- Submissions

Full Text
Cohesive Journal of Microbiology & Infectious Disease
Candida Parapsilosis Sensu Stricto Can Act as a Pathobiont in Conditions of Oral Dysbiosis
Rodríguez L1,2*, Rosa A3, Nastri L3, Nastri N4 and Jewtuchowicz V1,5
1Research Microbiology and Parasitology Institute, Argentina
2Semiology and Clinical Department, Argentina
3School of Dentistry, Microbiology Department, Argentina
4School of Dentistry, Argentina
5HIGA Gandulfo Hospital, Laboratory Service, Argentina
*Corresponding author: Rodríguez L, Research Microbiology and Parasitology Institute, Argentina
Submission: October 10, 2019; Published: December 20, 2019

ISSN 2578-0190 Volume3 issues3
Abstract
Recent publications have reported high prevalence of Candida parapsilosis Sensu Stricto in oral cavity niches. Our research group performed a pilot study in 2017 which showed that of the psilosis complex, Candida parapsilosis Sensu Stricto is the species most frequently isolated from oral cavity niches. Under inflammatory conditions, the probability of recovering it is almost four times higher, and it displays higher biofilm-forming capacity in-vitro, differing significantly from isolates of the same species obtained in conditions of eubiosis. This led us to hypothesize that an oral environment in dysbiosis overregulates virulent genes, promoting a more pathogenic phenotype. Accordingly, we decided to re-evaluate the in-vitro biofilm formation assay using a larger sample, under two nutritional conditions, using colorimetric methods for quantification. In addition, the results obtained were validated by imaging techniques. For both clinical conditions (eubiosis and dysbiosis), high biofilm forming phenotype predominated with both reading methods, and for both nutritional conditions tested. XTT showed significant difference between absorbance values for isolates from buccal dysbiosis and eubiosis (P=0.0025). Candida parapsilosis Sensu Stricto cells that colonize oral cavity niches are basically strong biofilm formers, regardless of in vitro growing conditions. However, the oral cavity in dysbiosis probably promotes virulence in this species due to inheritable epigenetic modifications.
Keywords: Candida parapsilosis sensu stricto; Biofilm; Oral eubiosis; Oral dysbiosis
Introduction
Reports from 2005 suggest that the organisms residing in the oral microbiome and their genomes are critical to health and disease [1,2]. In 2010, the first paper was published determining fungal microbiota in healthy humans by using independent culture approaches based on the analysis of the ITS region (fungal internal transcriptional spacer). It reported that, under the study conditions, the oral cavity would be colonized by over 70 fungal genera, with Candida being the most prevalent, having been isolated from 75% of participants. This is consistent with global consensus, which establishes that an average 40-60% of the healthy population carries species of the genus Candida in saliva or oral mucosa [3-6]; and that there are local and systemic conditions that may transform this ecological niche into a favorable site for overgrowth of this genus [5,7]. The species C. albicans is the most prevalent, in both immunocompetent and immunocompromised subjects [3,7-11]; with other species such as Candida parapsilosis, Candida dublinensis, Candida tropicalis, Candida guillermondi, among others, being isolated in smaller proportions, and having been recovered from both periodontal sites and oral mucosa [11,12-16]. Nevertheless, in the past ten years, some studies have reported greater frequency of recovery of non-albicans Candida (NAC) species in oral mucosa. Such is the case of Candida parapsilosis, whose frequency of isolation in that niche has increased from 10% in 1996 [3] to 15.0% in 2010 [9], 15.4% in 2011 [10], and 25.0 - 80.0% in 2017 [16,17]. Among the local and/or systemic factors which have been reported as associated to this shift in the oral distribution of Candida species are the use of prosthetic devices [13], old age [18], old people with low body mass index [19], oncological patients undergoing chemotherapy and radiotherapy [20,21], diabetics with poor metabolic control [22], males taking androgenic steroids [23], and females taking oral contraceptives [24]. Of the NAC species, Candida parapsilosis has arisen in recent years as a high-impact hospital pathogen [25], attracting the attention of medical staff. Candida parapsilosis belongs to a complex of three species (Candida parapsilosis sensu stricto, Candida orthopsilosis and Candida metapsilosis) which are phenotypically indistinguishable but genetically diverse, called Candida parapsilosis complex (psilosis complex) [26]. According to the literature, of the three species in the psilosis complex, Candida parapsilosis sensu stricto predominates in different human ecological niches, both in sickness and in health, and is the species most frequently recovered from invasive infections, both in adults and in premature neonates [27-29].
For many years, Candida albicans was the Candida species most frequently isolated from the oral cavity, whether pathological or healthy. However, a recently published paper on a prospective, comparative study reported greater prevalence of Candida parapsilosis sensu stricto in the oral cavity, with 83.3% isolation frequency higher than Candida albicans (56.7%)-in both pathological and healthy oral conditions, without significant differences between clinical groups [17]. Our research team conducted a pilot study in 2017, which showed that of the psilosis complex, the species Candida parapsilosis sensu stricto was the most frequently isolated from oral cavity niches. Under inflammatory conditions, the probability of recovering it was almost four times higher, and it displayed increased biofilm forming capacity in-vitro, with a significant difference over isolates of the same species obtained from conditions of eubiosis or health [30]. This outcome led us to hypothesize that an oral environment in dysbiosis overregulates virulence genes through epigenetic changes, promoting a more pathogenic phenotype. We therefore decided to evaluate the in-vitro biofilm formation assay under two nutritional conditions, using two colorimetric methods for quantification. In addition, the results were validated with imaging techniques: optical microscopy (OM) and confocal laser scanning microscopy (CLSM).
