- Submissions

Full Text
Experiments in Rhinology & Otolaryngology
SOM CLEFT Scoring/Staging System for Tympanomastoid Cholesteatoma
Ramadan H Sayed1, Mahmood A Hamed1* and Seiichi Nakata2
1Department of Otorhinolaryngology, Sohag University, Egypt
2Department of Otorhinolaryngology, Fujita Health University School of Medicine, Japan
*Corresponding author: Mahmood A Hamed, Department of Otorhinolaryngology, Sohag Faculty of Medicine, East District, 82524, Sohag, Egypt
Submission: April 16, 2018;Published: May 01, 2018

ISSN: 2637-7780Volume1 Issue4
Introduction
Cholesteatoma has the capacity for invasion, migration, change in differentiation, proliferation and recurrence which is quite similar to neoplasms; however, with no DNA affections, that rules out the hypothesis of being a neo plastic lesion [1,2]. Bone erosion is a fundamental biologic behavior of cholesteatoma that can cause serious complications. It is stimulated by a variety of factors that are still debated [3,4]. In the early 1962, the American Joint Committee for Cancer Staging reported a standardized and widely accepted staging system for head and neck cancer. This allowed precise definition of the disease stage, planning treatment protocol and comparing outcome between different modalities and surgeons [5]. On the other hand, tympanomastoid cholesteatoma has no accepted scoring or staging system up till now. Many classifications/staging were adopted worldwide to find a universally accepted staging system to allow a meaningful exchange of information, standardize decisions regarding selection of the proper surgery, compare the outcome and expect prognosis; [6-17] yet, all these attempts failed to gain acceptance due to lack of clinical relevance [9,11]. With the exception of the Japan Otological Society (JOS) scoring/staging system and the European Academy of Otology and Neuro-otology system, none of these systems were authorized [10,12,17,18]. This staging dilemma of cholesteatoma is probably attributed to many variables including etiopatho genesis, location, extent, destructive capacity, variable surgical procedures and functional outcomes.
Review of Literature
First, Lien et al. [6] reported the CAO classification and staging, where “C” referred to the extent of cholesteatoma, “A” degree of tympanic membrane atelectasis and “O” the severity of ossicular destruction. A year later, Austin et al. [7] proposed another classification of chronic otitis media and staged the extent of cholesteatoma into four groups. This was followed by a more detailed attempt done by Meyerhoff et al. [8] who classified cholesteatoma on the basis of patho physiologic types, sites, Eustachian tube function, condition of the ossicular chain at surgery and presence or absence of complications. Saleh et al. [9] proposed the SOC staging system according to the site of origin and extension(S), ossicular damage (O) and preoperative complications (C). They used seven sites including the attic, antrum, middle ear, mastoid, Eustachian tube, labyrinth and middle cranial fossa. In 2009, Telmesani et al. [11] utilizing the microscope and endoscope with thin cuts CT scan, proposed the ATM clinical staging system based on the extent of the disease in the attic (A), tympanum (T) and mastoid (M).
The committee on nomenclature of JOS proposed a consensus based cholesteatoma staging system that was modified over years. It started in 2008 for staging of pars flaccida (attic) cholesteatoma [10] and expanded in 2010 to include the two major types of retraction pocket cholesteatoma, pars flaccida and pars tensa cholesteatoma [12]. They divided the tympanomastoid space into four sections: protympanum (P), tympanic cavity (T), attic (A) and mastoid (M). Accordingly, three stages were defined: stage I, the lesion is confined to the attic or the tympanic cavity; stage II, the lesion extends beyond the attic or the tympanic cavity; and stage III, intra temporal and/or intracranial complications.
Their final version was declared in 2015 to expand the range of cholesteatoma by adding cholesteatoma secondary to a tensa perforation and congenital cholesteatoma. They classified cholesteatoma into 4 stages: stage I: cholesteatoma localized in the primary site; stage II: cholesteatoma involving two or more sites; stage III: cholesteatoma with extra cranial complications and/or intra temporal pathologic conditions; stage IV: cholesteatoma with intracranial complications [17]. In 2012, Belal et al. [14] presented the TMC staging system, where (T) referred to the site of pathology in the tympanic cavity, (M) mastoid cavity involvement and (C) the presence of complications and staged cholesteatoma into 5 stages. Many other attempts for cholesteatoma staging were documented in the literature including Maresh et al. [13], TMC radiological classification [15] and the revised CAO staging system assumed by Kuo et al. [16]. Finally, the European Academy of Otology and Neuro otology and the Japan Otological Society (EAONO/JOS) collaborated and produced a Joint Consensus Statements on definitions, Classification and Staging of Middle Ear Cholesteatoma, presented at the 10th International Conference on Cholesteatoma and Ear Surgery in Edinburgh, June 5–8, 2016. The final copy of this system was published in 2017, and was similar to that of the final version of the JOS staging [18]. We adopted a newly designed, objective, broad and decisive scoring/staging system, similar to cancer staging, for all types of tympanomastoid cholesteatoma that focused on the basic biologic behavior of this serious disease to cover all the black spots of the previous scoring and staging systems.
