- Submissions

Full Text
Environmental Analysis & Ecology Studies
A Study of Constructed Wetland on Overflow Land Type Upgrading Campus Sewage Effluent
TY Yeh1* and CS Yuan2
1 Department of Civil and Environmental Engineering, Taiwan
2 Institute of Environmental Engineering, Taiwan
*Corresponding author: TY Yeh, Department of Civil and Environmental Engineering, Taiwan
Submission: February 08, 2019;Published: February 26, 2019

ISSN 2578-0336 Volume5 Issue2
Abstract
The merits of constructed wetlands are ecosystem protection, water quality enhancement, and wastewater treatment. Campus wastewater treatment effluent was studied in this research. Nutrients and organic matters have demonstrated effectively via sedmentation, microbio-degradation, and plant uptake. SS, COD, DO, NH4+, NO3-N were also researched. Total Nitrogen, total Phosphorus, Ammonium Nitrogen removal rate was 84.1%, 47.9%, and 84.6%, respectively. The microbial genetic monitoring results showed Salmonella spp. and Listeria monocytogenes were not existed in soil and sediment. Acrophytes harvesting and sediments dredging are the promising approaches to remove nutrients and organic matters in this studied constructed wetland [1].
Keywords: Constructed wetland; Organic matters; Nutrients; Sediment; Microbial identification; PCR
Introduction
Constructed wetland is a man-made facility to mimic natural ecosystems treating wastewater. The removal mechanisms include filtration, adsorption, precipitation, microbial degradation, and microbial adsorption. Constructed wetland also can provide ecosystem and landscape enhancement [2]. In Taiwan, constructed wetland has been employed to treat sewage, piggery, and aquaculture wastewater. Campus waster often fluctuates up and down and hard to manage. By virtue of constructed wetland, used wastewater can be recycled and sustainable water resources can be achieved [3].
Materials and Method
Study site
Table 1:Wetland overland flow system overall removal efficiency.
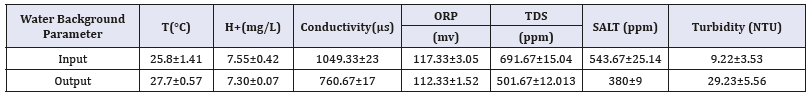
The constructed wetlands are situated in the University of Kaohsiung campus (22° 73’N, 120° 28’E). The length, width, average depth and flow velocity were 585m,5.2m, 0.5m and 67.5m/ hr (1.88cm/s) respectively, while the detention pond at point has a depth of 1.2m Table 1.
Water quality analysis
Water parameters, including organic matter (BOD and COD), Particulate matter (SS), Nutrients (Nitrogen and Phosphorus), Chlorophyll A, and Pathogenic Indicators (E. coli) were investigated [4].
Microbial PCR analysis
Deoxyribonucleic Acid (DNA) extraction and polymerase chain reaction-denaturing gradient gel electrophoresis (PCRDGGE) analysis were analyzed according to Boon et al. (2001). The Wizard DNA Clean-Up Kit (Promega, Madison, Wisconsin) was used to detect microbial community dynamics in the process of nitrogen transformation in the 1g sediment samples from surface-flow wetlands. Bacterial 200-bp fragments of 16S rDNA V3 region for subsequent denaturing gradient gel electrophoresis (DGGE) analysis were obtained with the primer combination of 341f with a gas chromatography clamp (40-nucleotide GC-rich sequence, 5-CCTACGGGAGGCAGCA G-3) and 534r (5-ATTACCGCGGCTGCTGG-3) [5].
The PCR mixtures contained 10ng of DNA extract, 4pmol of each primer, and 5U of Taq polymerase (Takara, Shiga, Japan) in the final concentration of 2.5mM of Mg C12 and 0.12mM of deoxyribonucleoside triphosphates in PCR buffer. The PCR amplification was conducted for 35cycles: denaturation at 94 °C for 1min, annealing at 55 °C for 1min, and extension at 72 °C for 2min. The equal concentration of each amplified PCR product (2500ng) was further performed with DGGE using a Bio-Rad Decode system (Bio-Rad, Hercules, California), as described by the manufacturer [6]. The 10% Polyacrylamide gel with a 30 to 60% denaturant gradient was used, and electrophoresis was performed at 60 °C and 70V for 14h Table 2.
Table 2:Wetland overland flow system overall removal efficiency.
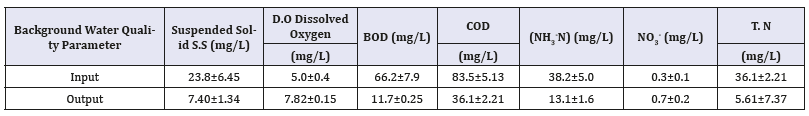
The gels were then stained with SybrGreenl and photographed. The relative intensity of amplified bands in the gels was analyzed with Phoreti ID software (Nonlinear Dynamics, Newcastle upon Tyne, United Kingdom). The PCR-amplified products were electroeluted from gel and then sequenced by provider (MdBio, Taipei, Taiwan) [7].
Those sequences were evaluated using the basic local alignment search tool (BLAST) to determine the closest relatives in the GenBank databases (www.ncbi.nlm.nih.gov). Alignment of nucleotide sequences of PCR-amplified products generated a matrix of similarity coefficients with the neighbor-joining method (Saitouand Nei, 1987). The dendrogram based on the similarity coefficient was plotted with un-weighted pair-group method with Arithmetic mean (UPGMA) method for clustering (Felsenstein, 1993).
Data and statistical analysis
Figure 1:overland flow system and aerial schematic side.
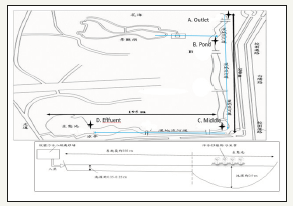
Data were evaluated relative to the control to understand their statistical variations. A triplicate of water and sediment samples were measured and recorded for statistical analyses. Statistical significance was assessed using a mean comparison test. Differences between treatment concentration means of parameters were determined by Student’s t test. One-way ANOVA was also employed to show the variation among sample groups, level of p< 0.05 considered statistically significant was used in all comparisons. Means are reported mean ± standard deviation. All statistical analyses were performed with Microsoft Office EXCEL 2007 Figure 1.
Results and Discussion
Figure 2:Soil and sediment microbio analysis, Salmonella.

