- Submissions

Full Text
Annals of Chemical Science Research
Overview of Recent Analytical Development and Commonly Used Analytical Techniques in Pharmaceutical Cleaning Verification: A Review
Apu Sarwar1,2, Conor McSweeney1 and Eric Moore2*
1Pfizer Ireland Pharmaceuticals, Ringaskiddy, Co Cork, Ireland
2University College Cork, Cork, Ireland
*Corresponding author: Eric Moore, Senior Lecturer, School of Chemistry, University College Cork, Cork Ireland
Submission: March 16, 2021;Published: April 06, 2021

Volume2 Issue3April, 2021
Abstract
The pharmaceutical cleaning verification can be a challenging process due to the technicality of the analytical process and the hazardous nature of the contaminant on the surface of the manufacturing equipment. There are a number of parameters to be considered before the analytical method is selected for the cleaning verification. The most commonly used methods fall under two main categories: direct and indirect method. The direct method is where the cleanliness is verified directly by visual inspection or any other means where the contaminant is not removed from the surface of the manufacturing equipment. The principle of indirect methods involves the quantitative removal and analysis of the contaminant chemical from the surface of the equipment. This article provides an overview of the current analytical process used by the pharmaceutical industry and the trend of the future pharmaceutical cleaning verification process.
Keywords: Real-time cleaning verification; FTIR for cleaning verification; Cleaning verification; Direct and indirect cleaning; Future of cleaning verification
Introduction
From both the Regulatory and Pharmaceutical industry compliance perspectives, cleaning verification is deemed as an obligatory activity to ensure that the products manufactured on the pharmaceutical equipment are free from cross-contamination, thus ensuring patient safety and product quality [1]. Prior to performing the cleaning verification study, the drug residue is removed from the production equipment by following the pre-defined and validated cleaning procedure that may include acidic, basic, detergent-based cleaners or solvents, which may be a manual or an automated process. The cleaning verification study confirms the effectiveness of the cleaning procedure during the product changeover and provides a high degree of satisfaction that the target molecule from the previous batch is below the Residue Acceptance Limit (RAL) or completely removed from the equipment [2].
The RAL is established to ensure the safety of the patient, and the RAL is established based on the toxicological and pharmacological data, dose strengths, size of manufacturing batch, equipment size, number of dosages per batch, smallest strength manufactured, and product contact surface area [3]. Previous reports addressed the problem of analyzing the low RAL molecule as it is difficult to recover the targeted molecule from the equipment surface. Studies also reported that the analytical procedure for the highly potent compounds is often slow and required advanced analytical skills in order to produce consistent results [4]. Although the pharmaceutical industry follows a validated cleaning procedure to clean the manufacturing equipment, cleaning verification during the changeover is still a requirement by the regulatory authorities in order to ensure that the cleaning procedure is effective as intended [5].
The regulatory expectation for cleaning
In the late 1980s and early 1990s, the US Food and Drug Administration (FDA) and other regulatory agencies began to consider cleaning of the pharmaceutical manufacturing equipment as a manufacturing process, and, as such, it is required to be “validated” in a way comparable to process validation. Many companies started to focus on the validation of cleaning procedures but not on the process of cleaning itself. In many instances, organizations were validating cleaning procedures as they existed without interrogating whether the cleaning procedure is most effective or whether the proper cleaning agents or detergents were being used. Subsequently, the cleaning procedures that were validated may not have been the best choice for a given situation, as stated by Andrew [6]. Therefore, the cleaning verification was introduced by the regulations in order to ensure that the validated cleaning procedure is performed as intended.
