- Submissions

Full Text
Experimental Techniques in Urology & Nephrology
A Novel Strategic Clipping Technique in Pelvic Lymphadenectomy to Safely Avoid Routine Pelvic Drainage Placement During Laparoscopic Radical Prostatectomy
Vitagliano Gonzalo, Rico Luis*, Rios Pita Hernando and Ameri Carlos
Hospital Alemán, Buenos Aires, Argentina
*Corresponding author: Rico Luis, Aleman Hospital of Buenos Aires Av Pueyrredon 1640 CP 1425, City of Buenos Aires, Argentina email id: luisrico_01@hotmail.com
Submission: June 05, 2020;Published: July 15, 2020
ISSN 2578-0395Volume3 Issue2
Abstract
Purpose: To safely avoid the routine placement of a pelvic drain after laparoscopic radical prostatectomy (LRP), a novel strategic clipping technique during pelvic lymphadenectomy (PLND) is presented to primarily decrease postoperative lymphatic leakage.
Material and Methods: We performed 76 LRP in which pelvic drainage was not placed. We described a novel technique in PLND and evaluated results and postoperative complications.
Results: Only one patient (1.31%) presented a complicated lymphocele that required percutaneous drainage under computed tomography guidance and antibiotic therapy. There were no cases of urinoma or anastomotic stricture.
Conclusion: The performance of a strategic clipping technique during the PLND associated with a bladder neck preservation and absence of urinary leakage allowed us to safely avoid the placement of a pelvic drain after the LRP.
Keywords: Prostate cancer; Radical prostatectomy; Pelvic lymphadenectomy; Pelvic drainage
Introduction
The active treatment (surgery or radiation therapy) is recommended for the localized prostate cancer [1]. Placement of a pelvic suction drain following a laparoscopic radical prostatectomy (LRP) is considered routine, however, there is limited evidence on the real benefit of a routine pelvic drainage [2]. The presence of lymphatic metastases is a prognostic factor of relevant importance in prostate cancer. Pelvic lymphadenectomy (PLND) is the most reliable tool when staging lymphatic involvement compared to available imaging studies and also prolongs survival in patients with lymph node metastasis [3]. The perioperative complication rate after laparoscopic PLND is approximately 4-20% and the lymphocele formation is the most frequent [4]. The subclinical lymphocele rate ranges from 27-30% [5], although most of them usually occur asymptomatically, the presence of abdominal pain with fever or sepsis may suggest the presence of lymphoceles that require percutaneous drainage [6]. The most frequent reasons for the placement of a routine drainage were PLND, an inaccurate anastomosis between the urethra and urinary bladder and intraoperative factors (bleeding). However, it has been associated with surgical site infections, pain at the drain site, bleeding, arteriovenous fistulae, pseudoaneurysms and retained foreign bodies requiring reoperation [7]. To safely avoid the routine placement of a pelvic drain after LRP, a novel strategic clipping technique during PLND is presented in order to primarily decrease postoperative lymphatic leakage.
Materials and Methods
Following institutional review board approval, we used retrospectively our institutional database to identify 182 patients who underwent LRP from November 2016 to October 2019. One hundred six patients were excluded from the analysis (placing a postoperative drain) because did not present de inclusion criterial. There were 76 cases in which drain was not placed and we performed the novel technique in our PLND and represent the patients included in our study. We performed a 5 port transperitoneal technique with the modifications of the Walsh technique [8]. Bladder neck preservation with careful dissection was performed when possible and the urethrovesical anastomosis was performed using the Van Velthoven technique with 2 continuous barbed sutures [9]. At the end of it, approximately 100ml of saline solution was instilled through a urethral catheter into de bladder to check for the presence or absence of urine leakage. The extended PLND was performed under the novel strategic clipping technique which consists of placing polymer clips (Hem-O-Lok) in previously selected strategic areas: the bifurcation of the primitive iliac artery, the distal limit of the iliac lymph node package, distal and proximal limits of the obturator lymph node package and visible adenopathy (Figure 1 & 2). The Hem-O-Lok clip is a nonabsorbable polymer clip with lock-engagement feature as well as teeth within the jaws, all of which may provide greater security.