Materials and Methods
We created a scoring system to document the degree of tympanomastoid cholesteatoma aggressiveness. Our scoring system relied on clinical, radiological findings on a high resolution, thin cuts CT scanning of temporal bone and brain, confirmed with the intra operative findings. All bony items in the tympanomastoid area that could be affected by cholesteatoma were listed and scored in an ascending scale with a minimal score of 0 and a maximum one of 3. These items include: scutum (S), ossicles (O), mastoid air cells (M), posterior bony canal wall (C), lateral sinus plate (L), inner ear structures including SCCs and cochlea (E), facial nerve canal (F) and tegmen (T). These items were termed collectively SOM CLEFT scoring system. Cranial and intracranial complications were included as the highest score for each bone specifically in Table 1. Concerning the facial canal dehiscence, we followed Aslier et al. [19]. Who scored only cases with direct contact with cholesteatoma debris and/or granulation tissue [19]. According to our scoring system we classified cholesteatoma cases into 2 distinct groups.
Table 1: SOM CLEFT scoring system for cholesteatoma aggressiveness.
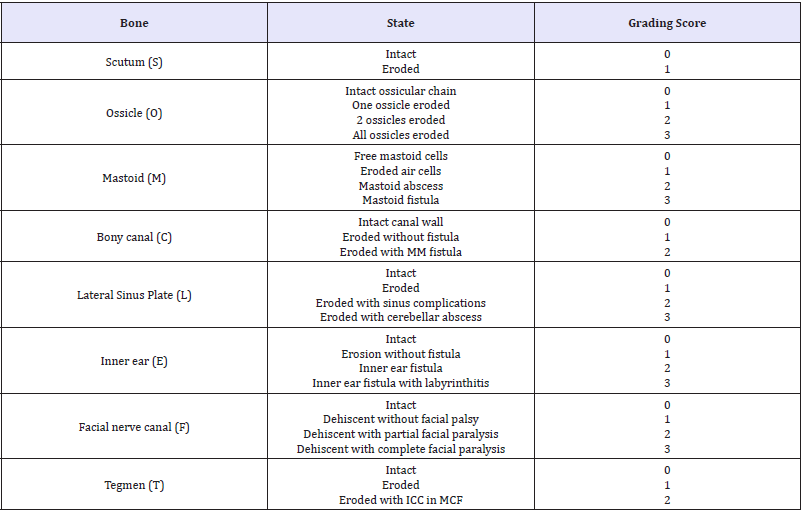
MM: Meato Mastoid; ICC: Intra Cranial Complications; MCF: Middle Cranial Fossa
Group (A)
Non-invasive cholesteatoma, cholesteatoma confined to the ME cavity with no or limited bone erosion involving the scutum and/ or ME ossicles. This type of cholesteatoma included all cases with S0-1O0-3.
Group (B)
Invasive cholesteatoma, cholesteatoma that extended beyond the confines of ME cavity to involve the mastoid air cells, with bone erosion of non ossicular (dangerous) structures, and/or crania land intracranial complications. This type of cholesteatoma included all cases with SanyOany+ any Mastoid CLEFT cholesteatoma. To be validated, we used the main framework of this scoring system in a previous original published data based on immune histo chemical detection of Ki-67, cytokeratin 13 and cytokeratin 17 in cholesteatoma tissues collected from 19 patients with nonrecurrent acquired cholesteatoma compared to 8 deep meatal skin tissues used as control. Based on the number of eroded bones seen radiologically and confirmed intraoperatively, our patients were classified into 2 groups; non-invasive (score 0~3, n=9) and invasive (score 4 or more, n=10). In that work, we found that both the proliferation/differentiation markers, Ki-67 and CK-17, were over expressed in the invasive group of cholesteatoma, and positively correlated with the grading score of bone erosion. In the present work, we added few modifications to our previously assumed scoring system to specify it more. Our new scoring system ranged from 0~20according to the extent of cholesteatomatous bone erosion and complications.
Results
Based on our scoring system, we could stage cholesteatoma into 5 stages (Table 2):
Table 2: Staging of cholesteatoma and topography.
