- Submissions

Full Text
Environmental Analysis & Ecology Studies
The Effect of mycorrhizal Species on Growth, Essential Oils, Yield and Morphophysiological Characteristics of Lemon Balm (Melissa Officinalis L.) under Water-deficit Conditions in Tabriz Region
Shamizi N1, Yarnia M2*, Mohebalipor N3, Ali F3 and Ajjali J3
1PhD Student, Faculty of Agriculture, Miyaneh Branch, Islamic Azad University, Iran
2Professor, Faculty of Agriculture, Tabriz Branch, Islamic Azad University, Iran
3Asistant Professor, Miyaneh Branch, Islamic Azad University, Iran
*Corresponding author:Mehrdad Yarnia, Professor, Faculty of Agriculture, Tabriz Branch, Islamic Azad University, Tabriz, Iran
Submission: September 22, 2020;Published: August 17, 2021

ISSN 2578-0336 Volume8 Issue5
Abstract
The aim of this study was investigating the effect of irrigation levels (irrigation after 70, 100, 130 and 160mm evaporation from pan) and mycorrhizal fungi species (Mycorrhiza non-application, Rhizophagusintradices, Funneliformismosseae, G. hoei and combined application of all three species) on growth and physiological attributes of lemon balm over two years and based on combined analysis of variance with a completely randomized block design. The highest yield achieved with from irrigation after 100mm evaporation + application of all three species of mycorrhizal fungi. Irrigation after 100mm evaporation increased this trait by 33% compared to irrigation after 70mm evaporation due to increase in essential oil percentage under dehydration conditions, but dehydration increment a decrease in each two components of essential oil yield (essential oil percentage and dry matter yield) led to a significant decrease in essential oil yield. Dehydration led to an increase in proline content and antioxidant activity. In the present study, despite the diminishing effect of dehydration, somewhat the mycorrhizal use was able to reduce the negative effect of dehydration. In general, given to the results and considering the economic importance of essential oil yield, lower water deficit stress and mycorrhizal application can increase the lemon balm essential oil yield, but planting this plant under severe water deficit condition drastically can decrease essential oil yield.
Keywords:Lemon balm; Dehydration; Mycorrhizae; Essential oil yield
Introduction
Lemon balm (Melissa officinalis) that belongs to the mint family is an aromatic medicinal plant. Many of its therapeutic characteristics are dependent on its essential oils, which are rich in aldehydes and terpenic alcohols [1]. Drought, like other stress, has detrimental effects on plant performance. Drought has various physiological effects on plants. Drought stress reduced leaf potential, stomatal conductance, nitrate recovery and cell elongation. Decreasing in chlorophyll content of plants exposed to dehydration is caused by dehydration and oxidative stress, as well as by photosynthesis of pigments and degradation of chlorophyll [2]. Drought is a multi-dimensional stress that affects plants at different levels in space and time. So the physiological response to Drought is very complex and unpredictable. In fact, the signs of Drought are the discoloration of the leaves from green to gray. At the same time the stomachs are closed and photosynthesis is drastically reduced.
Organic farming is dependent on natural soil microflora that enhances plant growth and yield by employing a variety of beneficial bacteria and fungi, including arbuscular fungi and plant growth promoting bacteria [3]. According to available reports, 60 to 90 percent of the total fertilizer used is lost and only 10 to 40 percent is absorbed by the plant. Studies have shown that microorganisms can play an important role in the integrated management of fertilizers to maintain soil productivity and fertility. Plant growth promoting bacteria and mycorrhizal fungi can increase fertilizer use efficiency [4]. Biofertilizers maintain the soil environment through nitrogen fixation, phosphorus and potassium solubilization or mineralization, release of growth stimulants, production of antibiotics and decomposition of organic matter in the soil, rich in macro and micro elements. Previous studies have shown that mycorrhizal fungi affect plants in a variety of ways, including accelerated growth, nutritional status, water intake, disease resistance and resistance to stress. The response of plants to root colonization by mycorrhizal fungi is largely dependent on the plant type and strain of the fungus and environmental conditions such as soil nutrient levels, light intensity and temperature. Also root colonization of plants by several strains of fungi has more positive effect than single strain application [5].
The presence of arbuscular mycorrhizal fungi is important for ecosystem stability, plant establishment and biodiversity conservation. The role of mycorrhizal fungi in biodiversity and ecosystem function has been well illustrated, in particular by their role in plant diversity and production power. Aram et al. [6] confirmed the positive relationship between plant diversity and colonization of mycorrhizal fungi. Symbiotic fungi increase the nutrient uptake of host plants and can increase plant growth, quality, and resistance to environmental stress [7]. mycorrhizal fungi increase the uptake of nutrients, especially phosphorus, there by enhancing the growth and yield of plants. mycorrhizal fungi, like other fungi, do not spread their spores through the wind, but are transmitted from plant to plant by soil transfer. One of the important effects of mycorrhizal fungi is to increase crop yields, especially in low yield soils. mycorrhizal fungi increase plant resistance to dehydration by increasing water uptake and a number of nutrients such as zinc and copper, improving leaf cell biogenesis, regulating stomatitis activity, root growth and development. mycorrhizal fungi also increase plant resistance to dehydration by regulating plant hormonal activities. So, this study was conducted for determination of the effect of different levels of irrigation and different mycorrhizal species on growth characteristics and yield of essential oil of lemon balm.
