- Submissions

Full Text
Clinical Research in Animal Science
Cabergoline Monotherapy in a Chilean Creole Horse with Pituitary Pars Intermedia Dysfunction (Cushing’s Disease): A Case Report
Adolfo F Godoy* and Christian I De la Fuente
Department of Clinical Science, Chile
*Corresponding author:Adolfo F Godoy, Equine Clinic and Surgery, Department of Clinical Science, School of Veterinary Sciences and Livestock, Chile
Submission: June 27, 2022;Published: August 19, 2022

ISSN: 2770-6729Volume 2 - Issue 3
Abstract
Chilean Creole mare, 25 years old, with bilateral support claudication in both forelimbs for 3 months. The clinical inspection showed signs of hypertrichosis, hirsutism, fat deposits at the base of the tail and neck, excessive sweating, muscular atrophy and visible horizontal rings in both hooves. In the radiological study of distal phalanges of forelimbs, septic pedal osteitis and subsolar abscesses were seen. Based on the clinical history, a dexamethasone suppression test was performed, obtaining conclusive Pituitary Pars Intermedia Dysfunction (PPID) results. Cabergoline treatment was initiated at a dose of 2mg orally every 24 hours. After 3 months of therapy, a total resolution of the clinical signs was obtained and a normal response to the dexamethasone suppression test was observed. The patient was monitored for 30 months while continuing the treatment, without observing recurrence of the condition or adverse effects to the treatment.
Keywords:Equine; Carbegoline; Cushing’s disease; Pituitary
Abbreviations: PPID: Pituitary Pars Intermedia Dysfunction; AAEP: American Association of Equine Practitioners; ACTH: Adrenocorticotropic Hormone; POMC: Pro-Opiomelanocortin; α-MSH: Α-Melanocyte Stimulating Hormone
Introduction
The dysfunction of the Pars Intermedia of The Pituitary (PPID) is observed as a neurodegenerative disease with loss of dopamine inhibition on melanotropic cells. [1]. Commonly referred to as equine Cushing syndrome, this disease usually occurs in older animals. The drug of choice for medical treatment is pergolide, a synthetic derivative of the ergot alkaloid, a dopamine receptor agonist administered to horses with PPID to restore the dopaminergic inhibition of melanotrophs [2]. Like pergolide, the cabergoline is also an agonist of dopamine D2 receptors. [3]. A greater degree of effectiveness and duration of cabergoline activity has been demonstrated in horses compared to pergolide to counteract sulpiride’s antidopaminergic activity in prolactin production [4]. In 2007, the pergolide was prohibited for use in humans due to reports of cardiac side effects. Despite this, its use is allowed in veterinary medicine by the Food and Drug Administration, but the drug is not available in many countries. Given this situation, cabergoline could be a valid option for the treatment of PPID. According to the authors’ knowledge, this is the first report of a case of suspected PPID treated with oral cabergoline, in which the clinical evolution of the patient is described, as well as the changes in some hematological parameters.
Case Presentation
Chilean Creole mare, 25 years old, presented severe claudication of both forelimbs, grade 4/5 according to AAEP classification for the last 3 months. In standing position, the animal presented protraction of the forelimbs to reduce the pressure exerted on them. The clinical examination of the hooves presented heat, digital pulse, pain with hoof tester pressure and horizontal rings. The animal was attentive and reactive to the environment presenting a body condition of score 5 (range 1-9). On inspection, abnormalities in hair development were observed, including hypertrichosis and hirsutism, areas of thinning and partial hair loss (Figure 1). Excess fat was also observed at the base of the neck, excessive sweating throughout the body and epiaxial muscular atrophy (Figure 2). According to the information provided by the owner, the signs of polydipsia and polyuria were evident. In the clinical examination, the animal presented all normal vital signs. Three x-ray views (dorsopalmar, lateromedial, dorsoproximalpalmarodistal oblique) were obtained from the distal phalanx in both forelimbs, which showed evident radiolucent areas in the third phalanx compatible with subsolar abscesses and septic pedal osteitis. The blood count and blood biochemical profile results showed only a slight increase in the albumin/globulin ratio, with a value of 1.86 (range 0.80-1.00). Based on the signology shown by the mare, an intermediate Pituitary Gland Dysfunction (PPID) was suspected, whereby ACTH was measured resulting in a value of 60pg/ml. Then a dexamethasone suppression test was performed [5], meaning that a serum sample for basal cortisol measurement was collected at 5:00pm, followed by the administration of 40μg/ kg of intramuscular dexamethasone. Then a second serum sample was obtained 19 hours after the administration of dexamethasone. Basal cortisol values and after dexamethasone administration were 2.90μg/dl and 2.80μg /dl respectively. This result, associated with the clinical signs observed in the animal, suggested a diagnosis of dysfunction of the intermediate pairs of the pituitary gland [5].
Figure 1:Hypertrichosis, hirsutism, areas of thinning and incomplete hair loss at the cervical level of the equine.
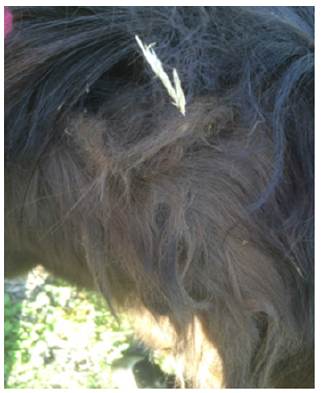
Figure 2:Excess fat was also observed at the base of neck, excessive sweating throughout the body and epiaxial muscular atrophy.

