- Submissions

Full Text
Environmental Analysis & Ecology Studies
Horizontal Gradient of Polycyclic Aromatic Hydrocarbons (PAHs) in Roadside Soil of a Semi-Arid North Central Part of India-A Case Study
Amit Masih*
Environmental Research Lab, Department of Chemistry, St. Andrew’s College, Gorakhpur, India
*Corresponding author:Amit Masih, Environmental Research Lab, Department of Chemistry, St. Andrew’s College, Gorakhpur, India
Submission: December 06, 2023; Published: February 09, 2024

ISSN 2578-0336 Volume11 Issue5
Abstract
Polycyclic Aromatic Hydrocarbons (PAHs) formed as a result of unfinished burning of organic compounds which may cause toxic, carcinogenic, mutagenic and teratogenic harm. PAHs can be found in industrial wastes, garbage, cigarette smoke, pesticides and flue gases and can contaminate air, water, soil and food. Although more than 100 PAH compounds are detected in nature out of which at least 16 PAHs compound have more harmful effects. The soil is a long-term sink for airborne PAHs and the concentrations of PAHs are influenced by a wide variety of environmental factors i.e. changes in emissions with traffic volume. To observe the spatial variation in the concentration of PAHs in soil the PAHs were detected in roadside soil collected from different distances i.e. 1m, 5m, and 10m from roadside during winter season. The samples were extracted with hexane by ultrasonic agitation. The extracts were then fractioned on a silica-gel column and the aromatic fraction was subjected to HPLC-UV for analysis of 14 priority PAH pollutants specified by Environment Protection Agency (EPA). The total mean concentrations of PAHs were 15.34, 9.25, and 6.45μg g-1 at 1m, 5m and 10m respectively. The results show negative correlation coefficient between concentration of PAHs and distances from the road. The carcinogenic potencies associated with the total B(a)P exposure was also calculated by using Potency Equivalent Factor (PEFs) and found to be 1.34, 0.93 and 0.42μg g-1 at 1m, 5m and 10m from the roadside respectively. Factor analysis suggests that at roadside motor vehicle exhausts are the dominant source of PAH in the soil of Agra.
Keywords: Polycyclic aromatic hydrocarbon; Roadside soil; HPLC; Semi-arid region
Introduction
Polycyclic Aromatic Hydrocarbons (PAHs) are chemicals containing two or more benzene rings in a linear, angular or cluster arrangement. They contain only Carbon and Hydrogen. PAH are usually generated under inefficient combustion conditions, such as insufficient oxygen by primary natural sources which are forest fires and volcanic activity, but most of the PAHs released into the environment arise from anthropogenic sources such as burning of fossil fuels, petroleum refinery, industrial processes, as a constituent of coal tar and motor vehicle exhaust [1,2]. Airborne particulate matter derives from a wide variety of sources. Probably the major sources within most urban areas of developed countries are road traffic emissions, resuspension of soils and road dust [3]. Sources of PAHs in urban atmosphere include automobiles, refineries and diesel generators. Automobile exhaust gases contain many non-polar PAHs such as benzo(a)pyrene, benzo(a)anthracene and benzo(b)fluoranthene which are associated with diseases such as cancer, asthma, and hay fever [4]. The lighter PAH (2-3 rings) are mostly found in the gas phase while the heavier ones are mainly associated with airborne particles. Heavier PAH (with more than three rings) are rapidly attached to existing particles, usually soot particles, by adsorption or condensation upon cooling of fuel gas [5,2]. Atmospheric deposition constitutes the main input of semi-volatile organic compounds to soil [6]. Once entered in the soil they accumulate in horizons rich in organic matter where they are likely to be retained for many years due to their persistence and hydrophobicity [7]. Consequently, soil is an important reservoir for these compounds and exchanges between soils and the atmosphere is a widely studied process [8]. Since PAHs are one of the most serious pollutants because of their carcinogenicity and mutagenicity, it is important to determine the amounts of PAHs from mobile sources in order to investigate possible adverse effects caused by air and soil pollution. Roadsides have received much attention from environmental scientists as this area is heavily exposed to automobile exhaust gases. Many researchers have used highways to obtain pollution data from diesel and petrol cars [4,2].