Materials and Methods
We designed a basic, retrospective, cross-sectional comparative study using a collection of 50 Candida parapsilosis sensu stricto oral isolates defined by conventional phenotypic methods (differential chromogenic medium “CHROMagar-Candida” [Becton-Dickinson]; micromorphology, and Vitek2), and molecular methods (endpoint PCR with specific primers derived from the region ITS1-5.8S-ITS2; plus Sanger sequencing and bioinformatic analysis with nucleotide BLAST [Blastn]). The isolates were obtained from a previous study on adult patients aged 18 to 65 years, immunocompetent, non-smokers, with no history of use of antimicrobial agent use during the 6 months prior to sampling; with or without prosthetic devices; with or without diagnosed gingival-periodontal disease, using the criteria of the American Association of Periodontology for diagnosis of periodontal diseases and conditions [31]. Samples were taken by a calibrated dentist following a procedure approved by the ethics committee of the School of Dentistry of Buenos Aires University, resolution number 012/2016CETICAFOUBA.
Isolates and media
To study the potential impact of a dysbiotic environment on the virulence of Candida parapsilosis sensu stricto, we used isolates of Candida parapsilosis sensu stricto (defined by phenotypic and conventional studies) from patients diagnosed with gingival-periodontal disease (GPD). GPD is a model par excellence for chronic inflammatory disease and is characterized by an association of alterations in oral pH, REDOX imbalance, oxidative stress, and changes in diversity and composition of the microbiota towards a polarization of the red and orange complex [32,33]. These attributes make GPD a good model for studying the impact of the oral microenvironment on the phenotype of a given microbial model. For the experimental phase, we used an strain of Candida albicans ATCC 10231 as positive control for the run, because it has been declared a pathogen according to the criteria of the CLSI (Clinical and Laboratories Standard Institute) [34], in addition to the 50 isolates of Candida parapsilosis sensu stricto taken from different sites in the oral mucosa (cheek, tongue, palate) and subgingival niches. The preserved isolates were reconstituted in BHI (brain-heart infusion) broth (Merck) at 37 °C. The growths obtained in BHI were plated in Sabouraud Dextrose Agar (SDA) supplemented with chloramphenicol (Becton Dickinson), incubated at 28 °C [34]. The biofilm assay was conducted in two nutritional media: YPD supplemented with chloramphenicol (Becton Dickinson) and RPMI 1640 supplemented with L-glutamine (Life technologies).
Analysis of virulence in-vitro
We measured in-vitro virulence through biofilm-forming ability in 96-well polystyrene microtiter plates (catalog number 167008 from Nunc or from Techno Plastic Products AG) by total biofilm biomass quantification assay with crystal violet (CV), in two different nutritional conditions: 1) YPD broth 2X (supplemented with antibiotic), described by Peteers [35], and 2) RPMI 1640 medium 1X (supplemented with L-glutamine), described by Treviño et al. [36]. Each sample was analyzed in 4 replicas and in two independent experiments. To increase the confidence level of the results, we used an alternative method for quantification of biofilm in-vitro based on measuring metabolic activity in each well by enzymatic reduction of the tetrazolium salt XTT (2,3-bis[2-methoxy-4-nitro-5-sulfophenil]-5-[(phenylamino) carbonyl]-2H-tetrazolium hydroxide) by mitochondrial dehydrogenases, which are only active in viable fungal cells [35].
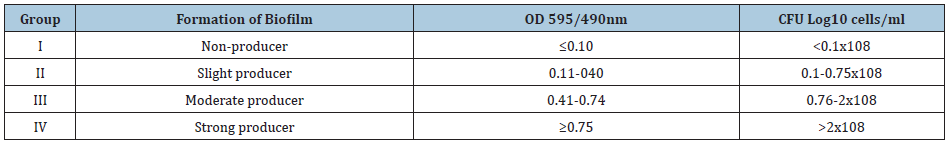
The quantity of biofilm formed by an isolate was classified according to the guidelines by Sánchez [37] & Treviño [36] as: high producers (≥0.41), low producers (0.11-0.40), and null producers (≤0.10). Sánchez et al. [37] break down high producers into strong and moderate biofilm producers (Table 1).
Induction of biofilm in YPD broth
Exponential phase isolates (18 hours) were obtained in SDA agar supplemented with chloramphenicol, with which to prepare suspensions in sterile saline solution with optical density 1x107 cells per milliliter. For each isolate, 100uL of inoculum was dispensed in 100uL of YPD 2X broth in each well. Four replicas were prepared for each isolate, with a positive control for the run (Strain ATCC Candida albicans 10231). As a blank, we used sterile saline solution without inoculum, in 100uL of YPD broth, in order to normalize absorbance in each assay. Plates were incubated with shaking (125rpm) for 24 hours at 37 °C [38]. After that time, they were evaluated with 0.1% CV only [35].
Table 1: Classification of biofilm-forming ability for optical density 595 and 490nm.
Induction of biofilm in RPMI 1640 medium supplemented with L-glutamine
Isolates in exponential phase (18 hours) were obtained in SDA agar supplemented with chloramphenicol, with which to prepare suspensions in RPMI 1640 medium, with optical density 1x107 cells per milliliter. For each isolate, 100uL of inoculum was dispensed per well, with 4 technical replicas plus one positive control for the run (strain Candida albicans ATCC 10231). As a blank, we used RPMI medium without inoculum, in order to normalize absorbance in each assay. Plates were incubated with shaking (125rpm) for 24 hours at 37 °C [36], after which they were evaluated by CV and by XTT salt.