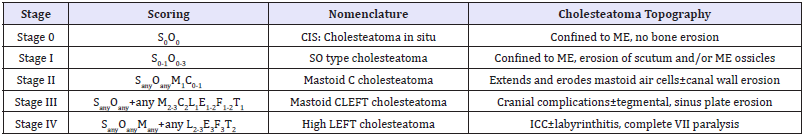
a) Stage 0: CIS (Cholesteatoma in situ) = S0O0 cholesteatoma i.e. cholesteatoma confined to the ME cavity with no evidence of bone erosion.
b) Stage I: S0-1O0-3(M,I,S) Cholesteatoma i.e. cholesteatoma confined to the ME cavity with limited bone erosion of the scutum and/or ME ossicles (malleus, incus and stapes).
c) Stage II: SanyOanyM1C0-1 Cholesteatoma i.e. cholesteatoma extended to and eroded the mastoid air cells with or without bony canal wall erosion.
d) Stage III: SanyOany+ any M2-3C2L1E1-2F1-2T1 cholesteatoma i.e. tympanomastoid cholesteatoma with cranial complications (mastoid abscess/fistula, meato mastoid fistula, labyrinthine fistula, incomplete facial nerve paralysis) with or without tegmental and sinus plate erosion.
Stage IV: SanyOanyMany+ any L2-3E3F3T2 cholesteatoma i.e. tympanomastoid cholesteatoma with intracranial complications and/or labyrinthitis, complete facial nerve paralysis.
This immunohistochemically confirmed scoring system documented cholesteatoma aggressiveness into 2 distinct groups, staged into 5 clinical stages that followed the protocol of cancer staging: Non-invasive cholesteatoma: including CIS (stage 0) and the SO type cholesteatoma (stage I) and Invasive cholesteatoma: included stages II~IV i.e. Mastoid CLEFT types of cholesteatoma.
Discussion
Many scoring/staging systems had been proposed for cholesteatoma; however, none of them gained acceptance. These reports focused either on cholesteatoma types [8,10,12,17] that are actually academic and of little clinical importance; or cholesteatoma extension [6-12] with or without ossicular erosion and associated complications [14-17]. Bone erosion is the most serious biologic behavior of cholesteatoma. Despite that, many scoring/staging systems focused only on ossicular erosion and neglected the more serious non ossicular bone erosion that could pave the way for serious complications [6,8,9,13,16,17]. In other systems, bone erosion was neglected completely [10,12,14].
Complications are the most serious outcome of cholesteatoma that could be fatal in some occasions. Surprisingly, many of these systems, as Meyerhoff et al. [8], SOC staging system [9], TMC staging system [14,15] and JOS staging systems [10,12,17] didn’t specify complications, mentioned them as numbers or collectively in broad terms. Other systems, like CAO classification, Austin et al. [7], Maresh et al. [13] rating system and the revised CAO staging system [16] neglected complications completely. Both Austin and ATM staging systems neglected both bone erosion and complications [7,11]. Actually, staging systems that neglected the most serious biologic behavior of cholesteatoma will be deceiving in evaluating this serious disease and should not be standardized. The last version of the JOS staging system and the EAONO/JOS system [17,18,20] although more broad and detailed ones that authorized by academic societies; yet, they are less decisive, more complicated, and time consuming systems that ignored non ossicular bone erosion and scored complications broadly. We suggested a new, strictly objective, broad, specific and decisive, simple and easily applicable scoring system for different types of tympanomastoid cholesteatomas. Our system was based on reporting eroded bones seen radiologically and confirmed intraoperatively, and various cranial and intracranial complications. Broad system means that it includes all temporal bone structures that could be eroded by cholesteatoma and various complications. Specific and decisive mean that it accurately specifies and scores each eroded bone in an ascending scale, and scores variant complications as the highest score for each related bone specifically. Thus, this scoring system gives a clear idea about cholesteatoma extension, aggressiveness and complications. In 2003, Sayed et al. [11] stated that cholesteatoma behaves like a locally malignant disease, yet it is non-neo plastic. Our staging system was based on the disease aggressiveness, similar to cancer staging protocols. According to the American Joint Committee [5] for Cancer Staging, the mere cartilage invasion in laryngeal carcinoma upgrades the disease from stage I to stage IV as it opens the way for cancerous spread outside the laryngeal confines. By emulating cancer staging protocol to cholesteatoma, extension of the disease beyond the ME confines, with bone erosion of non ossicular (dangerous) structures and/or cranial, intracranial complications upgrade cholesteatoma, not the mere extent of cholesteatoma, nor its less important effect on ME ossicles. Stage 0 (Cholesteatoma in situ) refers to cholesteatoma localized to the ME cavity with no structural invasion [21].
In stage I (SO type cholesteatoma), it is still localized to the ME cavity with erosion of the scutum and/or ME ossicles. Erosion of the scutum declares cholesteatoma to the otologist without opening the way for any serious effects. Ossicular erosion might necessitate ossiculo plasty; yet, the disease still has no serious outcomes. At these stages, cholesteatoma is considered non-aggressive and noninvasive and can be managed with a limited surgical approach. Stages II~IV classify the disease as an aggressive and invasive one that extends to the mastoid air cells, with bone erosion of non-ossicular temporal bone structures, and/or complications necessitating more radical surgery. This distinction was confirmed byan immune histo chemical study done by the authors and others to put an objective distinction between non-invasive and invasive cholesteatomas [20]. Being simple, decisive, specific and broad, one can say that our scoring/staging system is more advantageous than those described in previous reports. However, this system needs further researches and discussions to be universally approved and comparable to cancer staging systems.
