- Submissions

Full Text
Environmental Analysis & Ecology Studies
Development of a Mexican Caramel Jam: A Food Industry Alternative for Cheese Whey Valorization
Carmen Bulbarela Sampieri1, Lorena De Medina Salas1, Jorge Octavio Virues Delgadillo1, Eduardo Castillo González2, Raymundo Hernández Mateos1 and Micloth López-del-Castillo3*
1Facultad de Ciencias Químicas, México
2Facultad de Ingeniería Civil, México
3Instituto de Ciencias Básicas, México
*Corresponding author: Micloth López-del-Castillo, Instituto de Ciencias Básicas, México
Submission: June 28, 2019;Published: August 12, 2019

ISSN 2578-0336 Volume6 Issue1
Abstract
Development of a Mexican caramel jam, which can be consumed directly or used in confectionery and bakery, was made on the present research as an alternative for cheese whey valorization. Raw materials (cheese whey and cow milk) were obtained from different cheese producers in the central region of the state of Veracruz, Mexico. Samples were pasteurized at 65 °C, 30min and kept at 8 °C until they were used. To prepare the caramel jam all the compounds were obtained as food-grade from a local supplier. The Mexican caramel jam products were made by different ratios of cheese whey and milk. Physicochemical analyzes were performed on all samples. Besides, color, viscosity, and sensorial evaluation were also analyzed. Protein, fat, and lactose percentages were 3.2±0.98, 0.54±0.21, 5.2±1.42, respectively; pH had values above 7.1. In sensory evaluation, almost all the panelists attributed acceptance values between 6 and 9 for all formulations.
Keywords: Cheese whey; Caramel jam; Valorization; Cajeta
Introduction
Hernández & Velez [1] and Valencia & Ramírez [2], pointed out that industrial and artisanal cheese manufacturing produce a large flow of cheese whey as a byproduct. In general, to produce 1 or 2 kilos of cheese, it is necessary to have about 10 liters of milk, this generate between 8 to 9 liters of cheese whey. Božanić et al. [3], considered that 80-90% of the total flow of milk processed is discarded as cheese whey. Although cheese whey composition depends on the origin of the milk and the variety of cheese produced, in general it contains 93.1% water, 4.9% lactose, 0.9% protein, 0.6% ash (mainly phosphorus, calcium, sodium and potassium), 0.3% of fat, 0.2% of lactic acid and water-soluble vitamins. Hernández & Vélez [1] and Burguete & Caloca [4], showed that cheese whey is still discarded as an untreated byproduct in nearby bodies of water or in the sewer system. Rojas et al. [5] considered this situation causes a severe environmental impact due to the high concentrations of Chemical Oxygen Demand (COD) and the Biochemical Oxygen Demand (BOD) (35,000 to 68,000mg/L) which makes it highly polluting.
Santamaría et al. [6], indicated that the discarded cheese whey not treated generate an impact on the receptor bodies, due to the low pH value and high temperatures; the water becomes turbid and greenish and aerobic bacteria appear to consume the oxygen of the water oxidizing the organic matter; Prazeres et al. [7], explained that decreases the amount of dissolved oxygen in water and causes the death of microorganisms and destabilizes biological processes in rivers and even in municipal wastewater treatment plants. In addition, Carvalho et al. [8], considered that due to the total contents of nitrogen and phosphorus, cheese whey effluents have a considerable risk of eutrophication of groundwater, slow flow rivers and lakes. Smithers [9] and Palmieri et al. [10], explained that developed countries such as United States, Canada, Australia, New Zealand, and the European Union have established strict environment legislation against improper discharges of whey and in favor of their recycling and reuse.
Actually, thanks to its high nutritional value, cheese whey can be recycled or reused within the food and dairy chains into alternative products for human consumption or for feeding animals.
Božanić et al. [3], showed the in the protein fraction of whey for instance, 70% corresponds to β-lacto-globulin, α-lacto-albumin, immune globulins and enzymes, which together have a higher nutritional value than that of caseins. González [11] reported that cheese whey can be treated and reused in liquid form or in powder, as lactose or lactose free byproducts, and as cheese-whey-protein concentrates. Remón et al. [12] pointed out to that to achieve this, techniques such as nanofiltration or ultrafiltration must be applied. However, their disadvantage is the high cost; thus, it is not feasible to implement them on a small scale, as in the case of artisanal cheese manufacturing Sinia [13].
Królczyk et al. [14] and Motta & Mosquera [15], proposed other reuses of whey cheese include meat products, reduced-fat products, yoghurts and ice creams, cheeses, bakery products, confectionery and pastry products, infant formulas, and whey drinks. Henriques et al. [16], suggested, the improvement of yogurts and fresh cheese characteristics; meanwhile Pereira et al. [17] indicated about fermented and probiotics products as kefir; Rojas et al. [5], in the production of acid lactic; Gauche et al. [18], for lactulose; Yadav et al. [19] and Martins et al. [20], for bioactive peptides.