Water quality, soil and sediments PCR were also monitored [8,9]. The length of the water channel is 585m while velocity is 67.5m/hr (0.0188m/s), River water depth of about 0.15-0.2m. The W*D = 5.12m *0.9m. The sampling time is PM13:00, the duration is 3 months stared from March 5, 2016. The water quality parameters are reported in Table 3. Door coli (Salmonella spp.) Gram-negative bacilli, spore, Tony flagellated good at sports, under aerobic or facultative anaerobic, thermal weak acidic environment (PH< 4.5) their development will be inhibited. Listeria (scientific name: Listeria monocytogenes), and a list of monocytes Listeria, Listeria is a facultative anaerobic bacteria, the disease pathogen Listeria. It is mainly food-vector, is one of the deadliest food-borne pathogens, resulting in two to three percent of those infected. In which Listeria’s disease is a major cause of death, the death rate is even higher than salmonella and botulism. Listeria genus of grampositive bacteria, environmental adaptability, widely distributed in nature, often found in soil, plants and saprophytic many mammals’ feces, which results in Figure 2 & 3.
pH
The pH data are in Figure 4. The influent is 7.29, midpoint is 7.7 while effluent is 7.7. The decrease of pH indicates precipitation and nitrification occurred [10].
Figure 3:Soil and sediment microbio analysis, Salmonella.
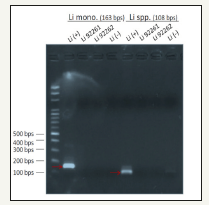
Figure 4:pH changes in Water.
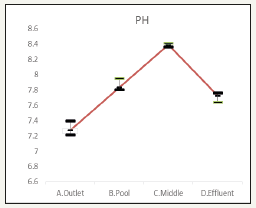
Table 3:Wetland overland flow system overall removal efficiency

DO
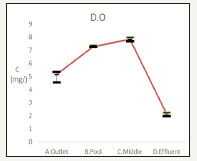
DO plot is in Figure 5. The influent is 5.0±0.4mg/L, the efflurnt is 7.3±0.07mg/L while the most eutrophic point recreation pond is 2.11±0.13mg/L. The DO results indicate eutrophication will reduce dissolved oxygen [11].
Figure 5:Changes of dissolved oxygen water quality.
SS
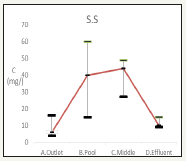
Suspended solid results are recorded in Figure 6. The influent is 9±6mg, the effluent is 58.3±54.8mg/L while the recreation pond is 11±3.4mg/L. Leaves, grit, and algae accumulation induced the sudden increase of suspended solid [12].
Figure 6:Changes of suspended solids in water quality.
Turbidity
The turbidity results are in Figure 7. The influent is 9.22±3.54(NTU), the effluent is 11.7±2.86(NTU) while the recreation pond is 29.2±5.58(NTU). Algae growth is strongly corrected with the turbidity Figure 8.
Figure 7:Change the water turbidity.
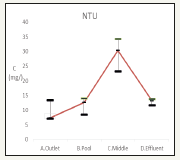
Figure 8:Wetland water through the filter case

