- Submissions

Full Text
Aspects in Mining & Mineral Science
Crystal Plane Anisotropy and the Collection Strengthening of Spodumene
Ning Sun, Haoran Chu, Yang Hu, Yuhua Wang*, Dongfang Lu and Xiayu Zheng
School of Minerals Processing & Bioengineering, Central South University, China
*Corresponding author: Yuhua Wang, School of Minerals Processing & Bioengineering, Central South University, Changsha 410083, China
Submission: July 12, 2021;Published: August 05, 2021

ISSN 2578-0255Volume7 Issue2
Abstract
To improve the separation efficiency of spodumene from gangue minerals, the strengthening of the collection of spodumene and the enlarging of the floatability difference between spodumene and gangue minerals are very important. Spodumene is a silicate mineral with chain structure, and its cleavage planes have obvious anisotropy, which affects the adsorption behavior of collector on each crystal plane. Oleate ions mainly adsorbed on the (110) and (100) planes of spodumene, while the combined collector of oleic acid and dodecyl amine promoted the adsorption of collector on the (001) and (010) planes without reducing the adsorption of collector on the (110) and (100) planes of spodumene, and then it strengthens the flotation collection of spodumene.
Keywords: Spodumene; Crystal plane anisotropic; Anionic collector; Anionic/Cationic Collector
Introduction
Lithium was acclaimed as the energy metal for world progress, and it is widely used in pharmaceutical, ceramic, battery, aerospace, and nuclear energy industries. Pegmatite type spodumene ore is one of the important sources for lithium extraction. Flotation is the most common technology for the separation of spodumene from the ores. Because the associated minerals in spodumene ore are feldspar, quartz and beryl, which have similar surface chemical properties as spodumene, so it makes the difficult to separate spodumene from other silicate minerals. Improving the separation efficiency of spodumene is a key issue to realize the efficient utilization and sustainable development of pegmatite lithium resources. It has been found that there is anisotropy of spodumene cleavage planes, and the adsorption behavior of collectors on the different cleavage planes is also different. Strengthening the adsorption of collectors on the different crystal planes of spodumene is an important way to strengthen the flotation of spodumene [1-14]. Based on these research, the crystal plane anisotropy of spodumene and the adsorption behavior of different collectors on crystal planes of spodumene were analyzed and summarized here, in order to provide a new idea for the follow-up research of spodumene flotation.
Crystal Plane Anisotropy of Spodumene
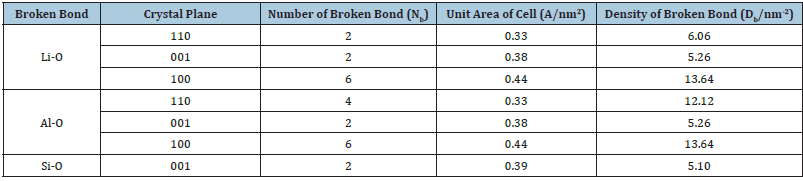
The common exposed cleavage planes of spodumene after the crushing and grinding are (110), (001) and (100), which will lead to the unique surface properties of spodumene. The Al in spodumene is located in the center of octahedron formed by six coordinated o, so the electrostatic strength of each Al-O bond is equivalent to 1/2 valence. As shown in (Figure 1 & Table 1), two Al-O bonds are broken for each aluminum on (110) plane, one Al-O bond is broken for each aluminum on (001) plane, and three Al-O bonds are broken for each aluminum on (100) plane [7]. (a)-(110), (b)-(001) and (c)-(100). (Color: red-O, white-h, pink Al, lavender Li and yellow Si).
Figure 1: Schematic diagram of bond breaking in different crystal planes of spodumene [7].

Table 1: Broken bond number of different crystal planes of spodumene [9].
Adsorption of anionic collector on crystal plane of spodumene
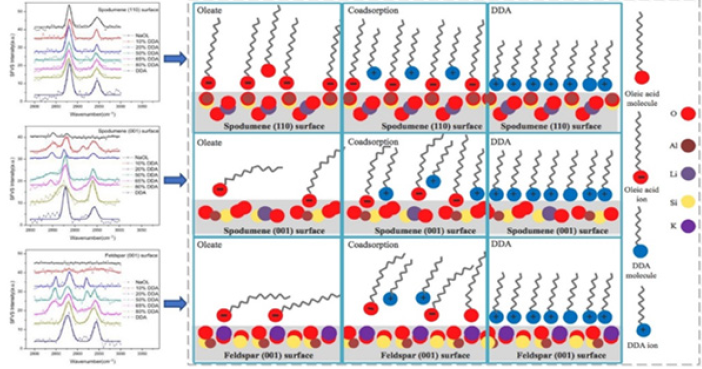
Different broken bond number of Al-O and Si-O bond on different cleavage planes of spodumene result in the adsorption anisotropy of flotation reagent on different cleavage planes in flotation process. As shown in Figure 2, when sodium oleate (NaOl) is used as collector, sodium oleate has two carboxyl oxygen atoms with -1/2 electrostatic charge, while on the spodumene (110) surface there are two aluminum atoms with +1/2 electrostatic charge that do not meet the coordination valance. (110) and (100) planes are the ideal plane for NaOl chemical adsorption, but there are weak adsorption of sodium oleate on (001) plane [7]. Therefore, it is undoubtedly one of the important ways to improve the flotation of spodumene by regulating the exposure ratio of (110) plane in the process of spodumene crushing and grinding [1,3,6].
Figure 2: Adsorption model of oleic acid on different cleavage planes of spodumene [9].