Crystal violet (CV) assay
After 24 hours’ incubation, each well was rinsed with sterile saline solution, left to dry for 20 minutes at 37 °C and stained with 0.1% CV. It was left to react for 45 minutes in the dark, then rinsed 3 times with sterile distilled water and left to dry for 20 minutes at 37 °C. To each well, 200uL of alcohol (96%) was added, and allowed to act for 45 minutes to induce discoloration. Finally, 100uL were taken from each well and transferred to a new 96-well polystyrene tray. Optical density (OD) was determined at a wavelength of 595nm in a Multiskan GO (Thermofisher), version 10040, series #1510-02746C, with Skanlt Software 3.2.1.4. The OD value of the negative or blank control was subtracted from the value of the test well [36].
XTT assay
After incubating for 24 hours, each well was rinsed twice with 200uL sterile saline solution. After rinsing, to each well was added 100uL sterile saline solution and 100uL XTT and menadione solution. The XTT stock solution was prepared by dissolving 4mg XTT (Sigma) in 10mL sterile saline solution preheated to 37 °C. This solution was complemented with 100uL of menadione stock solution (55mg menadione [Sigma] in 100mL acetone). This was prepared on each testing day. The plates were incubated at 37 °C in the dark for 4 hours, after which 100uL were removed from each well and transferred to a new plate. The concentration of the “formazan” product was determined by spectrophotometry at 490nm. The OD value of the negative control was subtracted from the OD value of each well test [36].
Evaluation of biofilm with optical microscopy (OM)
In order to observe Candida parapsilosis sensu stricto biofilm by MO, 4 isolates with different biofilm-forming capacity (high, low and null) according to the CV assay were selected at random. Suspensions of 1x107 cells per milliliter were prepared from each selected isolate to induce biofilm growth on 24-well polystyrene trays. A sterile Thermanox coverslip (Plastic Coverlips-Rochester, NY USA) 13mm in diameter was placed at the bottom of each well of the polystyrene tray under laminar flow. In each well with its coverslip, 500uL of the Candida parapsilosis inoculum was placed on 500uL culture medium (YPD broth) to induce biofilm growth for 24 hours, with shaking, at 37 °C. After the incubation period, the culture medium was removed by aspiration, and each well rinsed with 1mL sterile saline solution. Each Thermanox coverslip was removed from the bottom of each well using sterile tweezers and left to dry on filter paper at 37 °C for 20 minutes. Then the fixing and staining protocol was applied to each coverslip, and coverslip mounted on a slide for observation by MO, following the protocol described by Oggioni et al. [39]. Biofilm morphology and topology were examined under optical microscope (Olympus). Candida albicans ATCC 10231, confirmed as a pathogen by the CLSI and which has high biofilm-forming capacity was used as experimental control. For negative control, water was used instead of inoculum.
Evaluation of biofilm using Confocal Laser Scanning Microscopy (CLSM)
To obtain a 3-D reconstruction and evaluate biofilm architecture for this Candida species, we examined the biofilm formed by a representative isolate of each phenotype (high, low and null biofilm formers, according to the CV assay results) using CLSM. For CLSM analysis, biofilm growth was induced by following the same protocol as for MO, except for the staining system and mounting mechanism, for which the fluorescent dye acridine orange (AO) was used [40]. Each coverslip dyed with AO was mounted on a glass slide in inverted position. As a control, water was used instead of inoculum. Specimens were preserved in cold (4 °C) and protected from light until examination by CLSM. The study was performed with an Olympus FV1000 confocal laser microscope.
Statistical analysis
Data were processed and analyzed in Microsoft Excel 2010 and the InfoStat 2018 statistic package. Difference between means was evaluated by right-tailed Student’s test for two independent samples after verifying the assumptions of normality, independence and homogeneity of variances, with a 95% confidence interval, considering as significant a P value lower than error alpha (alpha= 0.05). Homogeneity of variances was tested with the F test, establishing a significance level of 10%, with the aim of increasing the power and minimizing error type II when not rejecting the null hypothesis of homogeneity of variances. Data normality was tested with Q-Q plot and Shapiro Wilks’ test with 20% significance level. This was done in order to increase precision when not rejecting the null hypothesis of normality. To determine whether there is significant association between biofilm-forming phenotype and clinical source of isolates, ANOVA with 2 fixed factors was used and K=1 observation, and Bonferroni’s test for pairwise comparisons. ANOVA validness was tested with Shapiro Wilks’ test, Q-Q plot test and Levene´s test on residuals. Presence or absence of outliers was determined by Grubb’s test for outliers.
Results
Measurement of biofilm biomass by crystal violet stain
Figure 1 shows that out of the total isolates studied (n=50) in RPMI medium, 94% (47/50 strains) exhibited biofilm-forming capacity. Only three isolates (0.06%) did not form biofilm invitro in RPMI. Bonferroni’s test showed that high biofilm-forming isolates differed significantly from low and null biofilm-forming isolates (P<0.0001), proving that the results observed are not random. In YPD medium, 74% of the isolates tested had high biofilm-forming phenotype. These results are like those obtained in RPMI. Bonferroni’s test showed that high biofilm-forming phenotypes differed significantly from low and null biofilm-forming phenotypes (P<0.0001), confirming that the observations are not random (Figure 2 & 3). Spearman’s test showed positive correlation (0.49) and statistically significant association (P=0.0001) between CV-YPD absorbance values and CV-RPMI absorbance values. Figure 4 confirms the result of Spearman’s test, showing that the correlation between the two variables is not only positive, but also linear. Considering this agreement in results, it can be said that the information obtained for biofilm-forming phenotype in the collection of study isolates is valid.