Therefore, the aim of this study was to reuse the cheese whey as a food industry alternative for valorization, by developing a Mexican caramel jam (“cajeta”), that can be consumed directly or used in confectionery and bakery. This “cajeta” is made generally with goat’s milk added with glucose and/or sucrose, evaporated and concentrated, reducing the volume of milk by more than 90%; Ramirez & Vélez [21], pointed out that “cajeta” is consumed in Mexico to spread on bread, as sweet with wafers, confectionery, as an additive for ice cream, filling of cakes and jelly to sweeten and flavor pancakes, hotcakes, dairy drinks and yogurt. Moro & Hough [22] and Ares et al. [23], mentioned that this type of caramels is also made in countries such as Argentina and Uruguay, where it is known as “dulce de leche”.
Methods and Materials
Materials
Cheese whey and cow milk were obtained from different cheese producers in the central region of the state of Veracruz (Acatlán, Naolinco, Jilotepec, Coatepec and Xico), in Mexico. This region corresponds to a mesophilic mountain forest, and milk was obtained as a mixture of different races according to the breeder. The samples were pasteurized at 65 °C, 30min and kept at 8 °C until they were used. To prepare the caramel jam all the compounds (sucrose, liquid corn glucose and sodium bicarbonate) were obtained as food-grade from a local supplier.
Making of Mexican caramel jam
Mexican caramel jam samples were made in triplicate using different ratios of cheese whey (0, 50, 75, 100%) and milk (0, 25, 50, 100%). The samples were labeled according with their whey content; the samples made with 25% of cheese whey (W) and 75% of cow milk (C) were then labeled as C25W, while the products labeled C100W, C75W, C50W and C0W, were obtained by using 100, 75, 50 and 0% of cheese whey. Each samples (C0W, C25W, C50W, C75W, and C100W) were stirred thoroughly at room temperature and 0.4% NaHCO3 was added. After that, the samples were heated to 60 °C, and then sucrose (15%w/v initial) and glucose (7.5%w/v initial) were added. This mixture was heated to evaporation with constant blended. As the sample reduced its initial volume to 1/3, sucrose (15%w/v initial) and glucose (7.5%w/v initial) were added again to reach 65-82 °Brix. The final product was packed and stored in sterilized glass vials at room temperature until analysis.
Physicochemical analyses
Cheese whey samples were analyzed physico-chemically. Indeed, pH was measured a room temperature by digital potentiometer (Beckman, Denver, CO, USA) using 1g of caramel jam in 10mL of water solution. Acidity content was quantified in 10mL sample solution by titration with NaOH (0.1N) and reported as percentage of lactic acid. Soluble solids were determined with a manual refractometer (ATAGO PR-1, Tokyo, Japan) at 20 °C and the results were expressed as °Brix. Moisture, total solids, nonfat solids, and residual ash content were analyzed according to Mexican Norms [24,25]. Proteins, lactose, and fat percentages were analyzed by spectroscopic ultrasonic (Lacti-Check Ultrasonic Analyzer model LC-02). Proteins were also quantified by Kjeldahl method. Fat content was also measured using the Warner-Schmid gravimetric method, which is based on acidic hydrolysis of fats and followed by ethylic ether extraction.
Color
Color was measured with a manual spectrophotometer (Konica Minolta 8D80) and the parameters L*, a* and b* were registered. In this case, L* stand for luminosity and has values between 0 (dark) and 100 (bright). The parameters a* and b* denote the color of samples from red (+) to green (-), and yellow (+) to blue (-) respectively. The value of ΔE was also calculated using Equation 1 where the color parameters of C0W samples were took as reference.
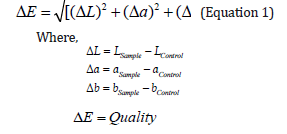
0 - 1 = Excellent
1 - 2 = Good
2 - 4 = Mean
4 - 5 = Enough
> 5 = Bad
Sensorial evaluation
A hedonic test was performed with 45 untrained judges, 27 women and 19 men, aged from 19 to 65 years old. Each judge evaluated the flavor, texture, odor, color and general acceptance parameters. The hedonic scale had nine points ranged from 1 (extremely disliked) to 9 (extremely liked) with a central point in 5 (either liked or disliked).
Viscosity testing
Viscosity test was performed with a Brookfield Viscometer (Model 4535, Lab-Line Instruments). Glass vessels were prepared with enough sample to use spindle number 5 (diameter 2.1cm) and number 6 (diameter 1.5cm). Shear rate was set to 1.05rad/s (10rpm) with a testing time of 300 seconds to achieve stationary state at a constant temperature of 40 °C. Samples apparent viscosity was registered in centipoises (cP) [26].