Materials and Methods
In order to evaluate the effect of different mycorrhizal species on some morpho-physiological traits of lemon balm under drought, an experiment was conducted during two cropping seasons of 2016- 17 and 2017-18 at the Agricultural Station of Tabriz Islamic Azad University. The area is located 5km from Tabriz, with longitude 46 degrees 17 minutes east, latitude 38 degrees north 5 minutes and altitude 1360 meters above sea level. According to the Domarton climate classification, the region has a semi-arid climate. The average annual temperature is 10 °C, the average annual maximum temperature is 16 °C, and the average annual minimum temperature is 2.2 °C. The average annual rainfall in this area is 272.3mm. The pH of the soils in the region is in the alkaline to medium range. Prior to the experiment to determine the physical and chemical properties of the soil, a depth of 0-30cm was sampled and 8 samples were transferred to the laboratory for analysis. The results of soil analysis showed that the field soil had sandy loam texture, salinity of 1.56ds/m and acidity of 7.53 (Table 1). According to fertilizer recommendations, phosphorus and potassium fertilizers were not used in this study. The experiment was conducted as split plot in a randomized complete block design with three replications. Experimental treatments including different levels of drought stress as the main factor in three levels (A1: Irrigation after 70mm evaporation from Class A pan (control), A2: Irrigation after 100mm evaporation from Class A pan, A3: Irrigation after 130mm evaporation from Class A pan, and A4: Irrigation after 160mm evaporation from Class A pan) and different species of mycorrhiza at five levels as a byproduct including non-use (control), Rhizophagus irregularis (Glomus intradices), Funneliformis mosseae (G. mossae), G. hoci and mixture of all three mycorrhiza species.
Table 1: Results of field soil decomposition during two crop years.

In order to carry out the experiment, land acquisition operations were conducted in March with appropriate climate conditions. On 11 June, after leveling the ground, the plot was dimensioned 3 × 1.5m. Each plot had 3 rows of plants. Sub plot was separated by a plot and 1.5m main plot. On June 16, 2016, irrigation was done before planting. Prior to planting, mycorrhizal species prepared from the Institute of Soil and Water of Iran, according to the experiment map, 9g per plant and in the mixed state 3g per plant was added in the planting place. In this study, 2-4 leaf transplants of lemon balm were used. The transplants were obtained from the Orumieh Golden Plant Institute and transferred to the main field on June 17, 2016, at the first year of the experiment.
Row spacing of 60cm and seedling spacing of 30cm and plant density of 5.5 plants/m2 were considered. Irrigation was performed immediately after planting. In the first week after transplanting, if some of the transplanted crops were destroyed, replanting was attempted. After transplanting, irrigation was done every 2-3 days, and irrigation was performed every seven days until complete plant establishment. The first stage of weed removal took place on July 9, 2016. Weed control was done manually. In order to provide the plant with nitrogen fertilizer based on soil test, urea was applied at 60kg/ha after transplanting and after the first weed removal stage, before irrigation treatments. In the first year of the study, the second stage of weeding was June 22, 2016. After weeding the field on July 26, 2016, the field was re-fertilized. On this date 60g were given per plot and used as a spread. After this date and transplants full establishment, irrigation levels were applied. Each irrigation period was 70, 100, 130 and 160mm evaporation from Class A pan. The daily cumulative evaporation rate from the pan, after reaching the desired value for each treatment (70, 100, 130 and 160mm evaporation from Class A pan), showed the irrigation time. The amount of irrigation water for each treatment was based on the calculated total water requirement of the plant which was calculated by random sampling from three different parts of each plot and determining the weight percent of soil moisture about 24 hours before irrigation. To determine the vegetation coefficient from two equations: ETc=Kc; ET0; and ET0 = Kpan×Ep were used, in which ETC and ETO are reference evapotranspiration and reference evapotranspiration, respectively. During each year, in the treatments of 70, 100, 130 and 160mm evaporation from the pan of evaporation 27, 18, 12 and 10 irrigation rounds, respectively.