Figure 1: Frequency distribution for biofilmforming ability in RPMI 1640 culture medium. Note: 68% of the isolates formed high levels of biofilm in RPMI medium (RF: relative frequency). The difference between the three groups was determined by ANOVA 1 factor.

Figure 2: Frequency distribution for biofilmforming ability in YPD broth. Note: 88% of the isolates formed biofilm in this culture medium. (RF: relative frequency). The difference between the three groups was determined by ANOVA 1 factor.
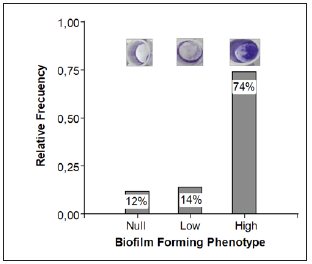
Figure 3: Dispersal diagram showing linear relationship between readings obtained for growth in RPMI medium and absorbance readings obtained for biofilm growth in YPD broth. Note: Pink lines indicate 95% confidence interval for each predicted or estimated value. Black line represents predicted or estimated value. Blue dots represent each value observed.
Figure 4: Comparison between mean absorbances (biofilm biomass level) according to clinical source (oral environment in presence and absence of GPD, equivalent to dysbiosis and eubiosis, respectively) in both nutritional conditions tested. Note: Data are expressed as mean and standard deviation and are representative of four technical replicas and two independent experiments. The difference in means was evaluated by the righttailed Student test for two independent samples.
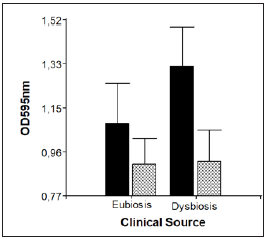
Figure 4 shows mean and standard deviation of absorbances for total Candida parapsilosis sensu stricto cells according to the oral condition in which they were isolated: with and without GPD, compared in two culture media: YPD and RPMI. In both culture media, the cells form greater biofilm biomass in conditions of dysbiosis or GPD (mean absorbance/YPD: 1.33±0.83; mean absorbance/RPMI: 0.93±0.62) than in oral health or eubiosis (mean absorbance /YPD: 1.08±0.95; mean absorbance/RPMI: 0.89±0.64). Nevertheless, comparison of means according to clinical origin using Student’s t-test found no significant difference [P= 0.047(YPD)/ P=0.1 (PRMI)] between the two nutritional conditions tested. Therefore, we cannot accept the hypothesis that the virulence of Candida parapsilosis sensu stricto increases in dysbiotic oral environments (Figure 5).
Figure 5: Frequency distribution for biofilmforming ability in RPMI medium shown by XTT. Note: Student’s test showed a significant difference (P<0.0001) between high and low phenotypes detected by this method (RF = relative frequency).

Measurement of biofilm metabolic activity by reduction of XTT salt
The choice of this technique as an alternative method for measuring biofilm formation in-vitro was based on the following criteria: a) The high inter-assay variability observed in the results with CV; b) the logarithmic relationship between the absorbance signal and the number of Candida albicans cells in two different strains in planktonic assays reported by Peteers et al. [35]; c) the correlation between absorbance values and microscopic image of biofilm reported by Pannanusorn [41]; and d) high repetitivity and minimal variation in absorbance among different experiments [41]. The XTT assay detected no null biofilm-forming ability; on the contrary, all isolates tested (50/50) displayed virulence. Figure 6 shows that only 16% of the isolates behave in vitro as low biofilm formers. The difference was statistically significant (P <0.0001), which also shows that within the species Candida parapsilosis sensu stricto there is intra-species variability in biofilm-forming ability, with a probability of error lower than 5% (Figure 7). Upon contrasting mean values for the variable OD490nm (metabolic activity in biofilm) according to clinical provenance, the t-test for two independent samples showed a statistically significant difference (P=0.0025). Figure 8 clearly shows this difference in biofilm metabolic activity according to clinical provenance (average absorbance of formazan in isolates from dysbiotic condition: 0.61±0.15 vs. average absorbance of formazan in isolates from eubiotic condition: (0.49±0.13). This result means that in-vitro biofilm-forming ability in this Candida species apparently also depends on clinical provenance. This raises the question of whether the biofilm-forming phenotype of Candida parapsilosis sensu stricto is significantly associated to clinical provenance of isolates. The hypothesis was tested by Bonferroni’s test, which provided a P-value of 0.04, indicating significant association. This result indicates that the high biofilm-forming phenotype is associated to a dysbiotic oral environment (Figure 7). The validity of the two statistical tests (t-test and ANOVA with Bonferroni’s test) was determined by the Shapiro Wilks’ test, Q-Q plot test and Levene’s test on residuals (distance between observed value and estimated or predicted value). The studentized residuals were used for normality tests. The absolute residuals according to clinical group were used for the homogeneity of variances with Levene’s test. The variable OD490nm did not follow normal distribution, however, Levene’s test generated a P-value greater than 0.10, therefore, the null hypothesis of homogeneity of variances was not rejected. This result enables the use of Student’s t-test and the Bonferroni test of the ANOVA. Due to the discrepancy between the results of the CV and XTT assays on biofilm-forming capacity and its association with clinical provenance, 3 isolates were randomly selected per phenotype and classified according to the CV assay results as high, low and null biofilm-forming, and used to evaluate the biofilm produced by MO and CLSM.
