat the viral intruder.
The gravity of CRS is clearly manifest in the case of COVID-19. The advance of autoimmune conditions consequent to COVID-19 infection could be related to two mutual reasons: transient immunosuppression of innate and acquired immunity leading to a loss of self-tolerance to self-antigens, and a form of inapt immune reconstitution in persons with predisposing conditions of autoimmunity. What is required is an efficient immunoregulator to block the CRS which is responsible for the associated morbidity and mortality. Repurposing existing medications would be an immediate option. There may be hidden immunotherapeutic drugs in different specialities of medicine.
More research is warranted into the exploration of autoimmune reactions that may help the growth of immunity-based psychiatry. The effective way of treating autoimmune reactions and disorders would be identifying “anti-autoantibodies.” The many processes involved in autoimmune reactions are unclear. So, attempting to identify the exact aetiology of autoimmune related conditions in the autoimmune sphere may look like finding a needle in the haystack. The emerging field of “immuno-neuropsychiatry” has the capability to broaden the exploration of a mechanism-based nosology and such an endeavour might bring us to the discovery of more effective personalised treatment strategies.
References
- Tan W, Zhao X, Ma X (2020) A novel coronavirus genome identified in a cluster of pneumonia cases-Wuhan, China 2019-2020. China CDC Weekly 2: 61-62.
- Lu R, Zhao X, Li J, Niu P, Yang B, et al. (2020) Genomic characterization and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. The Lancet 395(10224): 565-574.
- Parham Peter (2009) The immune system. Garland Science, New York, USA.
- Gerlach H, Toussaint S (2007) Organ failure in sepsis. Curr Infect Dis Rep 9(5): 374-381.
- Singer M, Deutschman CS, Seymour CW, Hari MS, Annane D, et al. (2016) The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 315(8): 801-810.
- Ruan Q, Yang K, Wang W, Jiang L, Song J (2020) Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Medicine 46(5): 846-848.
- Mehta P, McAuley DF, Brown M, Sanchez E, Tattersall RS, et al. (2020) COVID-19: consider cytokine storm syndromes and immunosuppression. The Lancet 395(10229): 1033-1034.
- Alegre ML, Vandenabeele P, Depierreux M, Florquin S, Lanckmanet DM, et al. (1991) Cytokine release syndrome induced by the 145-2C11 anti-CD3 monoclonal antibody in mice: prevention by high doses of methylprednisolone. J Immunol 146(4): 1184-1191.
- Ferrara JL, Abhyankar S, Gilliland DG (1993) Cytokine storm of graft-versus-host disease: a critical effector role for interleukin-1. Transplant Proc 25(1pt 2): 1216-1217.
- Aikawa N (1996) Cytokine storm in the pathogenesis of multiple organ dysfunction syndrome associated with surgical insults]. Nihon Geka Gakkai Zasshi 97(9): 771-777.
- Zhang W, Du R, Li B, Zheng X, et al. (2020) Molecular and serological investigation of 2019-nCoV infected patients: implication of multiple shedding routes. Emerg Microbes Infect 9(1): 386-389.
- Kaneko N, Hsuan KH, Boucau J, Farmer JR, Chamard HA, et al. (2020) Loss of Bcl-6-Expressing T Follicular Helper Cells and Germinal Centers in COVID-19. Cell 183(1): 143-157.
- Isenberg DA, Maddison P, Swana G, Skinner RP, Swana M, et al. (1987) Profile of autoantibodies in the serum of patients with tuberculosis, klebsiella and other gram-negative infections. Clin Exp Immunol 67(3): 516-523.
- Gerlach H (2016) Agents to reduce cytokine storm. F1000Research 5: 2909.
- Mohsen B (2020) Cytokine blood filtration responses in covid-19. Blood Purification 50: 141-149.
- Plaze M, Attali D, Petit AC, Blatzer M, Vinckier F, et al. (2020) Repurposing chlorpromazine to treat COVID-19: The reCoVery study. Encéphale 46(3): 169-172.
- Docherty AB, Harrison EM, Green CA, Hardwick HE, Pius R, et al. (2020) Features of 20 133 UK patients in hospital with COVID-19 using the ISARIC WHO clinical characterization protocol: prospective observational cohort study. BMJ 369: m1985.
- Wang D, Hu B, Hu C, Zhu F, Liu X, et al. (2020) Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA 323(11): 1061-1069.
- Carfì A, Bernabei R, Landi F (2020) Persistent symptoms in patients after acute COVID-19. JAMA 324(6): 603-605.
- Osman S, Mirsad M, Selma S (2020) COVID-19 pandemia: neuropsychiatric comorbidity and consequences. Psychiatria Danubina 32(2): 236-244.
- Callaway E (2020) Labs rush to study coronavirus in transgenic animals-some are in short supply. Nature 579(7798): 183.
- Li K, Lenane CW, Perlman S, Zhao J, Jewell AK, et al. (2016) Middle East respiratory syndrome coronavirus causes multiple organ damage and lethal disease in mice transgenic for human dipeptidyl peptidase 4. J Infect Dis 213(5): 712-722.