After flowering and in all treatment combinations, morphological traits were evaluated by sampling the secondrow competition from each experimental plot. At this stage of the experiment, the measured traits include leaf chlorophyll content index, chlorophyll a and b content, and antioxidant enzymes activity. In addition to the above-mentioned traits in both years of experiment, such as plant height, leaf area index, essential oil content and yield, Flower dry weight, dry matter, prolin content and root colonization with mycorrhiza were evaluated. Given that lemon balm is a perennial plant, the experimental field was ready for use in the second year. After overwintering the plants in the first year and growth of the plants, in the second year all operations were performed, and irrigation levels were applied exactly as in the first year and all traits evaluated in the first year were also evaluated in the second year. Statistical analysis of data was performed with SAS 9.1 software and means comparison were by Duncan multiple range test at 5% level. Charts were drawn with the help of Excel software.
Result and Discussion
In this study, the main effects of irrigation and application of mycorrhizal fungi on plant height, leaf area, flower dry weight and total dry yield were significant, while the interaction between the two treatments was significant in essential oil percentage and essential oil yield (Table 2). Drought stress caused a significant decrease in leaf area per plant, plant height, flower dry weight and total dry matter yield in all of these traits, with the greatest decrease being in irrigation after 160mm evaporation. So that in these traits, irrigation after 160mm evaporation from the pan reduced 36.5, 32.6, 39.3 and 30.3%, respectively (Table 3). Other researchers have also reported a decrease in growth due to dehydration. Gholizadeh et al. [8] in Dracocephalum showed that dehydration reduced plant height. Abbasi et al. [9] found a significant decrease in the height of Agropyron elongatum plant under dehydration. Shaw et al. (2008) reported that dehydration decreases cell growth due to the decrease in turgor pressure. Osmotic regulation can be effective in maintaining cell turnover for survival or maintaining plant growth under drought conditions. Shinozaki & Yamaguchi [10] reported that drought impedes cell development and consequently plant growth and height. Jamali [11] in Coriandrum sativum showed that dehydration caused a significant decrease in leaf area index. The researchers stated that dehydration reduced the leaf area by reducing the each leaf area and number of leaves. Allayi et al. found that dehydration significantly reduced the dry weight of Dracocephalum moldavica. Ghanbari & Ariafar [12] also showed that drought significantly reduced shoot dry weight of pepper mint.
Table 2:Composite analysis of variance of traits studied in lemon balm.
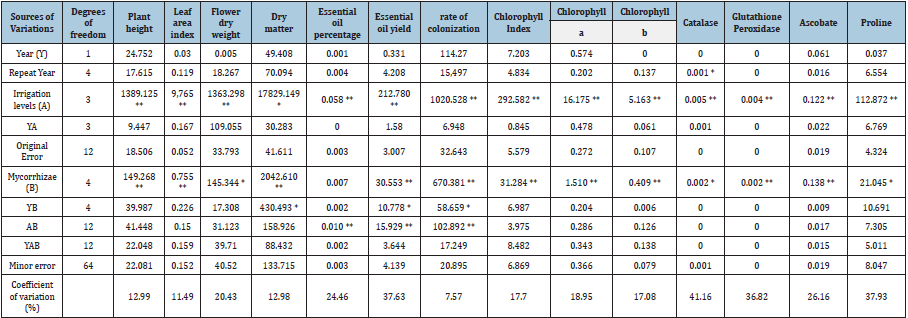
** and* represent significant at the one and five percent probability level, respectively.
Table 3:Comparison of mean traits affected by irrigation levels.
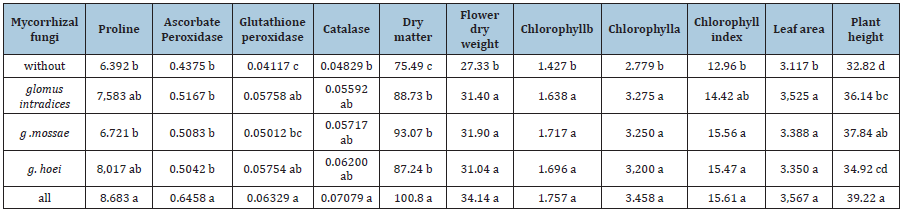
mycorrhizal application significantly increased vegetative traits of lemon balm. In leaf area and total dry weight, all mycorrhizal species had similar additive effects, but in plant height and total dry yield, the highest increase was due to combined application of mycorrhizal species. The combined application of mycorrhizal species increased this two traits by 33.4% and 33.5%, respectively (Table 4). The greater the diversity of mycorrhizal fungi found in soil, the better the absorption of water and nutrients through the mycorrhizas, the better. In this study, it was observed that only the combined application of fertilizers caused a significant increase in the dry yield of lemon balm plants. Murthy & Narayananapa [13] investigated the effect of two strains of Glomus mosseae and G. fasciculatum on plant height of Rutagraveolens. The researchers observed that the strains studied differ in their effects on plant height. Khalili & Yousef (2014) investigated the effect of different levels of phosphorus fertilizer and mycorrhizal fungi application on growth characteristics of the medicinal plant Lepidium sativum. The researchers observed that mycorrhizal fungi combined with phosphorus fertilizer caused a greater increase in plant height of Lepidium sativum.