Figure 6: Comparison of average absorbance of the formazan product after reduction of XTT by viable fungal cells grown in RPMI 1640 medium, according to clinical provenance of isolates. Note: Data are expressed as mean and standard deviation and are representative of four technical replicas and two independent experiments. The difference between means was determined by Student’s right-tail test for two independent samples; (**): P<0.01.

Figure 7: Biofilm topography evaluated in isolates of Candida parapsilosis sensu stricto with different formative capacity viewed under MO (10X). Note: A: An isolate defined by CV assay as “high biofilm producer” forms a biofilm structure with the appearance of a spiderweb. B: An isolate defined by CV assay as “low biofilm producer” forms biofilm with reticular topology. C: An isolate defined by CV assay as “null biofilm producer” forms a biofilm structure based on strings of interconnected cells forming a net shape. In contrast to groups A and B, group C forms biofilm with lower cellular component, and the extracellular polymeric component is very scarce.

Figure 8: Biofilm metabolic activity measured at 490nm according to clinical provenance of isolates and biofilmforming capacity. Note: Mean and standard deviation for the variable OD490nm according to clinical provenance shown divided by biofilm-forming phenotype. Note that low biofilm-forming phenotype predominates in eubiosis. At the same time, the graph shows that the high biofilm-forming phenotype is significantly associated to oral disbiosis; (*): P<0.05. The statistical test applied for this analysis was ANOVA two factors, with K=1 observation. The results were corroborated by Bonferroni test afterwards.
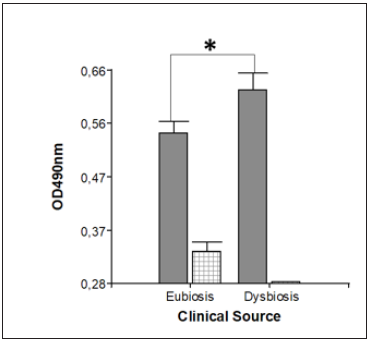
Figure 9: Biofilm topography evaluated in isolates of Candida parapsilosis sensu stricto with different formative capacity viewed under MO (40X). Note: Higher magnification shows the same results as described in Figure 9. We highlight the ultra-organized structure of fungal cells in group C, indicating that this group of isolates could not be classified as “null biofilmforming”.
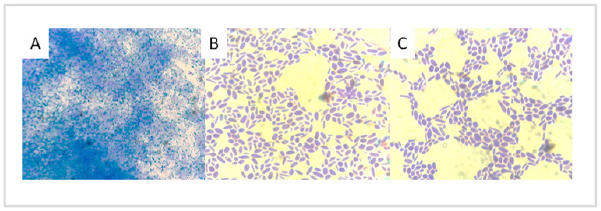
Figure 10: CLSM images of the architecture of biofilm in groups of Candida parapsilosis sensu stricto isolates with different biofilm-forming capacity (CV assay). Note: In group A (high biofilm formers), the number and size of peaks differs significantly from groups B (low biofilm formers) and C (null formers). There is no major difference in size and number of peaks between groups B and C.
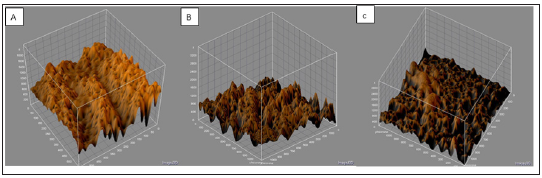
Figure 9 & 10 show that by MO, all Candida parapsilosis sensu stricto isolates form organized cell structures (purple) immersed in an extracellular substance (blue) with different topologies according to biofilm-forming capacity. Figure shows CLSM images the architecture of biofilm produced by each group of isolates (A, B and C). All groups form a 3D structure based on a series of peaks and valleys in differing numbers and amplitude according to biofilm-forming capacity.
Discussion
The etiopathogenesis of GPD has evolved over the past 5 decades, from being considered an infectious disease in which the microbial challenge was sufficient to cause clinical lesion, to being considered –according to the latest model by Hajishengallis and Lamont (2012) a disease determined by polymicrobial synergy and dysbiosis. This model claims that GPD is a pathology associated to an imbalanced biofilm, with increased levels of keystone pathogens, which not only increase in quantity, but also fulfill specific functions such as: a) reducing the host’s defense and b) interacting with other, less abundant pathogens and modulating the virulence of the entire microbial community. The interaction between this dysbiotic biofilm and the susceptible host determines the onset and progression of the disease [42-44].
According to a review published this year by Rodríguez the oral cavity under conditions of ecological equilibrium (Eubiosis) will probably acquire Candida parapsilosis sensu stricto strains (from the environment or direct person-to-person contact) with different potential virulence; but in a favorable context, under ecological and immunological balance (Th1-Th2-Th17-Treg), strains with low or null virulence capacity or symbionts are selected in the different oral niches. On the contrary, if homeostasis conditions in the oral cavity are altered, this imbalance may favor the selection of more virulent strains of Candida species, or pathobionts [45]. A pathobiont is a microorganism which, although it expresses attributes of virulence, nevertheless continues living in symbiosis with the host and therefore does not cause a clinically verifiable lesion [46]. This is possible if the Th1-Th2-Th17-Treg balance remains intact, which will depend mainly on an efficient Th17 response, since recent evidence shows that this response is crucial to anti-fungal defense in the mucosa, particularly oral mucosa [47-50]. When this balance breaks down, the clinical outcome may be oral candidiasis or invasive candidiasis.