Treatment
Subsolar abscesses and septic pedal osteitis were treated by surgical drainage and debridement, followed by aseptic bandage and daily washing with a 2% chlorhexidine solution. Pharmacological treatment, phenylbutazone 1000mg (SID), sodium penicillin G 22,000 IU/Kg IV (QID), Gentamicin 6.6mg/kg IV / (SID) was administered. The treatment focused on PPID, consisted of oral cabergoline 2mg administration once a day (SID). At 12 weeks posttreatment, ACTH measurement, basal cortisol measurement and post-dexamethasone suppression were performed. ACTH values 43.1pg/mL (10.0-110.0pg/mL), basal cortisol of 3.60mg/dL (1.30- 2.93ug/dL) were observed. At 19 hours after the dexamethasone suppression test, cortisol values of 1.00ug/dL (1.30 - 2.93ug/ dL) were observed. Regarding the clinical aspects of the patient, considerable improvement was observed in the specimen’s coat, and no more polydipsia-polyuria or excessive sweating was observed (Figure 3).
Figure 3:Mare 30 days after the start of therapy with cabergoline, a significant decrease in hirsutism is observed.

Discussion
Pituitary intermediate pars dysfunction is one of the most common endocrine diseases in horses. Dexamethasone suppression is an appropriate laboratory diagnosis test, despite the possible side effects that could be generated by the injection of corticosteroids [6]. Also, the performance of other tests such as the measurement of ACTH, can be affected by various factors such as breed, age, coat color, diet, health status, time of year and place of residence [7]. The treatment of PPID in horses is usually performed with pergolide, a dopamine agonist administered to restore the dopaminergic inhibition of melanotrophs. D2 receptor binding inhibits Pro-Opiomelanocortin (POMC) synthesis and decreases the secretion of αMSH, ACTH and other hormones derived from POMC [8]. This same effect is achieved with dopaminergic agonist cabergoline type D2, which due to its prolonged action may be a useful alternative in the treatment of PPID [9]. In horses, a greater degree of effectiveness and duration of cabergoline activity has been demonstrated compared to pergolide to counteract the sulpiride antidopaminergic activity, which stimulates prolactin production, [10]. Lactotropic secretion of prolactin is controlled by tonic suppression of hypothalamic dopamine in the same manner as αMSH secretion of melanotropes. In the studies by Herbert et al. [4] and Oberhaus et al. [10] it was observed that a dose of 5mg of cabergoline generated a great suppression of prolactin in mares. In this clinical case, Chilean Creole mare, 1mg of cabergoline was administered first orally every 24 hours. At 3 weeks of treatment, less sweating and hirsutism was observed. It was decided to increase the dose to 2mg orally every 24 hours. After 3 months of therapy a significant improvement of the animal was observed, in relation to hirsutism, hyperidrosis and polydipsia and polyuria, neither abscesses nor secondary infections were observed in the hooves.