Table 4: Comparison of mean traits affected by irrigation levels.
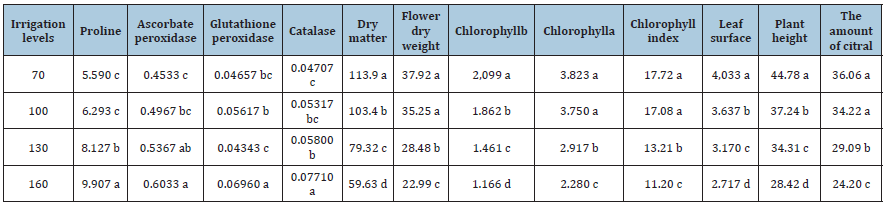
Increasing levels of phosphorus fertilizer resulted in a higher increase in plant height. Arango et al. [14] showed that application of mycorrhizal fertilizer significantly increased leaf area of pepper mint, but different strains of mycorrhizal fertilizer had different effect on total fresh weight. Glomus intraradices was the most effective strain in increasing this trait. In the present study, application of phosphorus fertilizer also caused a significant increase in the pepper mint leaf area, but the effect was dependent on mycorrhizal strain. The highest leaf area was obtained using phosphorus fertilizer and Glomus intraradices. A plant needs numerous growth factors such as fertilizer, water and nutrients for optimal growth. Studies have shown that mycorrhizal fertilizers increase the uptake of water and various nutrients such as nitrogen, phosphorus, iron, zinc and manganese in plants, as these microorganisms increase the root uptake of plants [15]. Murthy & Narayananapa [13] in a study on Rutagraveolens showed that application of mycorrhizal fertilizer significantly increased dry yield of Rutagraveolens. The researchers also found that the combined use of mycorrhizal fertilizers led to a greater increase in the dry yield of Rutagraveolens plants than in each alone. Kumar et al. [16] in a study done on lemon balm, found that the combination of mycorrhizal strains had a greater effect on lemon dry weight than either alone.
According to the results of the present study, the highest and lowest essential oil percentages were obtained with 0.36% and 0.16% in two irrigation treatments of irrigation after 100mm evaporation from pan with application of all three species of mycorrhizal fungi and irrigation after 160mm evaporation + without application of mycorrhizal fungi, respectively. These results indicate that the treatments under study caused significant changes in the percentage of essential oil of lemon balm. By application of Funneliformis mosseae, G. hoei and all three mycorrhizal fungi, reducing irrigation water had not significant effect on essential oil percentage, but with no application of mycorrhizal fertilizer and application of Rhizophagus intradices it was different. In the absence of mycorrhizal fertilizer application and Rhizophagus intradices, irrigation water reduction from irrigation after 70 and 160mm evaporation decreased essential oil by 38.4% and 40.9%, respectively. Therefore, the results of this study show that application of mycorrhizal fertilizers can reduce the negative effect of dehydration on essential oil percentage (Table 5). While Gholizadeh et al. [8] in lemon balm showed that dehydration reduced the essential oil percentage. Andalibi et al. [17] showed that in all plant organs, the percentage of essential oil decreased with increasing amount of available water and the lowest essential oil percentage was observed in treatments that were not drought tolerant.
Table 5:Comparison of mean traits affected by irrigation levels and mycorrhizal species.
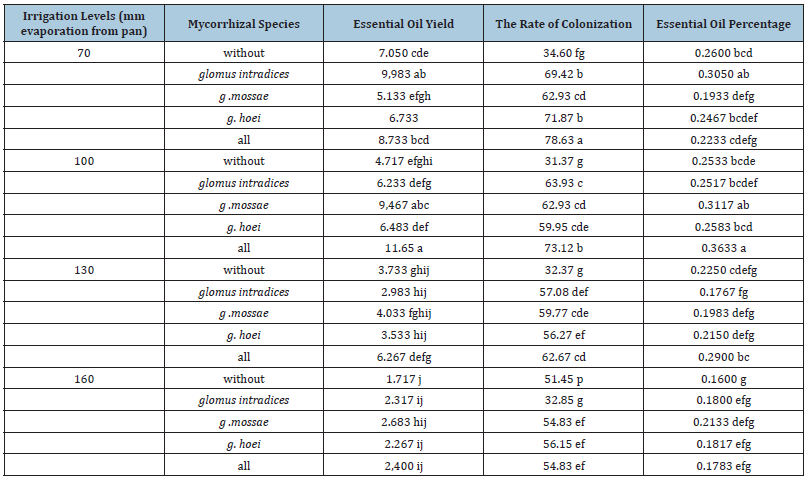
There are no proven reasons for the response of secondary metabolites of medicinal plants to drought stress. Only two hypotheses have been developed about how environmental conditions affect secondary metabolites of these plants. The first hypothesis, carbon balance of nutrients, explains the amount of carbon used to produce secondary metabolites as the balance between photosynthesis and growth. It is hypothesized that when nutrients are available, the plant will allocate carbon for growth. Deficiency of nutrients limits growth more than photosynthesis and results in the formation of carbonate hydrates that produce secondary carbon metabolites. The second hypothesis, or growthdifferentiation equilibrium, states that carbon is spent on growth until conditions allow for cell division and expansion. With the onset of dehydration, growth stops, cells differentiate and secondary metabolite reservoirs, and the carbon plant is devoted to the production of effective pharmaceuticals [17]. In this study, application of mycorrhizal fertilizer only in irrigation treatment after 100mm evaporation had significant effect on essential oil percentage. In this irrigation treatment, application of all three species of mycorrhizal fungi resulted in an increase of 43.26% in lemon balm essential oil percentage. An increase in the percentage of essential oils of medicinal plants by using mycorrhizal fertilizer by Zolghafari et al. [17] was also reported in Ocimum basilicum. Zolfaghari et al. [17] showed that the effect of different strains of mycorrhizal fungi was different in terms of the percentage of essential oil of Ocimum basilicum.