We hypothesize that an oral environment in dysbiosis associated to overgrowth of pathobionts exacerbates the virulence of commensal Candida parapsilosis sensu stricto strains through inheritable epigenetic changes, since the variability in DNA sequence between strains of this species has been shown to be minimal, even between isolates from different geographic regions [51]. In this regard, we found that isolates from oral niches under conditions of GPD or dysbiosis formed more biofilm biomass than isolates from conditions of eubiosis, although the difference was not significant according to the CV assay. However, the alternative method we used for measuring biofilm metabolic activity by reduction of tetrazolium salts showed statistically significant differences in the biofilm-forming ability between isolates of this Candida species according to clinical provenance (dysbiosis and eubiosis). Moreover, the XTT assay showed association between biofilm-forming phenotype and clinical provenance of isolates, since statistical analysis showed significant association between these two variables, suggesting that biofilm-forming ability in this fungal modal may also be modulated by the conditions in the ecological niche or the environment. These results were consistent with those published by Hasan [52] and Jain et al. [53] who also suggested that the provenance and selection of isolates may be an influencing factor in biofilm formation by Candida species. This is the first study to analyze the behavior of oral isolates of Candida parapsilosis sensu stricto according to the clinical conditions in which they were isolated and the impact of the conditions of the oral environment on its pathogenicity. The results for biofilm quantification using CV were corroborated with an alternative method based on measuring the metabolic activity present in the biofilm by using tetrazolium salts such as XTT salt. This is a colorimetric method for indirect quantification of biofilm, as it measures the activity of mitochondrial dehydrogenase. Viable cells will be capable of reducing the XTT to a water-soluble product: formazan [35]. This solubility makes the product easily measurable in cellular supernatants. According to Pananusorn et al. [41] this point is important in research into biofilm, as it enables biofilm to be studied intact, without alterations to its structure, and the method is being used increasingly in studies evaluating fungal growth and sensitivity to drugs [41]. We prioritized the results obtained with XTT over those obtained with CV, mainly based on the reports by Pananusorn et al. [41] and Sánchez [37], who showed the existence of a direct association between colorimetric signal and number of cells. Nevertheless, some studies show that although this method is useful for comparisons involving a single strain, it is more problematic to use when different fungal strains and species are to be compared. With regard to this dilemma, Khun et al. evaluated the association between colorimetric signal (OD490nm) and number of cells (different concentrations of a suspension of fungal cells), using two different strains and species of Candida (Candida albicans and Candida parapsilosis); at two different concentrations of XTT (1X-1mg/mL and 5X-5mg/mL). That study found significant differences in the absorbance signal between the two Candida species and between the two Candida parapsilosis strains, suggesting that there are differences within the genus Candida regarding tetrazolium salt metabolizing capacity, both between strains and between species. The authors thus concluded that the method is 100% reliable for measuring biofilm within the same species, but when different Candida strains or species are to be compared, detailed standardization is required [54]. However, the sample size in the study is deficient, and Khun’s results should be validated using a greater number of isolates. In our study, based on the research by Khun et al. we decided to use an inoculum of 1x107 cells per milliliter and a 1X concentration of substrate (XTT); because these were the two conditions showed the least variability among Candida parapsilosis strains analyzed [54].
The results obtained by colorimetric methods (CV and XTT) to define biofilm-producing capacity in each isolate in the study group were confirmed by morphology using optical microscopy (OM) and confocal laser scanning microscopy (CLSM). Both these methods provided similar results for biofilm architecture and topology, and, in agreement with reports by Pannanusorn et al. [41] the biofilm-producing capacity and biofilm architecture and morphology are highly dependent on the Candida parapsilosis sensu stricto strain. Our study recognized different biofilm architectures and topologies according to biofilm-forming phenotype. About topology, the isolates classified by CV as high biofilm producers formed denser, more complex biofilm structures that looked like “cobwebs”, with plentiful cellular component and extracellular polymeric substance (EPS). This is consistent with the observations of Pannanusorn [55]. Among the isolates with low biofilm-forming phenotype, there was a single architecture based on more- or less-dense aggregates of yeasts forming a network with more or less EPS deposit, in agreement with Pannanusorn [55] and Serrano F [56]. Three-dimensional reconstruction showed conical structures of variable height and area according to the biofilm-forming capacity of each isolate, as a result of the high level of spatial cellular organization. We found no study analyzing 3D biofilm structure for this Candida species. The imaging results confirmed the XTT colorimetric method results, which only recognized isolate with high and low biofilm-forming capacity. The findings in this study suggest a new hypothesis on the role of the epigenome in the regulation of genes in this fungal mode. To date, the impact of the epigenome on virulence has only been studied in Candida albicans, and it has been suggested that the phenotypical variation described for Candida albicans may be regulated by epigenetic changes [57]. At the same time, the results of this study provide an important basis for designing and conducting further longitudinal studies which could reliably demonstrate that the oral cavity under pathological conditions may be a source of invasive infections by Candida species.
Conclusion
Biofilm-forming capacity in Candida parapsilosis sensu stricto species is intrinsically heterogenous. Nevertheless, the conditions of the ecological environment or niche may impact or condition its potential virulence, probably via epigenetic mechanisms. These need to be demonstrated through approaches evaluating the epigenome in this fungal model. The results of this study show the role of ecological niche as another key factor modulating virulence in this Candida species, potentially turning it into a pathobiont under conditions of ecological imbalance. We suggest that these observations should be corroborated at transcriptional level. The oral cavity, like the skin, is probably a route of infection for invasive candidiasis by Candida parapsilosis sensu stricto under pathological conditions. Clinical studies, particularly longitudinal and prospective studies, are needed to prove this hypothesis.