Besides industries, the city of Agra also suffers from heavy vehicular pollution just as Delhi, Bombay and other Metropolitan cities of India. All these big cities account for more than 72% of the vehicular exhaust out of the total pollution from all sources [9]. In Agra, the main Mahatma Gandhi (MG) Road i.e from Sadar to Dayalbagh carries traffic densities of more than 105 vehicles per day. The local traffic within Agra city mostly depends on the 2 and 3 wheelers, which emit a lot of smoke and other pollutants. The total number of registered vehicles is close to 500000 [10]. According to conservative estimates at least 20 Metric Ton (MT) of diesel is sold in the city every day [11]. Thus, in Agra, traffic congestion has become very common, and this problem is expected to grow in coming years. However, in Agra the most important source of PAHs is expected to be vehicular emissions since motor vehicles contributed as much as 60% of pollution in Agra [10]. Therefore, the present study was a modest attempt to understand the impact of vehicular and other anthropogenic emissions on the roadside soil quality in terms of polycyclic aromatic hydrocarbon concentrations. In India, few studies have reported ambient PAH concentration in Ahmedabad [12], Mumbai [13], Delhi [14]. To our knowledge there has been a shortage of roadside soil PAH studies. Since PAHs are one of the most serious pollutants because of their carcinogenicity and mutagenicity [15] which have drawn attention of the scientific community, it is important to determine the amounts of PAHs in soil as their concentration in soil correlates significantly with the corresponding levels in air [16] and is a good indicator of the surrounding air pollution and the proximity of sources. The aim of this study was to determine the concentration and distribution of PAHs with increasing distances i.e. at 1m, 5m and 10m from roadside. Moreover, Potency Equivalent Factors (PEFs) are also calculated to assess the B(a)P equivalent exposure profiles with increasing distances from the probable sources.
Materials and Methods
Regional site description
Agra the city of Taj Mahal (E=27.1767° N, 78.0081°) is located in the north central part of India about 200 kms South of Delhi in the Indian state of Uttar Pradesh. It is considered as a semi-arid zone as two-thirds of its peripheral boundaries are surrounded by the Thar desert of Rajasthan. In winter the temperature ranges from 5.5 °C to 30.5 °C. The downward wind is west-north-west i.e. WNW 9.4% and north-north-west i.e. NNW 11.8% in winters [6]. The atmospheric pollution load is high because of the downward wind; pollutants may be transported to the different areas mainly from an oil refinery situated in Mathura (50 Kms from the center of Agra City). Measurements of PAHs were conducted at the side of St. John’s College (SJC). It is situated in the heart of Agra city. It is a major residential and commercial area. SJC lies by the side of MG road that carries mixed vehicular traffic. Traffic density is maximum about 105 vehicles per day that prevails throughout the day. A major road crossing halts traffic which results in the production of smoke and total suspended particulate matter by engine idling and gear changes. A major railway station is also located at a distance of 0.2km from the sampling site having a frequency of about 80 trains per day. Figure 1 shows the Map of Agra.
Figure 1:Map of Agra.
Source: https://hoidla.spordimuuseum.ee/?g=agra-city-map-vv-vDXwxouP
Sample collection
The upper 0-5cm soil was sampled at distances of 1m, 5m, and 10m from the kerb parallel to the roadway. A total of 60 soil samples were collected (20 from each distance and bulked together to obtain a representative sample) which is used for analysis. The collected samples were dried at room temperature in a dark room, sieved through 20-mesh sieve and stored in polybags in a refrigerator.