The highest essential oil yield was obtained after 100mm evaporation from pan + application of all three species of mycorrhizal fungi. In this treatment, essential oil yield was 11.65g/m2, while the lowest essential oil yield was 1.7g/m2 in irrigation treatment after 160mm evaporation from pan + no application of mycorrhizal fertilizer. According to the results, moderate dehydration not only had not a negative effect on essential oil yield, but also significantly increased this trait. So that in application of Funneliformis mosseae and application of all three species of mycorrhizal fungi by reducing irrigation water from irrigation after 70 and 100mm evaporation from pan, essential oil yield increased by 84.4 and 33.4%, respectively, while in application of Rhizophagus intradices, essential oil yield decreased after 100mm evaporation from pan (Table 5). Moderate drought stress can stimulate essential oil yield by stimulating the production of secondary compounds. The results of the present study also showed that severe drought stress significantly reduced the essential oil content of medicinal plants. In non-application of mycorrhizal fertilizer, application of Rhizophagus intradices, application of Funneliformis mosseae, application of G. hoei and application of all three species of mycorrhizal fungi, with reduction of irrigation water from irrigation after 70 and 160mm evaporation from pan, the essential oil yield decreased by 75.6%, 76.9%, 47.7%, 66.3% and 72.5%, respectively.
In the present study, in irrigation after 70mm evaporation from pan, only application of Rhizophagus intradices significantly increased essential oil yield and increased this trait by 41.6%. In irrigation after 100mm evaporation from pan, two treatments of Funneliformis mosseae and application of all three species of mycorrhizal fungi increased essential oil yield by 100 and 146%, respectively. In irrigation treatments of irrigation after 130mm evaporation from pan and irrigation after 160mm evaporation from pan, application of mycorrhizal fertilizers had no significant effect on essential oil yield. The positive effect of mycorrhizal fungi on essential oil yield has been shown in various studies. Ghushchi Mahdieh et al. studied the effects of different strains of mycorrhizal fungi which showed that application of mycorrhizal fertilizers caused a significant increase in essential oil yield. In the present study, the percentage of colonization of lemon balm roots significantly affected the interaction of irrigation levels and mycorrhizal fungi application (Table 2). The highest percentage of colonization of lemon root with 78.6% was in irrigation after 70mm evaporation from pan with application of all three species of mycorrhizal fungi. In this study, drought stress alone did not have a significant effect on root colonization percentage in the absence of mycorrhizal fertilizer treatment, while drought with mycorrhizal fungi caused a significant decrease in the colonization rate of lemon balm root.
In treatments of application of Rhizophagus intradices, application of Funneliformis mosseae, application of G. hoei and application of all three species of mycorrhizal fungi, with reduction of irrigation water from irrigation after 70mm evaporation from pan to irrigation after 160mm evaporation from pan, root colonization percentage decreased by 23.8 12.8%, 21.8% and 30.2% (Table 5). Nasr et al. [18] showed that colonization of maize roots and Citrus reticulate roots by mycorrhizal fungi was reduced by dehydration. The researchers attributed the decrease in carbohydrate secretion to the soil, alteration of root structure, and hormonal changes in root surface area.