Aaron Urbas and Robert Lodder [7] noted an increasing trend observed by the FDA that product manufacturing-related problems leading to product recalls and manufacturing disruption due to cross-contamination [7]. Therefore, the regulatory authority is now placing greater emphasis on equipment cleaning at product changeover due to the higher number of citations related to the cleanliness of the production equipment [8]. Jenkins [9] stated the increase in scrutiny from the regulatory authorities putting pressure on the pharmaceutical manufacturing companies to understand their cleaning validation and verification process better, as an ineffective cleaning process could lead to adulated products and decrease the process efficiency. The risk of crosscontamination is greater in a multi-product facility where the same equipment is used to manufacture more than one type of Active Pharmaceutical Ingredient (API).
The regulatory authorities expect to have a written procedure with detailed information on the cleaning verification and validation process. There is an expectation that the manufacturing plant will have a cleaning process/risk assessment to address equipment cleanliness between batches of the same product and for product changeover; regulators also expect the manufacturing sites to have written procedures for water-soluble residues and nonwater- soluble residue; as well as a written record for the person responsible for the cleaning and their training records. According to the FDA published document entitled ‘Guide to inspection of cleaning process’, the FDA also expects to have detailed written validation and verification protocols in advance for the studies to be performed on each manufacturing system or on a piece of equipment that should address the sampling plan and the analytical method. FDA also expects firms to have clearly defined acceptance criteria for the validation study.
Cleaning verification in the pharmaceutical industry
There are many analytical practices available in cleaning verification; choosing the appropriate analytical tool and technique depends on multiple factors. The most imperative aspect is to govern the specifications or chemicals to be measured. In most cases, the cleaning carryover limit is already established for the existing product. If the RAL is not well-known, the RAL must always be estimated prior to the selection of the investigative tool. In order to use an analytical method for cleaning verification in a GMP-regulated manufacturing facility, the analytical method must meet the requirements set by the regulator, such as the test method should be capable of detecting the targeted molecule or substances at the levels of RAL. The method should be able to detect the targeted molecule in the presence of other molecules such as excipient at the visual level. Forsyth [10] stated that the ability of the sampling method to collect and retrieve API from a surface is also known as the recovery study, which must be performed for a cleaning verification method. The recovery must be within the allowed range or within a pre-defined range.
One of the key criteria for a successful cleaning verification study is the sampling technique. The FDA outlined two sampling methods, direct and indirect. Each of these methods has advantages and disadvantages, but in general, the direct sampling method is preferred. Sandeep [11] indicates that the direct sampling method such as swabbing would work particularly well in more restricted and hard-to-clean areas such as isolators, hoods, and difficult to access areas of equipment. Therefore, the pharmaceutical industry carefully assesses the sampling and analytical technique prior to implementing an analytical procedure based on the following factors [12-14].
A. Objective, rapid, reliable, and reproducible
B. Processing time
C. The limit of detection below the RAL
D. No Interferences
E. Capable of capturing contaminants
F. Compatibility of the analytical technique and extraction solution
G. Process Robustness and type of sample
H. Manufacturing Infrastructure, production equipment, and operating costs
I. Analytical skills and training required
There are a large number of analytical techniques used for cleaning verification in the pharmaceutical industry. These range from visual inspection to using sophisticated technology depending on the physical and chemical nature of the target molecule and its toxicology index. The technique is selected based on the physicochemical properties of the selected molecule, RAL, and the Acceptable Daily Exposure [15]. The analytical techniques currently used by the Pharmaceutical industry for cleaning verification have advantages and disadvantages related to the assessment aspects listed above. But these methods are highly inefficient, and lots of time and effort are required to perform the analytical study. It is a necessity for the pharmaceutical industry to have a direct measurement technique that will allow interrogation of the equipment surface without sampling to generate surface residue results in near real-time. The goal of this article is to outline the past, present, and future (Figure 1) of the cleaning verification in a comprehensive manner so the industry can benefit by choosing the appropriate analytical technique for their cleaning study. Each method will not be described in detail as that is beyond the scope of this article; however, a brief overview of the future trend of a direct measurement technique that has enormous potential to eliminate sampling for cleaning verification will be outlined.
Figure 1:A, B and C Represent past, present and future the cleaning verification practice of pharmaceutical of manufacturing, respectively.
