- Submissions

Full Text
Cohesive Journal of Microbiology & Infectious Disease
Viable Monitoring During the Filling of a Terminally Sterilized Pharmaceutical Product-Risk Based Approach
Ahmed Assem*
Tabuk Pharmaceuticals, Saudi Arabia
*Corresponding author:Ahmed Assem, Tabuk Pharmaceuticals, Saudi Arabia
Submission: July 21, 2017 ;Published: July 18, 2018

ISSN 2578-0190 Volume2 Issue1
Abstract
This article addresses a risk based approach to establish a routine monitoring program of viable (I.e. microorganisms) in a controlled environment used to produce pharmaceutical products that are intended to be terminally sterilized. Current applicable quality system regulation & GMP require appropriate environment to be established, maintained and monitored for the manufacturing of the terminally sterilized pharmaceutical meanwhile the appropriate environmental monitoring plan is not clearly specified & a risk based approach will be a valuable tool to design a suitable program
Introduction
Current regulatory environment emphasize on the use of enhanced knowledge over the manufacturing processes & product performance that can help in identifying the risks associated to the patient safety & the product quality. This article will describe briefly the Quality risk management & apply its elements on a model which is the Environmental monitoring during the filling of terminally sterilized products [1]. Such approach will insure the following
A. Product quality and patient safety
B. Meeting the regulatory expectations
C. Minimize the manufacturing cost through minimizing the monitoring frequencies.
The following procedure will be followed during the Risk assessment procedure
A. Identify The process
B. Identify Risk
C. Implement Risk Assessment
D. Determination of the RPN (Risk Priority Number)
E. Establish the monitoring frequency according to the determined RPN
Procedure
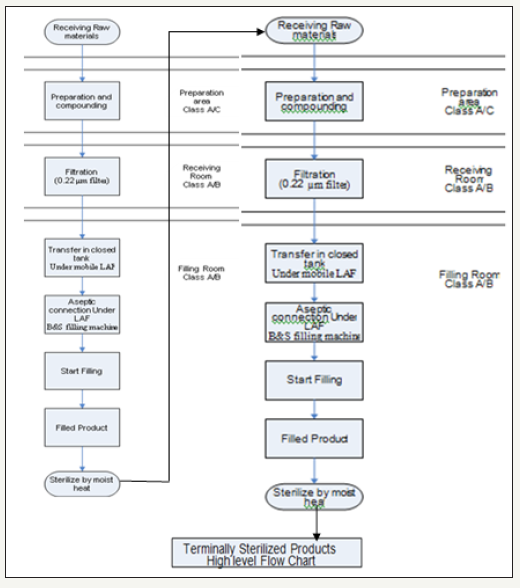
Identify the process
High flow chart for the manufacturing process of the terminally sterilized products:
Identify risk
Risk factors: The following factors represent the variables to be considered during the risk assessment process:
A. Risk factor (A): Amount of microbial contamination on, or in, a source.