These results indicate that drought stress had negative impact on the percentage of root colonization. Considering the fact that in irrigation after 160mm evaporation from the pan, there was no significant difference between the studied strains and control in root colonization percentage, it can be concluded that the strains under study had the potential to colonize the lemon balm rootstocks at low levels. In this study, in the irrigation after 160mm evaporation from the pan, mycorrhizal application had the least incremental effect. In irrigation after 70mm evaporation from pan, irrigation after 100mm evaporation and irrigation after 130mm evaporation with application of three types of mycorrhizal fertilizer, The percentage of root colonization was increased by 129, 135.3 and 93.6%, respectively and these results showed that the effect of mycorrhizal fungi decreased with exacerbation of dehydration. Silva et al. [19] obtained a significant increase in the percentage of pepper mint colonization by using mycorrhizal fertilizer. In this study, the application of Escotelospora calospora caused the highest increase in this trait at low levels of fertilizer. Combined analysis of variance showed that the main effects of irrigation and mycorrhizal application on chlorophyll index traits, chlorophyll a and b were significant, but interaction of studied factors had no effect on chlorophyll content index of lemon balm (Table 1). Severe drought stress caused the highest decrease in chlorophyll index and chlorophyll content. Irrigation after 160mm evaporation decreased the chlorophyll index, chlorophyll a and b by 40.3%, 40.8% and 44.4%, respectively (Table 4). Similar results have been reported by other researchers.
Hassan et al. [20] investigated the effect of irrigation levels on chlorophyll content index of Rosmarinus officinalis leaves and reported that dehydration at 60% of field capacity reduced chlorophyll content of Rosmarinus officinalis by 23.5%. decreases. Studies have shown that among the stressors of water scarcity, the greatest decrease in chlorophyll content of crop leaves is due to production of active forms of oxygen in thylakoids [21]. According to the results of this study, mycorrhizal fertilizer treatments often caused a similar and significant increase in chlorophyll index and chlorophyll content. mycorrhizal fertilizer application treatments increased chlorophyll index, chlorophyll a and b by 20.4%, 24.4% and 23.1%, respectively (Table 3). Heidari et al. [22] examined the effect of different bacterial strains on Ociumum basilicm and observed a significant difference between bacterial strains in terms of chlorophyll index. The researchers found the highest increase in chlorophyll index using Pseudomonades sp. (2015) also observed the positive effect of G. intradices on the chlorophyll content of Ocimum gratissimum. Tamizinian et al. also showed that the use of mycorrhizal biofertilizer significantly increased the chlorophyll b content of their medicinal plant, Coleus aromaticus.
Proline content, catalase activity, glutathione peroxidase and ascorbate peroxidase were affected by the main effects of irrigation levels and fertilizer application in the present study (Table 2). The results of this study showed that irrigation treatment after 160mm evaporation from pan caused the highest increase in these traits and increased proline content, catalase activity, glutathione peroxidase and ascorbate peroxidase by 31.2, 42.6, 49% and 28.3%, respectively (Table 4). Proline is an amino acid compound that plays a role in the osmotic regulation of cells. Cellular regulation helps maintain the cell’s motility and makes cells more open for longer. Drought stress increases the amount of this compound (Mann et al., 2011). ABA is one of the hormones that strongly affect proline production. But studies have shown that environmental factors such as the bacteria around the roots of plants affect the amount of proline in plants, and especially in conditions of dehydration it increases the amount of proline (Stathavi and Tavik, 2007). An increase in the proline content of medicinal plants has also been reported in studies by other researchers. Shafiqi & Pazuki (2014) in their study of the medicinal plant Trigonellafoenum-graceum found that dehydration caused a 98% increase in leaf proline content. In current study, the activity of other antioxidants also increased under the influence of dehydration. In this study, in most traits of proline content, catalase activity, glutathione peroxidase and ascorbate peroxidase, and all mycorrhizal species application increased significantly and increased these traits by 26.3, 41.7, 53.9 and 43.4% (Table 3). Naeemi et al. [23] investigated the effect of mycorrhizal application on Silybum marianum and observed that application of G. mossae and G. intradices significantly increased the content of catalase and glutathione peroxidase in this plant. The researchers attributed the decline to improved plant water uptake [24-35].
Conclusion
According to the results, although irrigation treatment had a positive effect on essential oil yield after 100mm evaporation from pan [36-40], but in total drought stress levels caused a decrease in general yield and lemon balm yield and, with intensification of dehydration, decreased severity. So that drought stress reduced up to 84% of the yield of lemon balm. However, the results showed that the application of mycorrhizal fungi, and particularly the combined application of the species, could somewhat reduce the negative effect of drought stress on the studied traits. The highest increase was in irrigation treatment after 100mm evaporation from the pan [41-42].
Refernces
- Ribeiro MA, Bernardo Gil MG, Esquivel MM (2001) Melissa officinalis L: Study of antioxidant activity in supercritical residues. Journal of Supercritical Fluids 21(1): 51-60.
- Lakzayi M, Sabbagh E, Rigi K, Keshtehgar A (2014) Effect of salicylic acid on activities of antioxidant enzymes, flowering and fruit yield and the role on reduce of drought stress. International Journal of Farming and Applied Sciences 3: 980-987.
- Sinha RK, Valani D, Chauhan K, Agarwal S (2014) Embarking on a second green revolution for sustainable agriculture by vermiculture biotechnology using earthworms: reviving the dreams of Sir Charles Darwin. Int J Agric Health Saf 2(7): 50-64.