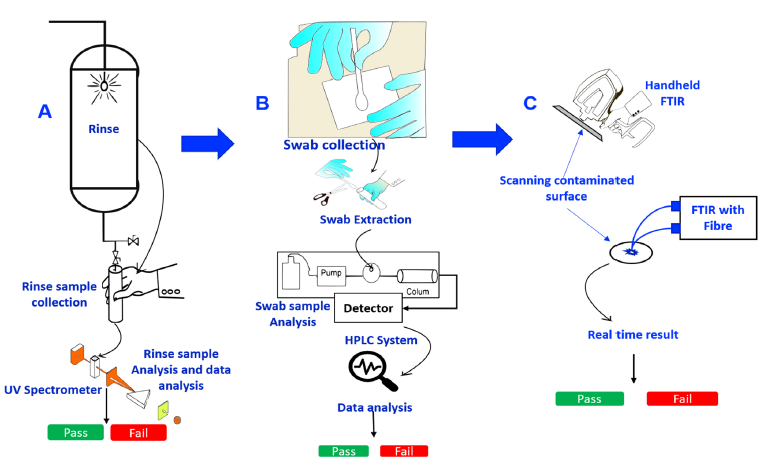
Pharmaceutical cleaning verification and visual inspection
In the pharmaceutical industry, the methods for quantifying the degree of surface residue during the product changeover can be branded as direct or indirect. The direct method refers to the techniques that require samples to be collected or measure directly from the surface; the indirect method, on the other hand, is referred to the technique where sampling or analysis is performed from a secondary source other than the surface. The analytical technique can be categorized as either specific or non-specific depending on the ability of the analytical instrument. For the specific method, only the target molecule is analyzed, while the non-specific method measures every chemical present in the sample. These methods could range from visual inspection to sophisticated instruments that are capable of detecting a very low amount of residues which is not possible to detect with the naked eye.
Visual inspection is one of the key techniques used in the pharmaceutical industry for cleaning verification. Visual inspection is a direct measurement technique for cleaning verification, and no sampling or direct analysis is performed unless the visual inspection is passed according to pre-defined criteria. The visible residue limit (VRL) can be used to monitor equipment cleanliness down to the part per million (PPM) or Part Per Billion (PPB) level [16,17]. The VRL is the contamination level below which the chemical of interest is not visible to the equipment inspector or analyst. Sometimes, multiple inspectors are required to reduce the subjectivity of the observers and improve precision. Sometimes, the visual inspection may be sufficient where the RAL is very high or above the VRL, and any residue left on the surface would be visible.
Visual inspection is also intended to make observations directly from the equipment surface rapidly. However, the pharmaceutical industry cannot solely rely on the visual inspection as residue may not be visible to the naked eye due to the lighting of the manufacturing area, and the reflection from the stainless-steel surface may cause an illusion of a clean surface due to the light angle. In addition, the visual inspection is typically performed at the hardto- clean locations; therefore, it may not be possible or extremely difficult to perform the visual inspection in these areas. Subjectivity always will be the primary concern with a visual inspection [18].
Sampling Techniques for Cleaning Verification
Surface sampling is required when the residue cannot be measured using a direct measurement technique (such as visual inspection or other means that does not require an extracted residue from the surface) that is capable of quantifying the surface residue without sampling at or below the RAL. There are a number of sampling techniques used for pharmaceutical cleaning verification, but the most commonly used techniques are swab and rinse techniques [19,20]. Other techniques such as coupon sampling which, involve the use of a stainless-steel coupon/plate or an actual removable piece of equipment that is dipped into purified water to extract chemical residues for analysis. In this method, it must be ensured that the coupons or plate utilized are equivalent to the surface under test. The placebo sampling method is utilized in the pharmaceutical and biopharmaceutical industry, which involves manufacturing a batch of a selected product without the active ingredient, and the Placebo is tested for any contamination from the previous batch. However, these methods are rarely used by the pharmaceutical industry for cleaning verification.