Figure 1: Proximal clipping with polymer clip. U: Ureter EIV: External iliac vein
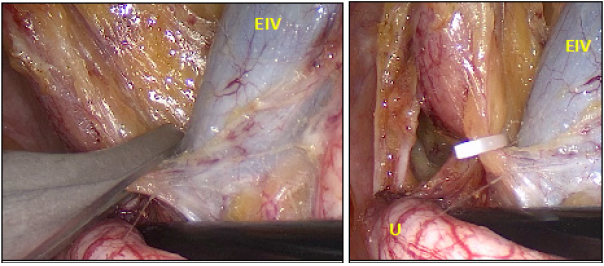
Figure 2: Distal clipping with polymer clip. ON: obturator nerve EIV: External iliac vein EIA: External iliac artery

The placement of a pelvic drain was omitted when the following criteria were met:
1. Bladder neck preservation
2. Absence of urine leakage
3. PLND with strategic clipping of lymphatic vessels
We consider these three points as fundamental and essential and were the inclusion criterial of this study. Those patients in whom these points were not perform were excluded. Routine imaging studies were not performed except in the presence of symptoms and all patients were discharged between the second and third day after admission. Catheters were removed on postoperative day 10 or 12 and we do not performed cystography before catheter removal.
Results
Routine placement of a pelvic drainage was avoided in 76 patients. The mean patient age was 61 years (range: 51-72) and the mean follow-up was 14±3.7 months (range: 2-34). (Table 1) lists patients characteristics. Bilateral extended PLND was performed in all patients using the technique described and the mean number of lymph nodes retrieved from the dissection was 14 (range: 7-22). Estimated blood loss was 200ml (range: 70-600) and no patient received a homologous transfusion. All the patients were followed up in the postoperative period. Only one patient (1.31%) presented a complicated lymphocele that required percutaneous drainage under computed tomography guidance and antibiotic therapy. There were no cases of urinoma or anastomotic stricture, and most common postoperative complications are shown in (Table 2).
Table 1:

Table 2:

Discussion
The placement of a pelvic drain in LRP has been a standard component of the procedure despite not having demonstrated an evidence-based benefit. Savoie et al. [10] were the first to describe the drainage-omission after 85 conventional radical prostatectomies. They reported a single case of a significant urinoma and no case of anastomosis stricture [10]. The same authors update this concept in a more recent publication with 552 patients and showed similar results [11]. Drain placement has not been shown to prevent complications at the time of another surgeries and is possibly associated with longer operative times, longer postoperative hospital stays and additional drain-related complications [12]. However, there are some studies suggest that a pelvic drain may be required for extended PLND [13]. Despite this suggestion, all our PLND were extended and bilateral. All reports agree that postoperative complications related to the pelvic drainage are uncommon and this is what is shown in our report. The rate of development of a symptomatic lymphocele during PLND depends on the surgical technique. Initially, Fried et al. [14] described a symptomatic and subclinical lymphocele rates of 1% and 30.4% respectively after laparoscopic PLND in 111 patients. In a more recent study, Orviedo et al. [15] reported a 51% development rate of lymphoceles after robot-assisted radical prostatectomy and 15.4% were symptomatic. The authors highlight the protective role of the peritoneum by promoting the lymphatic reabsorption [15]. In the present study, all our LRP were performed trans peritoneally and could lead to a low rate of development of clinical relevance lymphocele [1.31%]. It is important to understanding of the routes of lymphatic drainage. Sobotta el al. [16] described the lymphatic system as being predominantly a network of interconnected capillaries that form lymphatic plexuses composed of stems and trunks with interposition of nodes at the root of the main stems and trunks. These lymphatic vessels drain into the periprostatic subcapsular network from which three groups of ducts originate: the ascending duct from the cranial prostate drains into the external iliac nodes, and the lateral ducts to the internal iliac nodes and the posterior duct drain from the caudal prostate to the presacral nodes. According to lymphography studies, the four main regions for these ducts are: the internal iliac group as the primary region, the obturator nodes as the secondary, the external iliac as the tertiary, and the presacral nodes as the quaternary region [17]. Multiple advantages have been described in the bladder neck preservation technique, especially in terms of urinary continence. Furthermore, a larger bladder neck diameter can result in the need for time consuming, reconstructive tapering, which may increase susceptibility to anastomotic leak as a result of the longer suture line [18]. Postoperative cystography was not performed in any patient of our study. Contrast extravasation was historically described between 67-78% during the first five or eight days and 34% after seven days after conventional radical prostatectomy [19]. Currently, the series show minor results being 12% at 5 days of LRP and 4.6% at 10 days after robot-assisted radical prostatectomy [20]. Limitations of this study include the short-term follow up, however, most complications influenced by drain placement occur within the first few weeks or months after surgery.