- Adesemoye AO, Kloepper JW (2009) Plant-microbes interactions in enhanced fertilizer-use efficiency. Appl Microbiol Biotechnol 85(1): 1-12.
- Lu F, Lee C, Wang C (2015) The influence of arbuscularmycorrhizal fungi inoculation on yam (Dioscorea spp.) tuber weights and secondary metabolite content. Peer J 3: e1266.
- Aram H, Jorenoosh MH (2018) Application of arbuscular mycorrhizal fungi on zinc and iron concentration in wheat under cadmium Stress. JCHR 8(3): 239-243.
- Alizadeh Oskuie P, Baghban Cirus S (2015) The effect of Vesicular-arbuscular (VA) mycorrhizal fungi on vitamin c content of tomato in the presence of lead and different levels of phosphorus. Bulletin of Environment, Pharmacology and Life Sciences 4: 1-4.
- Gholizadeh A, Amin MSM, Anuar AR, Esfahani M, Saberioon MM (2010) The study on the effect of different levels of zeolit and water stress on growth, development and essential oil content of moldavian balm (Dracocephalum moldavica L.). American Journal of Applied Sciences 7(1): 33-37.
- Abbasi M, Pouzesh H, Enayati A, Hedayati A (2012) Investigation the effect of hydropriming and osmopriming treatments on seeds germination of Tall Wheatgrass (Agropyron elongatum) under drought stress. Annals of Biological Research 3(10): 4874-4879.
- Shinozaki K, Yamaguchi Shinozaki K (2007) Gene networks involved in drought stress response and tolerance. J of Experimental Botany 58(2): 221-227.
- Jamali MM (2013) Investigate the effect of drought stress and different amount of chemical fertilizers on some physiological characteristics of coriander (Coriandrumsativum L.) International Journal of Farming and Allied Sciences.) 2(20): 872-879.
- Ghanbari M, Ariafar S (2013) The effect of water deficit and zeolite application on growth traits and oil yield of medicinal pepermint (Menta piperita L). International Journal of Aromatic plants 3: 32-39.
- Srinivasa Murthy KM, Narayanappa M (2013) Influence of Vesicular Arbuscular Mycorrhizae (VAM) on Growth of Rutagraveolens - A Medicinal Plant. International Journal of Science and Research 4(5): 438.
- Arango MC, Ruscitti MF, Ronco MG, Beltrano F (2012) mycorrhizal fungi inoculation and phosphorus fertilizer on growth, essential oil production and nutrient uptake in peppermint (Menthapiperita L.). Rev Bras Pl Med Botucatu 14(4): 692-699.
- Habibzadeh Y (2015) Effects of phosphorus levels on dry matter production and root traits of chickpea plants in presence or absence of Arbuscular mycorrhizal fungi. Journal of Agricultural Science and Food Technology 1(1): 1-6.
- Kumar A, Mangla C, Aggarwal A, Srivastava V (2014) Rhizospheric effect of endophytic mycorrhiza and trichodermaviride on physiological parameters of Mentha Spicatalinn. Asian J of Adv Basic Sci 2(1): 99-104.
- Andalibi B, Zehtab SS, Ghassemi Gholezani K, Saba J (2010) Changes in essential oil yield and composition at different parts of dill (Anethumgraveolens L.) under limited irrigation conditions. Journal of Agricultural Science, University of Tabriz, Iran 21(2): 11-22.
- Nasr AH, Zare M, Alizadeh O, Mehdizadeh Naderi N (2013) Improving effects of mycorrhizal symbiosis on sorghum bicolor under four levels of drought stress. African Journal of Agricultural Research 8(43): 5347-5353.
- Silva FA, Silva FSB, Maia LC (2014) Biotechnical application of arbuscular mycorrhizal fungi used in the production of foliar biomolecules in ironwood seedlings [Libidibiaferrea (Mart. ex Tul.)L.P. Queiroz var. ferrea].Journal of Medicinal Plant Research 8(20): 814-819.
- Hassan FAS, Bazaid S, Ali EF (2013) Effect of deficit irrigation on growth, yield and volatile oil contenton Rosmarinus officinalis L Journal of Medicinal Plants Studies 1(3): 12-21.
- Kiani SP, Maury P, Sarrafi A, Grieu P (2008) QTL analysis of chloropHyll fluorescence parameters in sunflower (Helianthus annuus L.) under well-watered and water-stressed conditions. Plant Sci 175: 565-573.
- Heidari M, Mohsen Mousavinik S, Golpayegani A (2011) Plant growth promoting rhizobacteria (pgpr) effect on hysiological parameters and mineral uptake in basil (Ociumumbasilicm L.) under water stress. ARPN Journal of Agricultural and Biological Science 6: 78-91.