Conclusion
The study was supported by UBACYT subsidy from the University of Buenos Aires with number 2002017100050BA-2018.
References
- Jenkinson HF, Lamont RJ (2005) Oral microbial communities in sickness and in health. Trends Microbiol 13(12): 589-595.
- Avila M, Ojcius DM, Yilmaz O (2009) The oral microbiota: living with a permanent guest. DNA Cell Biol 28(8): 405-411.
- Kleinneger C, Lokhart S, Vargas K, Soll DR (1996) Frequency, intensity, species, and strains of oral candida vary as a function of host age. J Clin Microbiol 34(9): 2246-2254.
- Arendorf TM, Walker DM (1979) Oral candidal populations in health and disease. Br Dent J 147(10): 267-272.
- Yap M, Alnuaimi A, Adams G (2016) Oral candidal carriage in asymptomatic patients. Australian Dental Journal 61(2): 190-195.
- Samaranayake L (2009) Commensal oral candida in asian cohorts. Int J Oral Sci 1(1): 2-5.
- Javed F, Kheraif A, Kellesarian S (2017) Oral Candida carriage and species prevalence in denture stomatitis patients with and without diabetes. J Biol Regul Homeost 31(2): 343-346.
- Sanchez L, Estrada D, Pozos A (2013) Biofilm formation by oral clinical isolates of Candida species. Arch Oral Biol 58(10): 1318-1326.
- Ghannoum M, Jurevic R, Mukherjee P (2010) Characterization of the oral fungal microbiome (mycobiome) in healthy individuals. PLoS Pathogens 6 (1): e1000713.
- Yang Y, Leaw S, Wang A (2011) Characterization of yeasts colonizing in healthy individuals. Medical Mycology 49(1): 103-106.
- Terҫas A, Marques S, Monteiro C, Moffa EB, Alves MB, et al. (2017) Antifungical drug susceptibility of Candida species isolated from HIV-positive patients recruited at a public hospital in São Luis, Maranhão, Brazil. Front Microbiol 8: 298.
- Urzua B, Hermosilla G, Gamonal J, Morales-Bozo I, Canals M, et al. (2008) Yeast diversity in the oral microbiota of subjects with periodontitis: Candida albicans and Candida dubliniensis colonize the periodontal pockets. Medical Mycology 46(1): 783-793.
- Jewtuchowicz V, Brusca M, Mujica M (2017) Subgingival distribution of yeast and their antifungal susceptibility in immunocompetent subjects with and without dental devices. Acta Odontol Latinoam 20(1):17-22.
- Mushi M, Mtemisika C, Bader O (2016) High oral carriage of non-albicans Candida spp. among HIV infected individuals. International Journal of Infectious Disease 49: 185-88.
- Thanyasrisung P, Kesakomol P, Pipattanagovit P (2014) Oral Candida carriage and immune status in thai human immunodeficiency virus-infected individuals. Journal of Medical Microbiology 63: 753-759.
- Lourenco A, Rodrigues A, Nakao C (2017) Oral Candida spp carriage and periodontal disease in HIV-infected patients in Ribeirão Preto, Brazil. Rev Inst Med Trop S Paulo 59: 29.
- Peters B, Wu J, Hayes R (2017) The oral fungal mycobiome: characteristics and relation to periodontitis in a pilot study. BMC Microbiology 17: 157.
- Benito B, Aranda S, López F (2016) Oral Candida isolates and fluconazole susceptibility patterns in older Mexican women. Arch Gerontol Geriatr 9(65): 204-210.
- Zakaria M, Furuta M, Takeshita T (2017) Oral mycobiome in community-dpocillosing elderly and its relation to oral and general health conditions. Oral Dis 23(7): 973-982.
- Jain M, Shah R, Chandolia B (2016) The oral carriage of Candida in oral cancer patients of Indian origin undergoing radiotherapy and/or chemotherapy. J Clin Diagn Res 10(2): 17-20.
- Bulacio L, Paz M, Ramadan S (2012) Oral infections caused by yeasts in patients with head and neck cancer undergoing radiotherapy. Identification of the yeasts and evaluation of their antifungal susceptibility. J Mycol Med 22(4): 348-353.
- Melton J, Redding S, Kirkpatrick W (2010) Recovery of Candida dubliniensis and other Candida species from the oral cavity of subjects with periodontitis who had pocillos-controlled and poorly controlled type 2 diabetes: a pilot study. Special Care in Dentistry 30(6): 230-234.
- Brusca M, Verdugo F, Amighini C (2014) Anabolic steroids affect human periodontal health and microbiota. Clin Oral Investig 18 (6): 1579-1586.
- Brusca M, Rosa A, Olatz A (2010) The impact of oral contraceptives on women's periodontal health and the subgingival occurrence of aggressive periodontopathogens and candida species. Journal of Periodontology 81(7): 1010-1018.
- Trofa D, Gácser A, Nosanchuk JD (2008) Candida parapsilosis, an emerging fungal pathogen. Clin Microbiol Rev 21(4): 606-625.
- Gago S, García RR, Cuesta I (2014) Candida parapsilosis, Candida orthopsilosis, and Candida metapsilosis virulence in the non-conventional host Galleria mellonella. Virulence 5(2): 278-285.
- Canton E, Pemán J, Quindos G (2011) Prospective multicenter study of the epidemiology, molecular identification, and antifungal susceptibility of Candida parapsilosis, Candida orthopsilosis, and Candida metapsilosis isolated from patients with candidemia. Antimicrob Agents Chemother 55(12): 5590-5596.