- Li Y, Man L, Wang M, Zhou Y, Chang J, et al. (2020) Acute cerebrovascular disease following COVID-19: a single center, retrospective, observational study. Stroke Vasc Neurol 5(3): 279-284.
- Talbot PJ, Ekandé S, Cashman NR, Mounir S, Stewart JN (1993) Neurotropism of human coronavirus 229E. Adv Exp Med Biol 342: 339-346.
- Li YC, Bai WZ, Hashikawa T (2020) The neuroinvasive potential of SARS-CoV2 may play a role in the respiratory failure of COVID-19 patients. J Med Virol 92(6): 552-555.
- Gautier JF, Ravussin Y (2020) A new symptom of covid-19: loss of taste and smell. Obesity (Silver Spring) 28(5): 848.
- Toscano G, Palmerini F, Ravaglia S, Ruiz L, Invernizzi P, et al. (2020) Guillain-barré syndrome associated with SARS-CoV-2. N Engl J Med 382(26): 2574-2576.
- Zhao H, Shen D, Zhou H, Liu J, Chen S (2020) Guillain-Barré syndrome associated with SARS-CoV-2 infection: causality or coincidence? Lancet Neurol 19(5): 383-384.
- Mathew B, Thomas AP, Alsadair GR, Benedict DM, Timothy RN (2020) Neuropsychiatric complications of Covid-19. BMJ 371: m3871.
- Khandaker G, Dantzer R, Jones P (2017) Immunopsychiatry: Important facts. Psychol Med 47(13): 2229-2237.
- Leboyer M, Berk M, Yolken RH, Tamouza R, Kupfer D, et al. (2016) Immuno-psychiatry: an agenda for clinical practice and innovative research. BMC Med 14(1):173.
- Pandarakalam JP (2020) Immuno-neuropsychiatry. BMJ Rapid response BMJ 371: m3871.
- Eaton WW, Byrne M, Ewald H, Mors O, Chen CY, et al. (2006) Association of schizophrenia and autoimmune diseases: linkage of Danish National Registers. Am J Psychiatry 163(3): 521-528.
- Wright P, Sham P, Gilvarry C, Jones PB, Cannon M, et al. (1996) Autoimmune diseases in the pedigrees of schizophrenic and control subjects. Schizophrenia Res 20(3): 261-267.
- Chen JS, Chao LY, Chen CY, Chang CM, Wu ECH, et al. (2012) Prevalence of autoimmune diseases in in-patients with schizophrenia; nationwide population-based study. Br J Psychiatry 200(5): 374-380.
- Pandarakalam JP (2013) Is autoimmunity involved in the aetiology of schizophrenia? Progress in Neurology and Psychiatry 17(1): 24-28.
- Davison K (2012) Autoimmunity in psychiatry. Br J Psychiatry 200(5): 353-355.
- Nicholson T, Ferdinando S, Krishnaiah R, Anhoury S, Lennox B, et al. (2012) Prevalence of anti-basal antibodies in adult obsessive-compulsive disorder: Cross-sectional study. Br J Psychiatry 200(5): 381-386.
- Koralnik IJ, Tyler KL (2020) COVID-19: A global threat to the nervous system. Ann Neurol 88(1): 1-11.
- Román GC, Spencer PS, Reis J, Buguet A, Faris MEA, et al. (2020) WFN environmental neurology specialty group. J Neurol Sci 414: 116884.
- Wu Y, Xu X, Chen Z, Duan J, Hashimoto K, et al. (2020) Nervous system involvement after infection with covid-19 and other coronaviruses. Brain Behav Immun 87: 18-22.
- Nordvig AS, Rimmer KT, Willey JZ, Thakur KT, Boehme AK, et al. (2020) Potential neurological manifestations of COVID-19. Neurol Clin Pract 11(2).
- Lechien JR, Estomba CMC, Siati DR, Horoi M, Bon SD, et al. (2020) Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): A multicentre European study. Eur Arch Otorhinolaryngology 277(8): 2251-2261.
- Rogers JP, Chesney E, Oliver D, Pollak TA, McGuire P, et al. (2020) Psychiatric and neuropsychiatric presentations associated with severe coronavirus infections: a systematic review and meta-analysis with comparison to the COVID-19 pandemic. Lancet Psychiatry 7(7): 611-627.
- Stainsby B, Howitt S, Porr J (2011) Neuromusculoskeletal disorders following SARS: a case series. J Can Chiropr Assoc 55(1): 32‐39.
- Garg RK, Paliwal VK, Gupta A (2020) Encephalopathy in patients with COVID-19: A review. J Med Virol 93(1): 206-222.
- Scheidl E, Canseco DD, Naumov AH, Bereznai B (2020) Guillain-Barré syndrome during SARS-CoV-2 pandemic: A case report and review of recent literature. J Peripher Nerv Syst 25(2): 204-207.