- Naeemi N, Ladanmogaddam AR, Moradi P, Rezaei M, Abdoosi V (2017) Effect of vermicompost and mycorrhizae fungi on yield and growth of milk thistle and antioxidaant system activity. Iranian Journal of Plant Physiology 7(3): 2063-2074.
- Alaei S, Melikyan A, Kobraee S, Mahna N (2013) Effect of different soil moisture levels on morphological and physiological characteristics of Dracocephalum moldavica. Agricultural Communications 1(1): 23-26.
- Banchio E, Xie X, Zhang H, Pare PW (2009) Soil bacteria elevate essential oil accumulationand emissions in sweet basil. Journal of Agricultural and Food Chemistry 57(2): 653-657.
- Hazzoumi Z, Moustakime Y, Elharchli E, AmraniJoutei K (2015) Effect of arbuscular mycorrhizal fungi (AMF) and water stress on growth, phenolic compounds, glandular hairs, and yield of essential oil in basil (Ocimumgratissimum L). Hazzoumi et al. Chemical and Biological Technologies in Agriculture 2: 10.
- Jafari Ghoushchi M, Abbaszadeh B, Oraei M (2015) Effects of chemical and biological fertilizers on growth, yield and essential oil of Salvia officinalis. Journal of Medicinal Plants and By-Products 4(1): 31-37.
- Karaarslan E, Uyanöz R, Doğu S (2015) Morphological identification of vesicular-arbuscular mycorrhiza on bulbous plants (taurus mountain in turkey). Arch Biol Sci Belgrade 67(2): 411-426.
- Kazemi Nasab A, Yarnia M, Lebaschy MH, Mirshekari B, Rejali (2015) The Response of drought stressed lemon balm (Melissa officinalis L.) to Vermicompost and PGPR. Biological Forum. An International Journal 7(1): 1336-1344.
- Khalil SE, Ali El-Noemani A (2015) Effect of bio-fertilizers on growth, yield, water relations, pH otosynthetic pigments and carbohydrates contents of origanum vulgare L. plants grown under water stress conditions. American Eurasian Journal of Sustainable Agricultur 9(4): 60-73.
- Kheyrodin H (2014) Plant and soil relationship between Fungi. International Journal of Research Studies in Biosciences (IJRSB) 2(9): 42-49.
- Maazou AS, Tu J, Qiu J, Liu Z (2016) Breeding for drought tolerance in maize (Zea mays L.). American Journal of Plant Sciences 7(14): 1858-1870.
- Man D, Bao Y, Han L (2011) Drought tolerance associated with proline and hormone metabolism in two tall fescue cultivars. Hortscience 46(7):1027-1032.
- Roshanpour N, Taghi Darzi M, Haj Seyed Hadi M (2014) Effects of plant growth promoter bacteria on biomass and yield of basil (Ocimumbasilicum L.). International journal of Advanced Biological and Biomedical Research 2(6): 2077-2085.
- Roudi M, Salamatmanesh MM (2018) Study on the growth of (Achilleamillefolium L.) medicinal plant by soil inoculation of mountainous area with selected mycorrhizal fungi. Iranian Journal of Plant Physiology 9(1): 2601-2609.
- Sarkar J, Ray A, Chakraborty B, Chakraborty U (2016) Antioxidative changes in Citrus reticulata L. induced by drought stress and its effect on root colonization by arbuscular mycorrhizal fungi. European Journal of Biological Research 6 (1): 1-13.
- Shafighi A, Pazoki A (2014) Effects of drought stress and stimulus growth bacteria on some physiological traits of medicinal plants of fenugreek (Trigonellafoenum-graceum L). Trends in Life Science 3: 215-224.
- Shao HB, Chu LY, Shao MA, Abdul Jaleel C, Hong Mei M (2008) Higher plant antioxidants and redox signaling under environmental stresses. Comp Rend Biol 331(6): 433-441.
- Sheteawi SA, Tawfik KM (2007) Interaction effect of some biofertilizers and irrigation water regime on mung bean (Vigna radiata) growth and yield. Journal of Applied Sciences Research 3(3): 251-262.
- Yadav A, Aggarwal A (2015) The associative effect of arbuscular mycorrhizae with Trichoderma viride and Pseudomonas fluorescens in promoting growth, nutrient uptake and yield of Arachishypogaea L. New York Science Journal 8(1): 101-108.
- Zolfaghari M, Nazeri V, Sefidkon F, Rejali F (2013) Effects Effect of arbuscular mycorrhizal fungi on plant growth and essential oil content and composition of Ocimumbasilicum L. Iranian Journal of Plant Physiology 3(2): 643-650.
- Thamizhiniyan P, Panneerselvam M, Lenin M (2009) Studies on the growth and biochemical activity of coleus aromaticusbenth as influenced by am fungi and azospirillum. Recent Research in Science and Technology 1(6): 259-263.
© 2021 © Yarnia M. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