Ammonium nitrogen
The Ammonia Nitrogen is recorded in Figure 9. The influent is 38.2±5.0mg/L, the effluent is 28.3±2.59mg/L while the recreation pond is 28.3±2.59mg/L. The total Ammonium Nitrogen removal rate is 84.6%. External feeding caused in the jump of Ammonium Nitrogen concentration [13].
Figure 9:Changes of ammonia water quality.
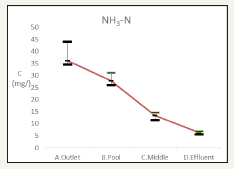
Figure 10:Change the water quality of T.N.
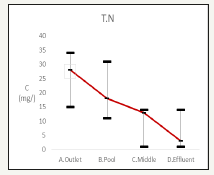
Total nitrogen
The total nitrogen results are in Figure 10. The influent is 35.67±25.38mg/L, the effluent is 5.67±7.37g/L while the recreation pond is 20±10mg/L. The promising total nitrogen removal indicated nitrification and denitrification occurred.
Total phosphorus
The total phosphorus data are in Figure 10. The influent is9.53±1.01mg/L, the effluent is 14.2±5.86mg/L while the recreation pond is 14.2±5.86mg/L. The total phosphorus removal rate is 47.9%. The phosphorus removal is not very significant in the studied system which is consistent with previous study.
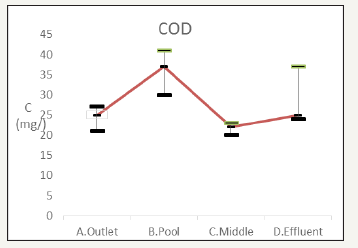
SOD
Determination of SOD is an indicator of river has a greater impact on the natural water of dissolved oxygen. Research has pointed out, SOD may account for the rivers of the total oxygen consumption of 40-50%, the ability to understand the purpose of measuring the sediment in water consumption of dissolved oxygen (DO) Figure 11.
Figure 11:The change of total phosphorus.
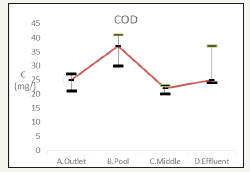
The wetland reaches the more oxygen-rich environment of the discharge orifice provided upstream and downstream river ecological pond aerator, Sediment Oxygen consumption was determined by linear regression 0.494367g O2/m2d and 0.466609g O2/m2d; in addition to the middle of the wetland 0.255416g O2/m2d.
Methane emission
The methane emission is increase in day time and decrease at night. Methane anaerobic microbial-degradation is the major reason [14].
Conclusion
Nitrification occurred in the low organic condition and enough oxygen. Plant uptake might induce Nitrogen decreasing. Nitrification and Denitrification also play the important roles Figure 12. It is promising to employ constructed wetlands to enhance water quality in tropical regions like Taiwan.
Figure 12:Change the water quality of BOD.
References
- An YJ, Kampbell DH, Breidenbach GP (2002) Escherichia coli and total coliforms in water and sediments at lake marinas. Environ Pollut 120(3): 771-778.
- Alvarez JA, Becares E (2006) Seasonal decomposition of Typha latifolia in a free-water surface constructed wetland. Ecological Engineering 28(2): 99-105.
- Aslam MM, Malik M, Baig MA (2007) Treatment performances of compost- based and gravel-based vertical flow wetlands operated identicaly for refinery wastewater treatment in Pakistan Ecological Engineering 30(1): 34-42.
- Bavor HJ, Roser DJ, Smalls IC (1989) Performance of solid-matrix wetland systems viewed as fixed film bioreactors. In: Constructed Wetlands for Wastewater Treatment Municipal, USA.
- Bartram J, Burch M, Falconer I, Jones G (1999) Situation assessment, planning and management. In: World Health Organization, Toxic cyanobacteria in water. Monitoring and Management, London, England.
- Bojcevska H, Tonderski K (2007) Impact of loads, season, and plant species on the performance of a tropical constructed wetland polishing effluent from sugar factory stabilization ponds. Ecological Engineering 29(1): 66-76.
- Brix H (1997) Do Macrophytes play a role in constructed treatment wetland? Water Science Technology 35(5): 11-17.
- Campbell CS, Ogden MH (2005) Constructed Wetlands in the Sustainable Landscape, John Wiley & Sons Lnc, Chichester, New York, USA.
- Calheiros CSC, Rangel, AOSS, Castro PML (2007) Constructed wetland systems vegetated with different plants applied to the treatment of tannery wastewater. Water research 41: 1790-1798.
- Chen XC, Kong HN, He SB, Wu DY, Li CJ, et al. (2009) Reducing harmful algae in raw water by light-shading. Process Biochemistry 44(3): 357-360.
- Chung AKC, Wu Y, Tam NFY (2008) Nitrogen and phosphate mass balance in a sub-surface flow constructed wetland for treating municipal wastewater. Ecological Engineering 32(1): 81-89.
- Davis K, Anderson MA, Yates MV (2005) Distribution of indicator bacteria in Canyon Lake California. Water Research 39(7): 1277-1288.
- Dierberg FE, DeBusk TA (2008) Particulate phosphorus transformations in south Florida stormwater treatment areas used for Everglades protection. Ecological Engineering 34(2): 100-115.
- Fleming SMS, Horne AJ (2006) Balancing wildlife needs and nitrate removal in constructed wetlands: The case of the Irvin Ranch Water District’s San Joaquin Wildlife Sanctuary. Ecologic Engineering 26(2): 147- 166.
© 2018 TY Yeh. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