Swab Sampling
Swab sampling involves rubbing a pre-defined surface area of manufacturing equipment using a swab that has been humidified with a selected solvent which is capable of extracting the target residue from the surface. The extracted sample is then analyzed using an alternative analytical technique such as HPLC, GC, UV, IMS, MS, etc., which is used to analyze the extracted sample [21]. Usually, the surface is carefully wiped according to the cleaning verification protocol. Generally, before the swab collection, visual inspection is performed, and no swab is collected if the residue is visible. Once the swab is collected, the swab is transferred into a known amount of solvent, which is known as the recovery solvent. The recovery levels depend on the solubility of the target molecules in the swabbing solvent, the swabbing procedure, and the physical nature and roughness of the surface. Preferably, the recovery should be as close as possible to 100% but, in the current swabbing method, any value greater than 70% is considered reasonable. In practice, the swab recovery could be as low as 50% of the actual value [22]. In addition, the recovery varies from operator to operator, which is one of the weaknesses of the swab technique when it comes to recovery and reproducibility. The time required to collect the swab and subsequent analysis could be up to a month, depending on the nature of the analysis and the lab resources.
Lower swab recovery values are generally considered unacceptable and require improved procedures. However, if no better alternative procedure is available, a recovery factor is built into the final calculation, which allows the analytical procedure to use for cleaning verification, but these pose a risk of false-positive results that require additional cleaning [23,24]. One benefit of the swab technique is that low soluble or insoluble molecules on the equipment surface are more easily removed by physically rubbing the equipment surface as compared with rinsing. This method also permits collected swabs from the hard-to-clean location of the equipment.
On the other hand, the surface residue is not uniformly distributed throughout the surface; therefore, sampling of a small area of large manufacturing equipment may not be illustrative of the entire production equipment. Other issues that need to be considered include the physicochemical characteristics of the swab’s materials and recovery levels. The swab material should not leach amalgams, such as adhesive used in the swab itself, that can affect the analytical measurement. Some researchers suggested that the swab recovery level can be enhanced significantly using swabs with two different extraction solvents, such as swabbing with organic solvent after swabbing with the aqueous solution, with the results from both swabs being combined [19,25,26].
Rinse sampling
Kohli [26] stated in his paper titled ‘Methods for Monitoring and Measuring Cleanliness of Surfaces,’ a sample of the solution is collected from the final rinse cycle of the cleaning process and subsequently analyzed for the target molecule and the recovery level calculated from the amount of the solvent and the contact surface area. This method is generally used to prepare a sample for the determination of the Nonvolatile Residue (NVR) level by the gravimetric method [27]. There are some concerns with rinse sampling; it is usually presumed that all the target contaminant is efficiently extracted or removed into the rinsing solution and that every corner of the equipment is equally cleaned. Since the surface residues are not quantified directly, there is the leeway that an unsatisfactorily high number of residues may be left on some areas, particularly in the areas with multifaceted geometries and hard-toreach locations. The solvent selection can also affect the cleanliness verification results significantly. A very poor solvent selection will result in large amounts of residues left on the equipment surface.
Kohli [26] also expressed concern about a further risk of rinse sampling, as an inappropriate application of a validated cleaning procedure could result in a false positive that the equipment is clean. A rinse sample collected from such a scenario could lead to disastrous consequences as the actual amount of contamination may remain undetected or an anomaly is detected. Even with all these concerns, rinse sampling does offer several compensations if executed appropriately. It is easy to accumulate a part of the last rinse solution out of the production equipment, and it permits the assessment of the target residue from the entire surface that is problematic to reach for a swab. This benefit makes rinse sampling appropriate for sealed systems, clean-in-place (CIP) systems, or very large-scale production equipment that is hard or impossible to disassemble or to be moved into a cleaning facility; instead, the manufacturing equipment is washed and verified for cleanliness where it is assembled [28]. The rinse analysis could be performed in real-time using an online UV-visible spectrometer, but the drawback of the method is that the sample still must be extracted from the surface, and the compound must be UV active. Another drawback of the method is that the method is highly non-specific.