A second dexamethasone suppression test was performed with the same considerations as the test that allowed the diagnosis and the values obtained were: basal cortisol was 3.60mg/dL (1.30- 2.93ug/dL). At 19 hours after the application of dexamethasone, values of 1.00ug/dL (1.30-2.93ug/dL) were observed, which is consistent with the improvements in the clinical aspect of the equine under study. After 30 months of starting the therapy, the mare has not presented recurrence, remain hair and hooves without problems (Figure 4), also, has presented baseline cortisol and ACTH values in normal ranges. The multi-systemic consequences of PPID make it necessary to establish management and pharmacological measures in the affected animals. In Chile there are no drugs with proven efficacy for the control of this disease in horses, such as pergolide. We decided to use cabergoline due to the pharmacological similarities with pergolide, as well as the results obtained from human studies on the control of the PPID. In regard to the dose used we based this decision on a previous published work [4] in which a dose of 5mg of cabergoline generated suppression of sulpiridestimulated antidopaminergic activity. Therefore, we proposed starting with a total dose of 1mg of cabergoline that would be evaluated based on the clinical signs changes, the baseline cortisol values and the response to the dexamethasone suppression test. Afterwards, it was increased to 2mg of carbegoline orally, obtaining excellent clinical results after 30 days treatment. A dose of 2mg of cabergoline orally caused the remission of clinical symptoms of an equine with Pituitary pars intermediate dysfunction, producing a satisfactory response to the dexamethasone suppression test of the clinical case, 30 days after the drug was administered, and no recurrence of clinical signs was observed while the treatment lasted as indicated.
Figure 4:Mare 90 days after the start of treatment, the disappearance of hirsutism and an important recovery of the muscle mass of the specimen is observed.

References
- McFarlane D (2007) Advantages and limitations of the equine disease, pituitary pars intermedia dysfunction as a model of spontaneous dopaminergic neurodegenerative disease. Ageing Res Rev 6(1): 54-63.
- Frank N (2015) Pituitary pars intermedia dysfunction. In: Sprayberry K, Robinson N (Eds.), In Robinson’s Current Therapy in Equine Medicine, Elsevier, Netherlands, pp. 574-577.
- Seeman P (2007) Antiparkinson therapeutic potencies correlate with their affinities at dopamine D2(High) receptors. Synapse 61(12): 1013-1018.
- Hebert R, Thompson D, Mitcham P (2013) Inhibitory effects of pergolide and cabergoline formulations on daily plasma prolactin concentrations in geldings and on the daily prolactin responses to a small dose of sulpiride in Mares. Journal of Equine Veterinary Science 33(10): 773-778.
- Dybdal N, Hargreaves K, Madigan J (1994) Diagnostic testing for pituitary pars intermedia dysfunction in horses. J Am Vet Med Assoc 204(4): 627-632.
- McFarlane D (2019) Diagnostic testing for equine endocrine diseases: Confirmation versus confusion. Veterinary Clinics of North America: Equine Practice 35(2): 327-338.
- Jacob S, Geor R, Weber P (2018) Effect of dietary carbohydrates and time of year on ACTH and cortisol concentrations in adult and aged horses. Domest Anim Endocrinol 63:15-22.
- Frank N, Tadros E (2014) Insulin dysregulation. Equine Veterinary Journal 46(1): 103-112.
- Godbout A, Manavela M, Danilowicz K (2010) Cabergoline monotherapy in the long-term treatment of Cushing’s disease. Eur J Endocrinol 163(5): 709-716.
- Oberhaus E, Thompson L, Arana N (2016) Effect of repeated cabergoline treatment on the vernal transition and hair shedding of mares (Year 1) and a subsequent comparison of the effect of starting date on prolactin suppression (Year 2). J Equine Vet Sci 40: 41-48.
© 2022 Adolfo F Godoy. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