Adsorption of anionic/cationic collector on spodumene crystal plane
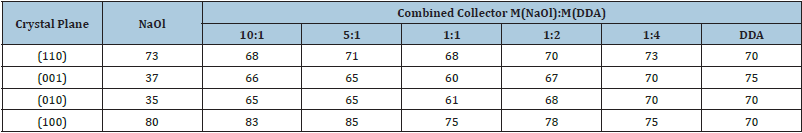
It has been proved that the combined collector can improve the collecting capacity and selectivity based on the synergism of different collectors, so the combined collector has been widely used in flotation practice. The combination of anionic/cationic collector has shown better collection capacity and selectivity for spodumene flotation and has become a new type of collector with the most application potential in spodumene ore flotation, such as NaOl/DDA [11], NaOl/DTAC [8,10], NaOl/DS [12] and sodium oxide paraffin salt/DDA [13]. The adsorption capacity of collector on spodumene is larger than that of oleic acid using alone instead of the combination collector of oleci acid and dodecyl amine. As shown in Table 2, the adsorption of sodium oleate on the spodumene (110) and (100) planes is mainly observed when sodium oleate is used as collector alone, and the contact angles are significantly higher than that on (001) and (010) planes. When sodium oleate and dodecyl amine were used as the combined collector, the contact angles of spodumene (110) and (100) planes changed slightly, but the adsorption of collector on (001) and (010) planes was significantly increased which results in the significant increase of the contact angles.
Table 2: Contact angles at spodumene crystal planes [5].
The same result was also found that oleate adsorbs on (110) plane of spodumene as a dense monolayer in the absence of DDA while its adsorption on (001) plane is weak [6]. As shown in Figure 3, a small amount of DDA can significantly promote the adsorption of collector on (001) plane but has little effect on the adsorption of collector on (110) plane of spodumene. This is an important reason why the combined collector of oleic acid and dodecyl amine can further strengthen the flotation of spodumene [14].
Figure 3: Grade/recovery and gangue rejection ratios versus the corresponding pH and collector concentration for collector 2 (top), collector 5 (middle), and the average of the collector 8 runs (bottom).
Conclusion
Spodumene, quartz and feldspar are all silicate minerals and have the similar surface properties, it is difficult to separate spodumene from gangue minerals by flotation. After the crushing, the main cleavage planes of spodumene are (110), (001), (010) and (100), which show differences in physical and chemical properties. It leads to the different adsorption behavior of collectors on different cleavage planes, the more adsorption of sodium oleate on (110) and (100) plane, but the less on (001) and (010) planes, which leads to the limitation of the collector capacity for sodium oleate. However, the combined collector of oleic acid and dodecyl amine can further strengthen the collector adsorption on (001) and (010) planes without affecting the adsorption of collector on (110) and (100) planes. In a result, the adsorption of collector on spodumene was strengthened and the effect of crystal plane anisotropy was also eliminated.
Acknowledgment
The authors gratefully thank the financial supports by the Natural Science Foundation Project of China (No. 51674290).
References
- Moon KS, Fuerstenau DW (2003) Surface crystal chemistry in selective flotation of spodumene (LiAl[SiO3]2) from other aluminosilicates. International Journal of Mineral Processing 72(1-4): 11-24.
- Yu F (2014) Theory and practice of spodumene ore flotation using mixed anionic and cationic collectors. Central South University, China.
- Zhu G, Wang Y, Liu X, Yu F, Lu D (2015) The cleavage and surface properties of wet and dry ground spodumene and their flotation behavior. Applied Surface Science 357: 333-339.
- Zhu G, Wang Y, Wang X, Miller JD, Lu D, et al. (2019) Effects of grinding environment and lattice impurities on spodumene flotation. Transactions of Nonferrous Metals Society of China 29(7): 1527-1537.
- Zhu G (2019) The selective surface treatment and interface interaction in spodumene flotation process. Central South University, China.
- Xu L, Peng T, Tian J, Lu Z, Hu Y, et al. (2017) Anisotropic surface physicochemical properties of spodumene and albite crystals: Implications for flotation separation. Applied Surface Science 426: 1005-1022.
- Xu L, Hu Y, Wu H, Tian J, Liu J, et al. (2016) Surface crystal chemistry of spodumene with different size fractions and implications for flotation. Separation and Purification Technology 169: 33-42.
- Xu L, Hu Y, Tian J, Wu H, Yang Y, et al. (2016) Selective flotation separation of spodumene from feldspar using new mixed anionic/cationic collectors. Minerals Engineering 89: 84-92.
- Tian J (2018) Fundamental study on enhanced flotation separation of pegmatite spodumene ore. Southwest University of Science and Technology, China.
- Tian J, Xu L, Deng W, Jiang H, Gao Z, et al. (2017) Adsorption mechanism of new mixed anionic/cationic collectors in a spodumene-feldspar flotation system. Chemical Engineering Science 164: 99-107.
- Wang Y, Zhu G, Yu F, Lu D, Wang L, et al. (2018) Improving spodumene flotation using a mixed cationic and anionic collector. Physicochemical Problems of Mineral Processing 54(2): 567-577.
- Xu L, Jiao F, Jia W, Pan Z, Hu C, et al. (2020) Selective flotation separation of spodumene from feldspar using mixed anionic/nonionic collector. Colloids and Surfaces A: Physicochemical and Engineering Aspects 594: 124605.
- Wu H, Tian J, Xu L, Fang S, Zhang Z, et al. (2018) Flotation and adsorption of a new mixed anionic/cationic collector in the spodumene-feldspar system. Minerals Engineering 127: 42-47.
- Zhu G, Wang Y, Wang X, Yu F, Miller JD (2018) States of coadsorption for oleate and dodecylamine at selected spodumene surfaces. Colloids and Surfaces A: Physicochemical and Engineering Aspects 558: 313-321.
© 2021 Yuhua Wang. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






