- Submissions

Full Text
Biodiversity Online J
Brief Description for the Development of a Protocol for the Aseptic Culture of Quercus Robur. Part Two
Chiavazza Paola Maria*, Nosenzo Antonio, Meloni Fabio
DISAFA, University of Turin, Italy
*Corresponding author: Chiavazza Paola Maria, DISAFA, University of Turin, Italy
Submission: August 08, 2020; Published: August 25, 2020

Volume1 Issue2August, 2020
Results of Micropropagation
Nodal and apical segments of Quercus robur, coming from both adult plants and germinals gave rise, with different modalities and times, to the formation of stalks of a few cm in length (Figure1). At this point, the new nodes formed were put in culture. The lengthening of the new stems continued for a few months, with slow growth (about 1cm in 20-30 days). Following further withdrawals, the nodes put in culture in October and November 2016 stopped their growth. A physiological arrest is hypothesized, to be expected also in plants grown in vitro. The increase in auxin concentration alone (IAA 0.75mg/l), tested in an attempt to promote faster elongation, did not give the expected results. Trials are underway to use both concentrations and plant growth regulators in combination, in an attempt to resume the elongation of the new stalks. As mentioned above, tests are underway to obtain any somatic embryogenesis from adult leaf tissues. At this regard, in the face of enlargements on the surface of the tissue, somatic embryos have not yet been differentiated. Rooting tests have begun on the first elongated stems obtained in vitro.
Figure 1: Normal stem lengthening in Quercus robur explants at about 1 month of age.
For this purpose, glass jars containing expanded clay on the bottom, moistened with mineral salts, were used to form a substrate, at the same time, for containing and nourishing the explant during rooting. All of this while still maintaining the conditions of sterility, to avoid explants that are still free of roots without water stress. About a month after the transfer, the explants were checked for the emission of roots. At the moment, although the vital explants remain, a true radical capillary has not yet formed, but at the base of the same, enlargements of various shapes have been noted, probable signs of the formation of radical sketches, to date not yet visible. The study is therefore continuing on two fronts, that of micropropagation, currently not yet stabilized due to the arrest of the growth of the nodes formed in vitro and, at the same time, that of rooting, an equally delicate phenomenon in plants considered recalcitrant, such as the genus Quercus.
Description of the Methodology and Confirmation of the Acclimation of the Plants
Experiments with different combinations and concentrations of plant growth regulators (NAA and IAA as auxins, BA and KIN as cytokinins), did not lead to positive results. After a first initial elongation (a few cm, Figure 1), the stem darkens and subsequently necrotizes (Figure 2). The optimization of protocols aimed at the standardization of propagation techniques and subsequent rooting of material free from pathologies and difficult to multiply in vivo, when present with some adaptability characteristics, takes time. The in vitro rooting phase of Quercus explants and their subsequent acclimatization are also critical phases of the propagation process, strongly influenced both by the type of substrate and the species used (Figure 3). Some authors have also highlighted the existence of a rooting seasonality of the different species, which maintain a socalled “seasonality memory” similar to that possessed in vivo. In our case, the insufficient lengthening of the stem (except in rare cases) prevented the rooting of an adequate number of microcuttings (Figure 4). It has been shown that the microcuttings cannot root if the cauline apex does not reach a certain distance from the root (cutting with at least two to three knots). At the moment, tests are underway that aim to lengthen the oak internode, which is always very close. The plant growth regulator used is gibberellic acid, at concentrations varying between 5 and 50ppm. The continuation of the work concerning the multiplication of Quercus robur will be subject to joint evaluation.
Figure 2: Evident growth arrest of the stem in explant of 30 days.

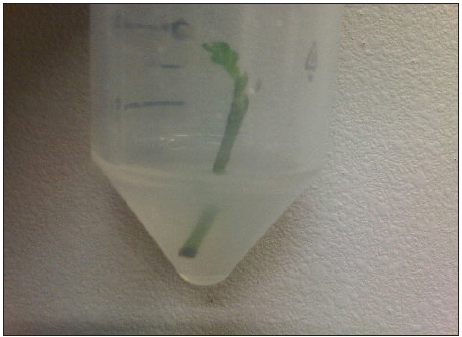
Figure 3: Application of in vitro rooting system.

Figure 4: Maintenance of viability conditions in explant about one month after rooting.

© 2020 Chiavazza Paola Maria. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






