- Submissions

Full Text
Aspects in Mining & Mineral Science
Microstructures of Dissimilar Materials Welded by Solid State Diffusion Bonding Process
Boumerzoug Z1*, Hamdi I1, Bedjaoui W1, Baghdadi L1, Chettah M1 and Delaunois F2
1LMSM, University of Biskra, Algeria
2Metallurgy Department, University of Mons, Belgium
*Corresponding author: Zakaria Boumerzoug, LMSM, University of Biskra, B P 145, Biskra, Algeria
Submission: August 18, 2021;Published: August 31, 2021

ISSN 2578-0255Volume7 Issue2
Abstract
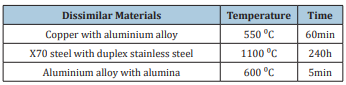
This article presents a microstructural study of three different pairs of dissimilar materials (copper with an aluminum alloy, X70 steel with duplex stainless steel and an aluminum alloy with alumina) welded by a diffusion bonding process. The bonded area was examined by light microscopy. Despite the difference in the physicochemical properties of the base materials, microstructural observation revealed that the three pairs of dissimilar materials were successfully joined by a solid state diffusion bonding process.
Keywords: Solid-state diffusion; Welding; Interface; Microstructure
Abbreviations: DSS: Duplex Stainless Steel; SSDW: Solid State Diffusion Welding
Introduction
It is known that bimetallic products, composed of two or more dissimilar materials, have been widely used in various industries due to their excellent properties [1]. Diffusion bonding is a solid state assembly process, used to join similar and dissimilar materials without coarse microscopic deformation and with minimum dimensional tolerances [2,3]. Diffusion bonding or diffusion welding is a solid state welding technique used in metalworking, capable of joining similar and dissimilar metals together. It works on the principle of solid state diffusion, in which the atoms of two solid metal surfaces become intercalated over time. This is usually accomplished at an elevated temperature, around 50-75% of the absolute melting temperature of the materials [4,5]. The temperature, pressure and bonding time are the three important parameters of the diffusion bonding process. A review of the literature suggests that most of the work on diffusion bonding has been done primarily to optimize the parameters of diffusion bonding process, namely pressure, time, temperature, and intermediate layers for diffusion bonding. of similar and dissimilar metals. Diffusion bonding has been widely applied to metals, such as aluminum and titanium alloys and stainless steel, and to silicon and other solid state electronic materials, where it is generally referred to as direct bonding of wafers, for joining special purpose materials where relatively large areas of contact effect are involved, and where other familiar deposition or assembly processes such as chemical vapor deposition or welding are generally not applicable [6-9].
For economic reasons, bonding may be preferred at low temperature due to ever increasing energy costs. Lower temperature bonding can also have the additional benefit of preserving the microstructure of the base metal near the bonded regions. Successful attempts have been made to develop diffusion bonds of similar metal types such as austenitic stainless steel [9], or dissimilar metals such as Fe-Al alloy and pure iron [10]. This article presents a microstructural study of three different pairs of dissimilar materials (copper with an aluminum alloy, X70 steel with duplex stainless steel and an aluminum alloy with alumina) welded by a diffusion bonding process.
Experimental Procedure
Table 1: Solid-state welding configuration applied for joining the dissimilar materials.
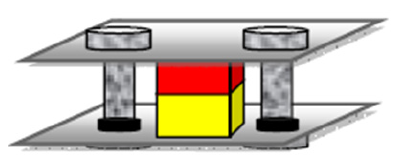
Pure copper, aluminum alloy, X70 steel, duplex stainless steel and alumina were chosen as the basic materials for this study. Before solid state diffusion bonding process, surfaces samples were polished and cleaned. Diffusion welding was performed in a vacuum chamber with heating the specimen at the appropriate temperatures (Table 1). The two dissimilar materials were fixed by a loading device. Figure 1 presents the schematic diagram of the diffusion bonding setup. For microstructural analysis, the samples were cross-sectioned, polished and then etched with a solution of a standard metallographic procedure.
Figure 1: Solid-state welding configuration applied for joining the dissimilar materials.
Results and Discussion
Figure 2 shows the microstructures of the three pairs of dissimilar joints (copper with aluminum alloy, X70 steel with duplex stainless steel and aluminum with alumina) welded by a solid state diffusion bonding process. During this process, the interdiffusion of atoms through the interface represents the main mechanism of the process. The heat treatment during the bonding process leads to the formation of an interface between the two dissimilar materials as observed in X70 steel/duplex stainless steel joint (Figure 2a) and aluminium/alumina joint (Figure 2b). However, some intermetallic layers were formed in copper/aluminium joint. The literature data for diffusion of copper and aluminum confirm a higher diffusion rate of copper in aluminum [11]. In addition, this study showed the possibility of joining metallic materials to non-metallic materials, because an aluminum alloy was welded to a ceramic material (Alumina) by the solid state diffusion bonding process. It is known the great difference of physicochemical properties between metallic materials and ceramics. Successful ceramic-metal bonding is of considerable practical interest for the development of electronic devices (Figure 2c).
Figure 2: Adsorption model of oleic acid on different cleavage planes of spodumene [9].

Conclusion
In summary, our investigation represents a contribution to the study of the welding of dissimilar materials. The microstructures of three different couples of dissimilar materials bonded by solid state welding process were investigated. The three couples consist of copper with aluminium alloy, low carbon steel with duplex stainless steel, and aluminium with alumina which were welded by diffusion bonding process at 550, 1100, and 600 ⁰C respectively.
The main conclusions are as follows:
a. Solid state diffusion welding technique can be successfully applied to joining dissimilar materials.
b. Interface in welded joint has been formed between the dissimilar materials.
c. Thin intermetallic layers were formed between copper.
Acknowledgment
The authors would like to thank WBI and DGRSDT of Algeria for their financial support. We also thank the entire team of the metallurgy department at Mons University for their technical support.
References
- Jin JY, Hong SI (2014) Effect of heat treatment on tensile deformation characteristics and properties of Al3003/STS439 clad composite. Mater Sci Eng A 596: 1-8.
- Cam G, Kocak M (1998) Progress in joining of advanced materials. Int Mat Rev 43(1): 1-44.
- Kazakov NF (1985) Diffusion bonding of materials. Mir Publishers, Moscow, Russia.
- (2011) Welding fundamentals and processes. Materials Park, ASM International, Handbook Committee, Ohio, USA, pp. 682-689.
- Stephenson DJ, David J (1991) Applied Science, Elsevier, London.
- Islam MF, Ridley N (1998) Isostatic diffusion bonding of a microduplex stainless steel. Scripta Mater 38(8): 1187-1193.
- Kokawa H, Tsuzuki T, Kuwana T(1995) Effect of initial microstructure of intermediate material on superplastic diffusion bonding of duplex stainless steel. Iron Steel Inst Jpn Int 35(10): 1291-1297.
- Tong QY, Gosele U (1999) Semiconductor wafer bonding. John Wiley & Sons, New York, USA.
- Gietzelt T, Toth V, Hüll A, Messerschmidt F, Dittmeyer R (2014) Systematic investigation of the diffusion welding behavior of the austenitic stainless steel 304 (1.4301). Advanced Engineering Materials 16(11): 1381-1390.
- Masahashi N, Kondo N, Hanada S (2002) Ann Chim Sci Mat 27: S231.
- Bergmann JP, Petzoldt F, Schürer R, Schneider S (2013) Solid-state welding of aluminium to copper-case studies. Weld World 57: 541-550.
© 2021 Boumerzoug Z. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