- Constante C, Monteiro A, Alves S (2014) Different risk factors for candidemia occur for Candida species belonging to the Candida parapsilosis, complex. Medical Mycology 52(4): 403-406.
- Caggiano G, Lovero G, Giglio O (2017) Candidemia in the neonatal intensive care unit: a retrospective, observational survey and analysis of literature data. BioMed Research International 1-13.
- Rodríguez L, Rosa A, Rodríguez J (2017) The oral cavity: a reservoir that favors colonization and selection of Candida parapsilosis sensu stricto strains with high pathogen potential under conditions of gingival-periodontal disease. J Dent Sci Ther 2(1).
- Armitage G (1999) Development of a classification system for periodontal diseases and conditions. Ann Periodontol 4(1): 1-6.
- Bullon P, Morillo J, Ramirez TM (2009) Metabolic syndrome and periodontitis: is oxidative stress a common link? Journal of Dental Research 88(6): 503-518.
- Aiuto F, Nibali L, Parkar M (2010) Oxidative stress, systemic inflammation, and severe periodontitis. Journal of Dental Research 89(11): 1241-1246.
- Durán E, Mujica T, Jewtuchowicz V (2007) Examination of the genetic variability among biofilm-forming Candida albicans clinical isolates. Revista Iberoamericana de Micología 24(4): 268-271.
- Peeters E, Nelis H, Coenye T (2008) Comparison of multiple methods for quantification of microbial biofilms grown in microtiter plates. Journal of Microbiological Methods 72(2): 157-165.
- Treviño R, Rodríguez I, Rosas A (2015) Biopelícula formation and genetic variability of BCR1 gene in the Candida parapsilosis Rev Iberoam Micol 32(3): 180-184.
- Sanchez L, Estrada D, Pozos A (2013) Biopelícula formation by oral clinical isolates of Candida Archives of Oral Biology 58: 1318-1326.
- Laffey S, Butler G (2005) Phenoype switching affects biofilm formation by Candida parapsilosis. Microbiology 151(4): 1073-1081.
- Oggioni M, Trappetti C, Kadioglu A (2006) Switch from planktonic to sessile life: a major event in pneumococcal pathogenesis. Molecular Microbiology 61(5): 1196-1210.
- Facultad de Ciencias Exactas y Naturales. Universidad Nacional de Buenos Aires. Departamento de Química Biológica Microbiología e Inmunología Técnicas de tinció Fundamentos.
- Pannanusorn S, Fernandez V, Romling U (2013) Prevalence of biofilm formation in clinical isolates of Candida species causing bloodstream infection. Mycoses 56(3): 264-272.
- Kinane D, Stathopoulou P, Papapanou P (2017) Periodontal disease. Nature reviews disease primers 3(17038).
- Michaud D, Fu Z, Shi J (2017) Periodontal disease, tooth loss, and cancer risk. Epidemiol Rev 39(1): 49-58.
- Tomás I, Camelo A, Balsa C (2016) Nuevo modelo de patogenia de la periodontitis crónica: de la enfermedad infecciosa a la disbiosis polimicrobiana. RCOE 21(3): 131-145.
- Rodríguez ML, Jewtuchowicz VM, Rosa AC (2018) La mucosa oral como fuente potencial de candidemia por Candida parapsilosis sensu stricto, bajo condiciones patológicas SOJ Microbiol Infect Dis 6 (2): 1-10.
- Hornef M (2015) Pathogens, commensal symbionts, and pathobionts: discovery and functional effects on the host. ILAR Journal 56(2): 159-162.
- Richardson J, Moyes D (2015) Adaptive immune responses to Candida albicans Virulence 6(4): 327-337.
- Gow N, Vande VF, Brown A (2012) Candida albicans morphogenesis and host defence: discriminating invasion from colonization. Nature Review Microbiology 10(2): 112-122.
- Panagoda GJ, Ellepola AN, Samaranayake LP (2001) Adhesion of Candida parapsilosis to epithelial and acrylic surfaces correlates with cell surface hydrophobicity. Mycoses 44(1-2):29-35.
- Branchini ML, Pfaller MA, Rhine CJ (1994) Genotypic variation and slime production among blood and catheter isolates of Candida parapsilosis. J Clin Microbiol 32: 452-456.
- Tavanti A, Hensgens L, Mogavero S (2010) Genotypic and phenotypic properties of Candida parapsilosis sensu strictu strains isolated from different geographic regions and body sites. BMC Microbiology 10(203): 1-11.
- Hasan F, Xess I, Wang X (2009) Biofilm formation in clinical Candida isolates and its association with virulence. Microbes and Infection 11(8-9): 753-761.
- Jain N, Kohli R, Cook E (2007) Biofilm formation by and antifungal susceptibility of Candida isolates from urine. Appl Environ Microbiol 73(6): 1697-1703.
- Kuhn D, Balkis M, Chandra J (2003) Uses and limitations of the xtt assay in studies of candida growth and metabolism. Journal of Clinical Microbiology 41(1): 506-508.
- Pannanusorn S, Ramírez B, Lünsdorfet H (2014) Characterization of biofilm formation and the role of bcr1 in clinical isolates of candida parapsilosis. Eukaryotic Cell 13(4): 438-451.
- Serrano I, López E, Reyna G (2015) Influence of culture media on biofilm formation by Candida species and response of sessile cells to antifungals and oxidative stress. Biomed Res Int 783639.
- Lohse M, Johnson A (2010) Temporal anatomy of an epigenetic switch in cell programming: the white-opaque transition of Candida albicans. Molecular Microbiology 78(2): 331-343.
© 2019 Rodríguez L. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