Analytical procedure
20gm air-dried soil sample were extracted for 45 minutes with hexane (30ml) in an ultrasonic bath extractor. The extract was decanted at the rate of 3100rpm in a decanter (Supelco) and then passed through silica-gel column for the purification of the extract [17]. The obtained extract was evaporated by a flow of nitrogen. This dried extract was redissolved in 1ml of acetonitrile for analysis of PAH. The analysis was carried out using a HPLC (Shimadzu LC- 10AD) with UV visible detector. The analytical column was of 250 mm length and 4.6mm i.d; packed with totally porous spherical RP-18 material (Particle size 5μm). Acetonitrile-Water mixture (70:30) was used as mobile phase at a flow rate of 1.5ml/min. Samples of 20μl were injected into the column through the sample loop. For the detection of compounds, the UV detector was set at 254nm for analysis. Standards were obtained individually as the solids from Supelco, USA. The following compounds were quantified: naphthalene, acenaphthene, acenaphthylene, fluorene, phenanthrene, anthracene, fluoranthene, pyrene, benzo(a)anthracene, chrysene, benzo(b)fluoranthene, benzo(k) fluoranthene benzo(a)pyrene and benzo(ghi)perylene. All these compounds are on the USEPA priority pollutants list. The procedure described above has been checked for recovery efficiencies using spiked PAH standards. Recoveries range between 61-78%, with the lower values corresponding to the lower molecular weight PAH compounds. Presented data are corrected accordingly with the means of triplicate analyses. Replicated analyses give an error between ±10-20% for PAH in soils.
Result and Discussions
Spatial variation of PAHs
The PAHs shown in Figure 2 were detected in roadside soil collected from different distances i.e. 1m, 5m, and 10m from roadside. The total mean concentrations of PAHs were 15.34, 9.25 and 6.45μg g-1 respectively. The mean soil concentration trends of individual PAHs at various distances from the road are also illustrated in Figure 2, which shows that the highest concentrations are at 1m from the road, the closest sampling location from the road, and decreases with increasing distance from the road. The decreasing trend of PAH concentrations probably reflects the decrease in the atmospheric concentration further from the road due to dispersion and deposition r=-0.96. This is confirmed by Figure 3 which shows the correlation coefficient between concentration of PAHs and distance from the road to be -0.96. Hence, there is a negative correlation between these two.
Figure 2:Concentration trends of Individual PAHs at different distances from the road.
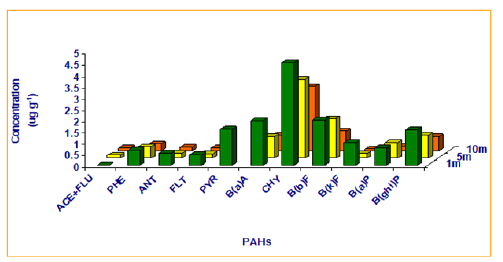
Figure 3:Correlation coefficient between concentration of PAHs and distances from road.
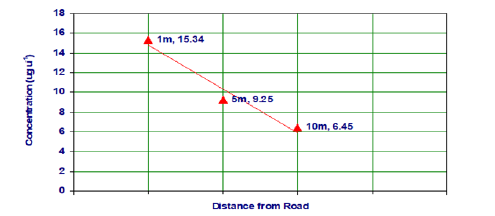
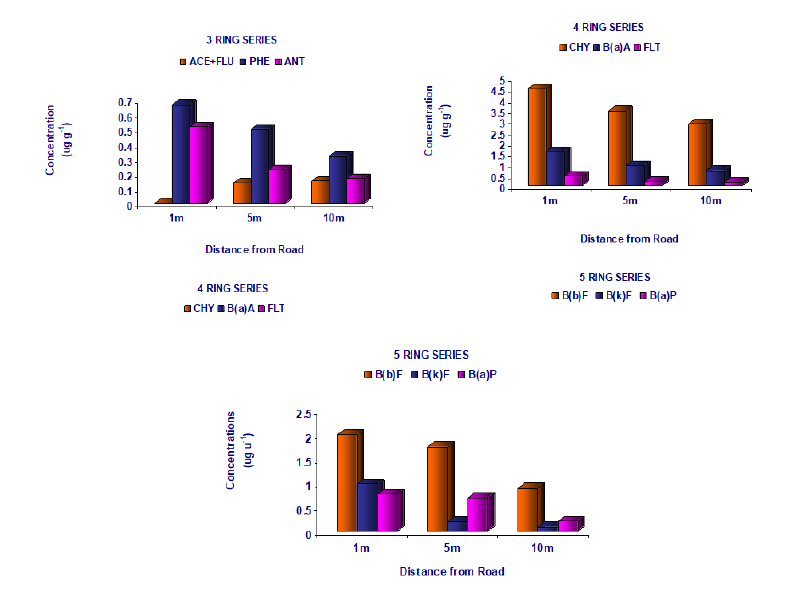