- Fokke C, Berg B, Drenthen J, Walgaard C, Doorn PA, et al. (2014) Diagnosis of Guillain-barré syndrome and validation of Brighton criteria. Brain 137(Pt 1): 33-43.
- Hao H, Shen D, Zhou H, Liu J, Chen S (2020) Guillain-Barré syndrome associated with SARS-CoV-2 infection: causality or coincidence? Lancet Neurol 19(5): 383-384.
- Mustafa MWM (2020) Audiological profile of asymptomatic Covid-19 PCR-positive cases. Am J Otolaryngol 41(3): 1024833.
- Grewal P, Pinna P, Hall JP, Dafer RM, Tavarez T, et al. (2020) Acute ischemic stroke and COVID-19: Experience from a comprehensive stroke center in Midwest US. Front Neurol 11: 910.
- Leykina I, Mayerb R, Shinitzkya M (1997) Short- and long-term immunosuppressive effects of clozapine and haloperidol. Immunopharmacology 37(1): 75-86.
- Abdelmawla N, Ahmed MI (2009) Clozapine, and risk of pneumonia. Br J Psychiatry 194(5): 468-469.
- Leung J, Hasassri M, Barreto J, Nelson S, Morgan RR (2017) Characterization of admission types in medically hospitalized patients prescribed clozapine. Psychosomatics 58(2): 164-172.
- Stoecker ZR, George WT, Brien JB, Jancik J, Colon E, et al. (2017) Clozapine usage increases the incidence of pneumonia compared with risperidone and the general population: a retrospective comparison of clozapine, risperidone, and the general population in a single hospital over 25 months. International Clinical Psychopharmacology 32(3): 155-160.
- Leon J, Ruan CJ, Verdoux H, Wang CY (2020) Clozapine is strongly associated with pneumonia and other infections: Clinical relevance of the relationship between clozapine and inflammation. General Psychiatry 33(2): e100183.
- Pandarakalam James Paul (2020) Revisiting clozapine in a setting of COVID-19. American Journal of Psychiatry and Neuroscience 8(3): 50-58.
- DeLisi LE, King AC, Targum S (1985) Serum immunoglobulin concentrations in patients admitted to an acute psychiatric in-patient service. Br J Psychiatr 145: 661-665.
- Ganguli R, Rabin BS, Kelly RH, Lyte M, Ragu U (1987) Clinical and laboratory evidence of autoimmunity in acute schizophrenia. Ann NY Acad Sci 496: 676-685.
- Chengappa KN, Ganguli R, Yang ZW, Shurin G, Brar JS, et al. (1995) Impaired mitogen PHA responsiveness and increased autoantibodies in Caucasian schizophrenic patients with the HLA B8rDR3 phenotype. Biol Psychiatr 37(8): 546-549.
- Wilke I, Arolt V, Rothermundt M, Weitzsch Ch, Hornberg M, et al. (1996) Investigations of cytokine production in whole blood cultures of paranoid and residual schizophrenic patients. Eur Arch Psychiatr Clin Neurosci 246(5): 279-284.
- Ganguli R, Rabin BS, Belle SH (1991) Decreased interleukin-2 production in schizophrenic patients. Biol Psychiatr 26(4): 427-430.
- Shintani F, Kanba S, Maruo N, Nakaki T, Nibuya M, et al. (1991) Serum interleukin-6 level in schizophrenic patients. Life Sci 49(9): 661-664.
- Masserini C, Vita A, Basile R, Morselli R, Boato P, et al. (1990) Lymphocyte subsets in schizophrenic disorders. Relationships with clinical, neuromorphological and treatment variables. Schizophr Res 3(4): 269-275.
- Cañas CA (2020) The triggering of post-COVID-19 autoimmunity phenomena could be associated with both transient immunosuppression and an inappropriate form of immune reconstitution in susceptible individuals. Med Hypotheses 145: 110345.
- Amft N, Cruz DD (1996) Post chemotherapy connective tissue diseases-more than just rheumatism? Lupus 5(4): 255-256.
- Margolin L, Cope DK, Sisser RB, GreenspanJ (2007) The steroid withdrawal syndrome: A Review of the implications, aetiology, and treatments. J Pain Symptom Manage 33(2): 224-228.
- Rivoisy C, Tubach F, Roy C, Nicolas N, Mariette X, et al. (2016) Paradoxical anti-TNF-associated TB worsening: Frequency and factors associated with IRIS. Joint Bone Spine 83(2): 173-178.
- Sereti I, Sheikh V, Shaffer D, Phanuphak N, Gabriel E, et al. (2020) Prospective international study of incidence and predictors of immune reconstitution inflammatory syndrome and death in people living with human immunodeficiency virus and severe lymphopenia Clin Infect Dis 71(3): 652-660.
- Somers EC (2020) Pregnancy and autoimmune diseases. Best Pract Res Clin Obstet & Gynaecol 64: 3-10.
© 2021 James Paul Pandarakalam. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