Current Surface Residue Analysis Methods
At present most of the cleaning verification techniques used in the drug manufacturing facilities are indirect, which requires the residue to be extracted prior to the analysis using existing analytical technology. The section below provides a high-level summary of the most commonly used analytical technique in the GMP-regulated pharmaceutical industry.
Total Organic Carbon (TOC)
Total Organic Carbon (TOC) analysis is a well-established testing method for cleaning verification in the pharmaceutical and biopharmaceutical industry [29-34] and is mostly used for rinse sample analysis. TOC is a highly non-specific method of analysis, all organic carbon present in the sample is detected. Therefore, any detected residue or chemicals is considered the chemical of interest. If the residue of interest is not water-soluble, it can limit the effective linear range of the residue assay. The sensitivity of the TOC method could be down to the ppm or ppb level, and method development for TOC is required less compared to other analytical methods such as HPLC [35].
Liquid chromatography
The use of High-Performance Liquid Chromatography (HPLC) for the testing of cleaning verification samples is well established [22,36] and can address most of the parameters. HPLC is a liquid chromatography technique that involves a liquid sample separates from the other components within the sample while the sample solution passes through a packed column. An HPLC method is capable of separating the residue of interest from other chemical component presents in the sample as well as the detergent in a single HPLC run.
Typically, HPLC is a reliable method when it is required to detect and quantify the low amount of targeted compounds in a surface residue. HPLC is a highly selective method and able to measure low levels of surface residue with great accuracy. In most cases, the swab sample (after extraction) is analyzed by HPLC, where specificity and low levels of residue detection are required. However, the sensitivity of HPLC is reliant on the chemical signature of the residue of interest and the detector. A molecule with no UV chromophore requires either a sophisticated detector such as electrochemical, fluorescence, or derivatization to achieve the required level of sensitivity [4]. HPLC is a time-consuming process and could take up to 40minutes per analysis, depending on the molecule and column. In addition, an extensive sample preparation process is required, which could be problematic for the quick turnaround of samples for analysis [37].
Gas Chromatography (GC)
Similar to HPLC, GC has also been used to analyze the cleaning verification samples after the appropriate sample preparation [38]. This analytical technique is corresponding to HPLC with equivalent accuracy, selectivity, and sensitivity. The sample carrier or mobile phase is in a stream of gas that passes through a column for separation. The method development and the method validation parameters and their demonstration are similar to HPLC. Gas chromatography can only be used for the residues that are thermally stable and volatile at the high temperatures generated in the instrument. The GC analysis could take up to 30minutes per analysis. The technique does not require an extensive amount of sample preparation compared to HPLC [39].
Mass spectroscopy
A Mass Spectrometer (MS) is a sophisticated analytical instrument that measures the atomic mass of an analytical sample. There are several types of MS instruments, but the atmospheric ionization models are widely used for cleaning validation and verification. MS detector can be coupled with an HPLC [40], or the samples for analysis can be inserted directly into the MS [41]. The separation of the target molecule occurs through mass selectivity from the components of the solution and the detergent. This analytical method is very sensitive and could detect a molecule of interest down to the ppm or ppb level. An internal standard is required to enhance the recovery range. The internal standard is referred to a chemical compound whose physicochemical characteristics and chemical structures are similar to the target residue of interest, which will interact with MS in a similar manner. If an internal standard is absent, the accuracy can be constrained to concentrations at or near the RAL [42].