Figure 4 also indicates that there is a decline in the concentrations of relatively high molecular weight PAHs increase in distance when concentrations of PAHs with three rings, four rings, and five rings were plotted versus distance. It was interesting to note that the mean concentrations of PAHs having the highest molecular weight, i.e. those with more than four rings, decrease substantially compared with the concentrations of PAHs with three and four rings. Earlier studies have suggested that PAHs with more than four rings occur almost entirely in the particulate phase [18]. Close to the road, rapid deposition of particles occurs by sedimentation, turbulent diffusion, inertial impaction and interception by vegetation surfaces [19,6]. On the other hand, PAHs with three or four rings exist both in the vapour and particulate phases [20]. These vapors do not sediment out of the atmosphere and can be widely dispersed by winds and vehicle-generated turbulence. It is suggested that these factors lead to the observed pattern of PAH distribution. The soil is a long-term sink for airborne PAHs and the concentrations of PAHs are influenced by a wide variety of environmental factors i.e. changes in emissions with traffic volume [21,22].
Figure 4:Concentrations of PAHs (3, 4, and 5 rings) at various distances from the roadway.
Principal component analysis
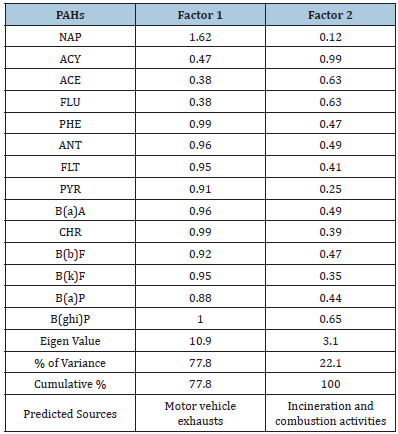
A varimax rotated factor analysis was performed to identify the main sources influencing the PAH concentration at the sampling sites. In this statistical method a set of multiples inter correlated variables is replaced by a small number of independent variables (factors) by orthogonal transformations (rotations). This is achieved by diagnosing the correlation matrix of the variable i.e. by computing their Eigen values and Eigen vectors. Factor loadings obtained after the rotation called varimax rotation gives the correlation between the variables and the factors. Each variable was also evaluated for its KMO value (Keiser Mayer Olvin), which gives sampling adequacy, and data was included in the matrix only if it had Eigen values greater than one. The varimax procedure was adopted for rotation of the factor matrix to transfer the initial matrix into one that was easier to interpret. In the present study, the SPSS (version 10.0) computer software was used to perform factor analysis. Results obtained by varimax rotated factor analysis are given in Table 1. Shown results in the table have loading > 0.5, because they are deemed to be statistically significant. As presented in Table 1, the roadside factor analysis reveals two factors with Eigen values > 1 accounting for 100% of the variance. The first factor contributes 77.8% of the total variance explaining the PAH loads of NAP, PHE, ANT, FLT, PYR, B(a)A, CHR, B(b)F, B(k)F, B(a)P and B(ghi) P. The main source of PAH is vehicular activities as number of the diesel and petrol vehicles especially two and three wheelers are used for local transportation of the public (traffic density is about 105 vehicles per day). The second factor accounts for 22.1% of the total variance, and contains ACY, ACE, FLU and B(ghi)P. This may be related to incineration and combustion activities taking place near roadside. In addition, ratios between pairs of individual PAH have also been calculated. Ratios of Phe/Ant (1.6), B(a)A/B(a)P (2.0) and Pyr/B(a)P (2.8) indicated the PAH origin is likely to be from motor vehicles exhaust [21,1]. Thus, it would appear that at roadside motor vehicle exhausts are the dominant source of PAH in the soil of Agra.