Result and Discussion
Physicochemical analyses
Physicochemical composition of the cheese. As food constituents, proteins, fats and carbohydrates give nutritional value to food products. Although classified as a waste effluent, cheese whey carries on several proportions of these nutrients. By Mexican law, percentage of such nutrients are higher than 0.72% for protein content, lesser than 0.10% for fat content, and 4.7% for lactose. On the cheese whey samples characterized in the present work, it was observed the following percentage of each constituent: 3.2±0.98 % protein, 0.54±0.21% fat, 5.2±1.42% lactose.
Physicochemical characterization of caramel jam. There were no significant statistical differences between products for the parameters of pH, total solids and moisture. However, protein, fat, ash and °Brix parameters shown a significant statistical differences between products. The highest values in proteins (3.5%) and fat (4.0%) were presented by C0W and the lowest values (2.4% and 0.7% respectively) were obtained in C100W. Differences observed in protein and fat values were probably due to concentrations of cheese whey. Neither the fat nor the protein was adjusted since it would increase again the possible making cost of products.
Color
Color of caramel jams were analyzed. It is illustrated that a 50% replacement of cheese whey was closer to C0W product which was made with 100% of milk. However, as the amount of cheese whey was increased in the products, and then the color difference (i.e., ΔE) was increased too. Parameter L* indicates the color luminosity and ranged from 0 (dark) to 100 (bright). The a* values represent the redness color intensity. The b* values indicate the yellowness. The product´s brownish color was probably due to a non-enzymatic browning reaction. Van Boekel [27], referred Maillard reactions could be occurred between the proteins and the carbohydrates, lactose form the cheese whey or milk, and sucrose and glucose added during the caramel jam making.
Food products substituted with cheese whey tend to be lest bright, as it can be observed in the L* values. On the other hand, parameter a* indicates the tendency to red (+) or green (-) color. In this work, it was observed that C0W sample presents a tendency towards red color, and as soon as the cheese whey percentage was increased, this value decreased, which was observed with a less intense color (i.e. sample C100W developed the less intense red color, with a value of 0.354). The effect of diminishing coloration due to the increase in the concentration of serum in the product can be attributed to the caseins, which are precipitated in the curd during the cheese making, and are the majority proteins responsible for the formation of reactions of Maillard compounds, which give brownish-red tones to the food products. Parameter b* indicates the tendency of the products towards yellow (+) or blue (-) color. In our case, values of b* indicated that the caramel jam products had a tendency to yellow color. The values show that all the products were significantly different; in particular, it was observed that caramel jam products tend more towards the yellow tones than the caramel jam products elaborated in this work.
Values of ΔE lesser than one indicated that the sample (product made with cheese serum) was equal to the reference product, while maintaining an excellent quality. Thus, the product made with 50% cheese whey was equal to the reference product (in this case, the caramel jam made with 0% cheese whey was used as reference), however, this product was not compared with the commercial caramel jam because it had several differences in terms of the manufacturing process and additives added.
Sensorial evaluation
In a general speaking, the judges gave a good score to all caramel jam formulations, scored it with values between 6 and 9. The C0W product obtained the highest score of 7.2 in the general acceptance parameter, equivalent to ‘liked moderately’. This score decreased slightly as the proportion of cheese whey increased in the product. Indeed, the C100W product (100% of cheese whey) obtained the lowest score of 5.87 equivalents to ‘neither like nor dislike’. The panelists were only able to observe the color difference between the product made exclusively with milk (C0W) and the one made entirely with cheese whey, C100W (ΔE = 4.19).
The alternative of reuse and recycling of cheese whey in the food industry, presents an important associated environmental benefit, since with this valorization it avoids its inadequate disposal that can cause a significant environmental impact in the water bodies, as in the case of dissolved oxygen depletion and acidification, which in turn would lead to the extinction of higher species of aquatic life, turbidity that causes inhibition of photosynthetic processes, generation of bad odors and deterioration of the landscape.
Viscosity testing
Apparent viscosity (in centipoises, cP) of samples made with 0% and 50% cheese whey, were the effect of testing time on the viscosity of both samples of the caramel jam product analyzed isobserved for the two spindles used in this work. The results show the viscosity of the caramel jam product made from milk (C0W) is almost three times more viscous than the product made with 50% cheese whey (C50W); therefore, the amount of solids present in this type of products is a variable which substantially modifies the viscosity
Conclusion
Physicochemical results of cheese whey samples from the study area, show that they still have considerable percentages of proteins, lactose, fat and mineral salts, which makes this by-product of the dairy industry feasible for its reuse in the elaboration of new products, of for its use in food fortification. Mexican artisanal dairy products are a good combination for their reuse taking advantage of the nutritional properties of cheese whey, with a partial replacement of the milk for processing up to 50%, without affecting the sensory properties of the caramel jam product. The viscosity properties are affected by a substitution of 50% of milk with cheese whey.