B. Risk factor (B): Ease of dispersion, or transfer, of microorganisms
C. Risk factor (C): Proximity (location) of source from critical area.
D. Risk factor (D): Effectiveness of control method.
Table 1:
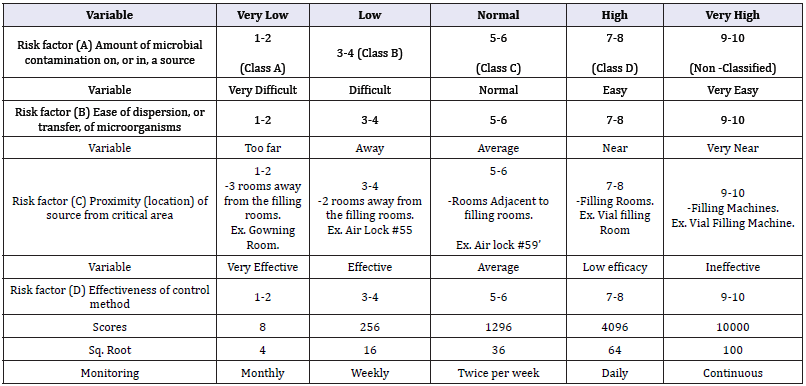
Risk numbering: The following table showing the risk numbering procedure (Table 1)
Risk priority number (RPN) determination: The RPN shall be determined by multiplying the risk number of the four variables [2]: (Figure 1)
Figure 1:
RPN= Risk factor (A) * Risk factor (B) * Risk factor (C) * Risk factor (D)
Monitoring frequency according to the determined RPN: As a safety factor the frequencies shall be determined [3] according to the square root of the determined RPN: The following table showing the environmental monitoring frequencies according to the determined RPN [4]: (Table 2)
Table 2:
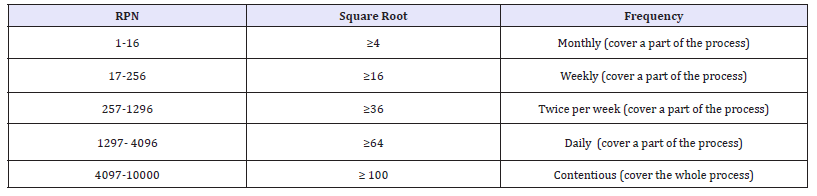
Table 3:
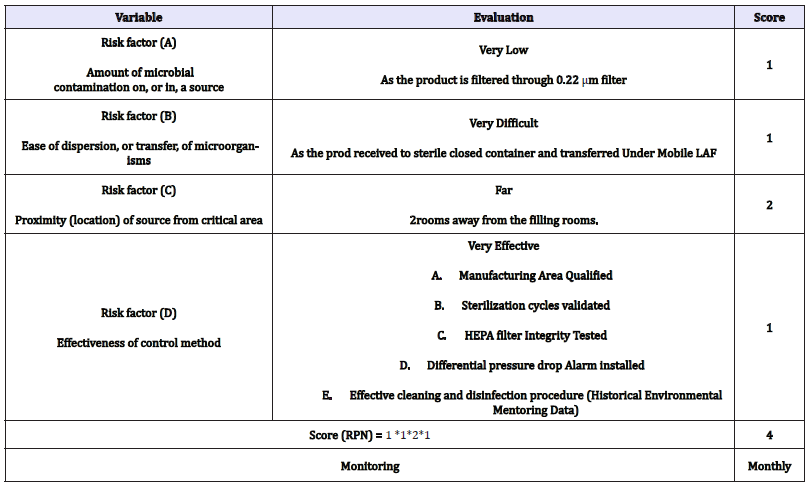
Table 4:

Risk assessment of the receiving process: Evaluation of impact of the environment on the product quality (Microbiological safety) during [5] the Receiving process: (Table 3) and Evaluation of impact of the environment on the product quality (Microbiological safety) during the Filling process [6]: ( Table 4)
References
- (2008) ICH Q9, Pharmaceutical Quality System, ICH Harmonised Tripartite Guideline.
- (2008) Technical Report 44. Quality Risk Management for Aseptic Processing. PDA J Pharm Sci Technol 62: (S1).
- (2012) Technical Report No. 30 (Revised 2012): Parametric Release of Pharmaceutical and Medical Device Products Terminally Sterilized by Moist Heat, Parenteral Drug Association.
- (2012) General Chapter <1116> Microbiological Control and Monitoring of Aseptic Processing Environments. USP 36/NF 31; US Pharmacopeia, USA.
- Eudra (2008) The Rules Governing Medicinal Products in the European Union: EU Guidelines to Good Manufacturing Practices for Medicinal Products for Human and Veterinary Use-Annex 1, Manufacture of Sterile Medicinal Products; European Commission, Belgium 4:
- Moldenhauer J. Environmental Monitoring, A Comprehensive Handbook. Parenteral Drug Association 1(2):
© 2018 Ahmed Assem. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