Table 1:Results of factor analysis with varimax rotation on PAHs at Roadside. Loading greater than 0.5 is significant
Assessing PAH exposure profiles
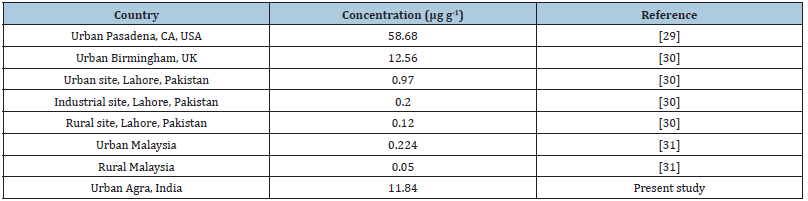
Until now, occupational exposure limit for TPAHs has not been established because of the complexity of PAHs in their chemical composition. Several PAH species including benzo(a)pyrene (as most carcinogenic compound) have been classified into probable (2A) or possible (2B) human carcinogens by the International Agency for Research on Cancer [23,24]. According to the literature [25], the potency equivalent factor for B(a)P is one (1), which is highest among all the PAHs. One approach in estimating the carcinogenic potency associated with the exposure of a given PAH compound can be obtained by calculating its BaPeq for each individual PAH species. In order to calculate the carcinogenic potencies associated with the total PAH exposures from particulate matter; we pragmatically used the sum of each individual BaPeq (i.e., total-BaPeq) as a surrogate indicator. Therefore, in the present study Potency Equivalent Factor (PEF) of the given species relative to B(a)P carcinogenic potency have been used. The above method has the main advantage of being relatively easy to apply in the environments affected by human sources, however, may underestimate risk due to not all PAH, but only limited compounds, are considered [26,27]. For pragmatic purpose, the list of PEFs compiled by Tsai et al. [28] was adopted in this study. Table 2 depicts the total mean concentration of roadside PAHs at 1m, 5m and 10m from roadside having 15.34, 9.25 and 6.45μg g-1 which corresponds to a B(a)P equivalent exposure of 1.34, 1.03 and 0.42 with reference to carcinogenicity, using PEFs given by Tsai et al. [28] respectively. B(a)P contributed the maximum carcinogenic exposure equivalent in the decreasing order of 0.77 at 1m. 0.59 at 5m and 0.21 at 10m which clearly indicates that the carcinogenic exposure risk from PAHs decreases with increasing distance from the road. This decrease has been found to be 72.7% at 5m and 10.38% at 10m. The present study is compared with the work done in other countries in Table 3 which depicts that the concentration of PAHs in roadside soil in Agra is higher than reported in Pakistan and Malaysia whereas, when compared to USA and UK the PAHs concentrations were found to be lower [29-31]. Thus, the molecular weight of a PAH strongly influences its dispersal from the sources at the roadway.