Conclusion
SOM CLEFT scoring/staging system is a newly designed, objective, broad, decisive system for all types of tympanomastoid cholesteatoma that focused on the basic biologic behavior of this serious disease and covers all the black spots of the previous systems. It gives all the merits of cancer staging. Moreover, it was confirmed by an immune histo chemical study. Our system needs to be discussed and criticized by different otologic societies to gain a worldwide acceptance similar to cancer staging. Further researches are needed to correlate between our staging system and outcome of cholesteatoma surgery.
References
- Desloge RB, Carew JF, Finstad CL, Steiner MG, Sassoon J, et al. (1997) DNA analysis of human cholesteatomas. Am J Otol 18(2): 155-159.
- Albino AP, Kimmelman CP, Parisier SC (1998) Cholesteatoma: a molecular and cellular puzzle. Am J Otol 19(1): 7-19.
- Bujía J, Schilling V, Holly A, Stammberger M, Kastenbauer E (1993) Hyper proliferation associated keratin expression in human middle ear cholesteatoma. Acta Otolaryngol 113(3): 364-368.
- etiopathogenesis, Wagner M, Bernal SM, Ebmeyer J, Dazert S, et al. (2004) Etio pathogenesis of cholesteatoma. Eur Arch Otorhinolaryngol 261(1): 6-24.
- American Joint Committee on Cancer (1962) Staging and end results reporting: clinical staging system for cancer of the larynx. pp. 1-2.
- Lien CF (1984) Staging of attic cholesteatoma. Chin Med J 33: 438-442.
- Austin DF (1985) Reporting results in tympanoplasty. Am J Otol 6(1): 85-88.
- Meyerhoff WL, Truelson J (1986) Cholesteatoma staging. Laryngoscope 96(9): 935-939.
- Saleh HA, Mills RP (1999) Classification and staging of cholesteatoma. Clin Otolaryngol Allied Sci 24(4): 355-359.
- Tono T, Okamaoto M, Sakagami M, Okuno T, Hinohira Y, et al. (2008) Staging of middle ear cholesteatoma. Otol Jn 18: 611-615.
- Telmesani L, Sayed H, Bahrani N (2009) Proposed clinical classification of cholesteatoma. Egypt J Ear Nose Throat Allied Sci 10(1): 50-53.
- Tono T, Aoyagi M, Ito T (2010) Staging of middle ear cholesteatoma. Otol Jn 20(2): 743-745.
- Maresh A, Martins OF, Victor JD, Selesnick SH (2011) Using surgical observations of ossicular erosion patterns to characterize cholesteatoma growth. Otol Neurotol 32(8): 1239-1242.
- Belal A, Reda M, Mehana A, Belal Y (2012) A new staging system for tympano mastoid cholesteatoma. Int Adv Otol 8(1): 63-68.
- Razek AA, Ghonim MR, Ashraf B (2015) Computed tomography staging of middle ear cholesteatoma. Pol J Radiol 80: 328-333.
- Kuo CL, Shiao AS, Chen CH, Lien CF (2016) Multi dimensional staging system for pediatric acquired cholesteatoma: A 30-year verification data. Auris Nasus Larynx 43(4): 387-394.
- Tono T, Sakagami M, Kojima H (2017) Staging and classification criteria for middle ear cholesteatoma proposed by the Japan Otological Society. Auris Nasus Larynx 44: 135-140.
- Yung M, Tono T, Olszewska E, Yamamoto Y, Sudhoff H (2017) EAONO/JOS Joint consensus statements on the definitions, classification and staging of middle ear cholesteatoma. J Int Adv Otol 13(1): 1-8.
- Aslier M, Erdag TK, Sarioglu S, Güneri EA, Ikiz AO (2016) Analysis of histo pathological aspects and bone destruction characteristics in acquired middle ear cholesteatoma of pediatric and adult patients. Int J Pediatr Otorhinolaryngol 82: 73-77.
- Hamed MA, Nakata S, Shiogama K, Suzuki K, Sayed RH, et al. (2017) Cytokeratin 13, Cytokeratin 17, and Ki-67 expression in human acquired cholesteatoma and their correlation with its destructive capacity. Clin Exp Otorhinolaryngol 10(3): 213-220.
- Ramadan HS (2003) One stage reconstruction in management of extensive cholesteatoma. International Congress Series 1240: 121-131.
© 2018 Mahmood A Hamed. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