Conclusion
In conclusion and in our experience, the performance of a strategic clipping technique during the PLND allowed us to safely avoid the placement of a pelvic drain after the LRP. Until such time, on the basis of the current analysis we have eliminated routine drain use in patients undergoing LRP.
References
- Mottet N, Bellmunt J, Briers E, Bolla M (2018) EAU-ESTRO-ESUR-SIOG guidelines on prostate cancer. EAU Annual Congress, Copenhagen, Denmark.
- Niesel T, Partin AW, Walsh PC (1996) Anatomic approach for placement of surgical drains after radical retropubic prostatectomy: long-term effects on postoperative pain. Urology 48(1): 91-4.
- Matthew EH, Jeffrey KM, Christian PP (2010) Pelvic node dissection in prostate cancer: extended, limited, or not at all? Curr Opin Urol 20(3): 211-217.
- Briganti A, Felix C, Andrea S, Nazareno S, Andrea G, Luigi FDP, et al. (2006) Complications and other surgical outcomes associated with extended pelvic lymphadenectomy in men with localized prostate cancer. Eur Urol 50(5): 1006-1013.
- Naselli A, Rossana A, Carlo I, Vincenzo F, Paolo P (2010) Predictors of symptomatic lymphocele after lymph node excision and radical prostatectomy. Urology 75(3): 630-635.
- Michael M, Virgilijus K, Ulla Roggenbuck, Darko Kroepfl (2008) Complications of pelvic lymphadenectomy in 1,380 patients undergoing radical retropubic prostatectomy between 1993 and 2006. J Urol 179(3): 923-929.
- Musser JE, Assel M, Guglielmetti GB, Prachee P, Jonathan LS, et al. (2014) Impact of routine use of surgical drains on incidence of complications with robot-assisted radical prostatectomy. J Endourol 28(11): 1333-1337.
- Walsh PC (1998) Anatomic radical prostatectomy: evolution of the surgical technique. J Endourol 160(6 Pt 2): 2418-2424.
- Van Velthoven RF, Ahlering TE, Peltier A, Skarecky DW, Clayman RV (2003) Technique for laparoscopic running urethrovesical anastomosis: the single knot method. Urology 61(4): 699-702.
- Savoie Marc, Mark SS, Sandy SK, Manoharan M (2003) A pelvic drain may be avoided after radical retropubic prostatectomy. J Urol 170 (1): 112-114.
- Araki M, Manoharan M, Sachin V, Alan M N, Mark SS (2006) A pelvic drain can often be avoided after radical retropubic prostatectomy-an update in 552 cases. Eur Urol 50(6): 1241-1247.
- Wang Z, Chen J, Su K, Dong Z (2011) Abdominal drainage versus no drainage post gastrectomy for gastric cancer. Cochrane Database Syst Rev 5: CD008788.
- Heidenreich A, Ohlmann CH (2005) The role of pelvic lymphadenectomy in the therapy of prostate and bladder cancer [in German]. Aktuelle Urol 36(3): 219-229.
- Freid RM, D Siegel, AD Smith, GH Weiss (1988) Lymphoceles after laparoscopic pelvic node dissection. Urology 51(5): 131-134.
- Orvieto MA, Rafael FC, Sanket C, Kenneth JP, Bernardo R, et al. (2011) Incidence of lymphoceles after robot‐assisted pelvic lymph node dissection. BJU Int 108 (7): 1185-1190.
- Sobotta J (1907) Atlas and textbook of human anatomy, vol. 3: vascular system, lymphatic system, nervous system and sense organs. WB Saunders 62, Philadelphia, USA.
- Malmström PU (2005) Lymph node staging in prostatic carcinoma revisited. Acta Oncologica 44(6): 593-598.
- Freire MP, Weinberg AC, Lei Y, Soukup JR, Lipsitz SR, et al. (2009) Anatomic bladder neck preservation during robotic-assisted laparoscopic radical prostatectomy: description of technique and outcomes. Eur Urol 56(6): 972-980
- Leibovitch I, G Rowland, J S Little, R S Foster, R Bihrle, et al. (1995) Cystography after radical retropubic prostatectomy: clinical implications of abnormal findings. Urology 46(1): 78-80.
- Guazzoni G, Andrea C, Richard N, Matteo R, Antonia C, et al. (2006) Intra-and peri-operative outcomes comparing radical retropubic and laparoscopic radical prostatectomy: results from a prospective, randomised, single-surgeon study. Eur Urol 50(1): 98-104.
© 2020 Rico Luis. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