Table 2:B(a)P Potency Equivalency Factors (PEFs) and BaPeq exposure profiles (μg g-1) *PEFs cited by Tsai et al. (2004)
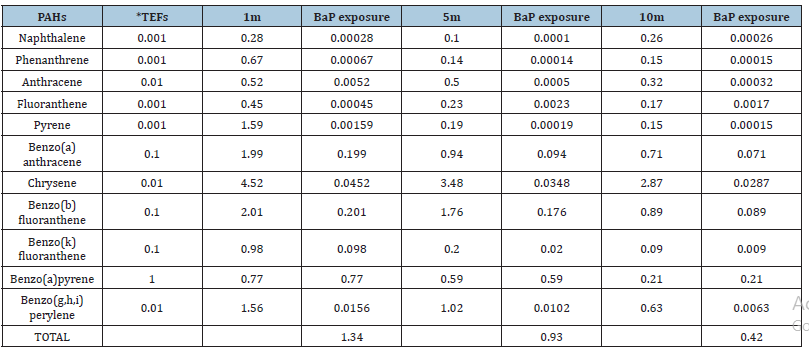
Table 3:Concentrations of roadside soil (PAHs) compiled from literature data.
Conclusion
This investigation was conducted at various distances of an urban roadside and shows that motor vehicles are the major sources of PAHs in soil. Total mean concentrations of PAHs were found to be 15.34, 9.25, and 6.45μg g-1 at 1m, 5m, and 10m respectively. A set of fourteen PAHs was determined in the roadside soil. This study also indicates that the highest molecular weight PAHs (4-6 rings) are deposited in higher proportion to the roadside, probably as particulates and the lower molecular weight compounds with a higher proportion in the vapour form, are more widely dispersed. The results show negative correlation coefficient between concentration of PAHs and distances from the road. A varimax rotated factor analysis was performed which suggests that at roadside motor vehicle exhausts are the dominant source of PAH in the soil of Agra. The carcinogenic potencies associated with the total B(a)P exposure was also calculated which exemplify that B(a)P contributed the maximum carcinogenic exposure equivalent at all the distances from road i.e. 0.77μg g-1 at 1m, 0.59μg g-1 at 5m and 0.21μg g-1 at 10m. This study also indicates that the highest molecular weight PAHs (4-6 rings) are deposited in higher proportion to the roadside and lower molecular weight compounds with a higher proportion in the vapour form are more widely dispersed.
References
- Martin Koller, Saleh HM (2018) Introducing heavy metals. In: El-Din M Saleh, Aglan RF (Eds.), Heavy Metals, IntechOpen, London, UK.
- CBS Mornings (2019) Heavy metals like arsenic and lead found in 45 packaged fruit juices, report finds.
- Eric Estevez (2021) Consumer goods, Investopedia, USA.
- Consumer Reports (2010) Heavy metals: Despite progress, our tests still find products with lead and cadmium.
- Some consumer products contain lead, mercury, arsenic.
- Mark Hyman (2021) Heavy metals and health: The untold story.
- Adam Husney (2020) Chelation therapy, University of Michigan Health, Michigan, USA.
- (2020) India amends its food safety and standards (contaminants, toxins, and residues) regulations, SGS, Geneva, Switzerland.
- (2020) Item no. 4 of the food safety and standards (contaminants, toxins and residues) first amendment regulations.
- (2019) India announces new food packaging regulations.
- Avijit Das, Anindya Das (20187) Heavy metals in common food items in Kolkata, India. Euro Mediterranean Journal of Environmental Integration 3.
- Santhi D, Balakrishnan VB, Kalaikannan A, Radhakrishnan KT (2008) Presence of heavy metals in pork products in Chennai (India). American Journal of Food Technology 3(3): 192-199.
- Mathaiyan M, Natarajan A, Rajarathinam X, Rajeshkumar S (2021) Assessment of Pb, Cd, As and Hg concentration in edible parts of broiler in major metropolitan cities of Tamil Nadu, India. Toxicology Reports 8: 668-675.
- Swati S, Chhaya S (2019) Levels of selected heavy metals in food packaging papers and paperboards used in India. Journal of Environmental Protection 10(3): 360-368.
- Thomas Sumner (2014) Did lead poisoning bring down ancient Rome?
- Sharma RK, Agrawal M, Marshall FM (2009) Heavy metals in vegetables collected from production and market sites of a tropical urban area of India. Food and Chemical Toxicology 47(3): 583-591.
© 2023 © Monalisa Saha. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