In this work we attempted to show the reuse of cheese whey in a foodstuff as the caramel jam, and in this way reduce its environmental impact
References
- Hernández RN, Vélez RJF (2014) Suero de leche y su aplicación en la elaboración de alimentos funcionales. Temas selectos de Ingeniería de Alimentos 8(2): 13-22.
- Valencia, Ramírez (2009) La industria de la leche y la contaminación del agua. Elementos 73: 27-31.
- Božanić R, Barukčić I, Lisak K, Jakopović, Tratnik L (2014) Possibilities of whey utilization. Austin J Nut Food Sci 2(7): 1036.
- Burguete AR, Zea Caloca SG (2016) Gelatina a base de suero de quesería y extracto de soya. Inv Des Cienc Tecnol Alim 1(1): 619-625.
- Rojas (2015) Production of lactic acid from whey using Lactobacillus delbrueckii subsp. bulgaricus and Streptococcus thermophilus. Rev Colomb Quím 4(3): 5-10.
- Santamaría (2015) Characterization of water quality parameters to reduce pollution during the processing of milk. Agroind Sci 5: 13-26.
- Prazeres (2012) Cheese whey management: A Review. J Environ Manage 110: 48-68
- Carvalho F, Prazeres AR, Rivas J (2013) Cheese whey wastewater: Characterization and treatment. Sci Total Environ 445-446: 385-396.
- Smithers (2008) Whey and whey proteins- from “gutter-to-gold”. Int Dairy J 18(7): 695-704
- Palmieri, Maria BF, Elisabetta S (2017) Environmental impacts of dairy cheese including whey feeding: An Italian case study. J Clean Prod 140: 881-889.
- González MI (1996) The biotechnological utilization of cheese whey: A review. Biores Technol 57(1): 1-11.
- Remón (2016) Cheese whey management by catalytic steam reforming and aqueous phase reforming. Fuel Proc Technol 154: 66-81.
- SINIA (2017) Sistema Nacional de Información Ambiental.
- Królczyk B, Tomasz D, Emilia J, Sołowiej B (2016) Use of whey and whey preparations in the food industry - A Review. Pol J Food Nutr Sci 66(3): 157-165.
- Motta, Mosquera (2015) Aprovechamiento del lactosuero y sus componentes como materia prima en la industria de alimentos. Rev Alimentech Cienc Tecnol Alim 13: 81-91.
- Henriques MHF, Gomes DMGS, Pereira CJD, Gil MHM (2012) Effects of liquid whey protein concentrate on functional and sensorial properties of set yogurts and fresh cheese. Food Biop Technol 6(4): 952-963.
- Pereira (2015) Fermented dairy products based on ovine cheese whey. J Food Sci Technol 52: 7401-7408.
- Gauche C, Tomazi T, Barreto PLM, Ogliari PJ, Bordignon LMT (2009) Physical properties of yoghurt manufactured with milk whey and transglutaminase. LWT-Food Sci Technol 42(1): 239-243.
- Yadav, Yan S, Pilli S, Kumar L, Tyagi RD, et al. (2015) Cheese whey: A potential resource to transform into bioprotein, functional/nutritional proteins and bioactive peptides. Biotechnol Adv 33(6 Pt 1): 756-774.
- Martins IFS, Jongen MF, Svan Boekel AJ (2001) A Review of maillard reaction in food and implications to kinetic modeling. Trends in Food Science & Technology 11(9-10): 364-373.
- Ramírez, Vélez (2013) Physicochemical, rheological and stability characterization of a caramel flavored yogurt. LWT - Food Sci Technol 51: 233-241
- Moro, Hough (1985) Total solids and density measurements of dulce de leche, a typical Argentine dairy product. J Dairy Sci 68(3): 521–525.
- Ares G, Giménez A, Gámbaro A (2006) Preference mapping of texture of dulce de leche. J Sensory Studies 21(6): 553-571
- DGN (1986) NMX-F-083-1986. Alimentos. Determinación de humedad en productos alimenticios. (Foods. Moisture quantification in food products). Normas Mexicanas. Dirección General de Normas.
- DGN (1978) NMX-F-066-S-1978. Determinación de cenizas en alimentos. (Foods. Quantification of ashes). Normas Mexicanas. Dirección General de Normas.
- DGN (2014) NMX-F-721-COFOCALEC-2012. Suero de leche (líquido o en polvo)-Especificaciones y métodos de prueba. Consejo para el fomento de la calidad de la leche y sus derivados, A.C.
- Van Boekel (1998) Effect of heating on Maillard reactions in milk. Food Chem 62(4): 403-414.
© 2019 Micloth López-del-Castillo. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