Ion chromatography
Ion Chromatography (IC) is commonly used for the analysis of surfactants, inorganic and organic compounds present in the cleaning sample (swan or Rinse sample). Most cleaning detergents used in the pharmaceutical industry contain sodium and/or potassium. IC detection technique of suppressed conductivity is more sensitive to potassium ions than to sodium ions. This analytical technique is very rapid, and the detection level is also very low. Therefore, a low amount of cleaning agents can be detected and or can be quantified by using Ion chromatography technique [43]. HPLC, GC, Ion Chromatography, and MS all require method development and validation before performing the verification study. Method development and validation can vary depending on the molecule of interest and other potential compounds in the sample. Therefore, the analytical method development and validation can range from a few days to several months.
The Future of the Cleaning Verification
The use of Process Analytical Technology (PAT) in pharmaceutical manufacturing is increasing, and the regulatory authorities are also encouraging the use of PAT in the manufacturing process to enhance the process-understanding. In recent years, a number of new technologies were developed for pharmaceutical cleaning, which can be extremely beneficial for pharmaceutical manufacturing. These technologies are novel to the pharmaceutical industry and are still under development. The aim of this section is to provide a holistic overview of this development.
Fluorescence spectroscopy
Luminescence is a form of emission that occurs from electronically active states of any substance or chemicals. There are two types of luminescence, fluorescence and phosphorescence, and this depends on the characteristics of the excited state. “In excited singlet states, the electron in the excited orbital is paired (of opposite spin) to the second electron in the ground state orbital. Consequently, a return to the ground state is spin allowed and occurs rapidly by the emission of a photon. Typical emission rates of fluorescence are 108 s-1, meaning that a typical fluorescence lifetime is around 10 nanoseconds” stated by Lakowicz [43-45].
Fenske et al. and others [46-49] mentioned that the use of Fluorescence spectroscopy is very common in the biomedical field for evaluating dermal-exposure to chemical contaminants, as well as contamination in hospitals and bio-pharmaceutical. Also, this technology is used in surface cleanliness measurements in industrial applications. It is a fast, non-invasive, and non-destructive analytical technique that can be utilized for online or offline contamination identification and quantification analysis [46-49]. The benchtop fluorescence-based spectrometer has been used in the pharmaceutical industry for many years and is an established measurement technique. However, the use of this technology for direct surface measurement is a new concept, and the instrument had to be redesigned to enable reflective measurements.
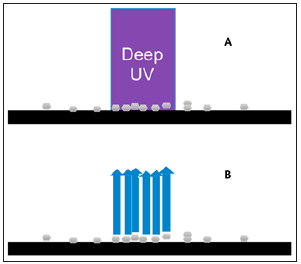
A hand-held cleaning verification instrument was developed for a rapid measurement system that could be used to verify equipment cleanliness. Fluorescence spectroscopy is used to provide the information necessary to measure the concentration of the Active Pharmaceutical Ingredient (API) material or detergent. Using optimum excitation wavelength from a Light Emitting Diode (LED) and detection using multiple emission wavelengths, the hand-held cleaning verification instrument is capable of validating the cleanliness of production equipment. The principle is demonstrated in Figure 2. The data collected will be compared against a calibration curve to quantify the levels of materials (API or detergents) on the equipment surface [50].
Figure 2:(A) Deep-UV florescence illuminates on the surface that contains residual API. (B) the scattered light collection in 180-degree backscatter and analysed by the detector [55].
The fluorescence spectrometer provides the intensity counts according to the Beer-Lambert law, and no chemical signature is produced by the instrument. Therefore, the instrument does not have specificity. It is a known phenomenon that some molecules have fluorescence quenching over time due to chemical and environmental interference, which is a limitation of this technology. As the instrument is fluorescence-based, in the event of fluorescence quenching of the target molecule, the use of this technology will be limited for pharmaceutical cleaning verification [51].
Mid-infrared spectroscopy
FTIR spectroscopy is used in the pharmaceutical industry since the last century, and it is now a well-known analytical method for qualitative and quantitative analysis within the pharma manufacturing process [52,53]. However, the use of the Fourier Transform Infrared spectroscopy (FTIR) for pharmaceutical cleaning verification is new. A measurement system based on FTIR was developed using a specially designed flexible Mid-infrared fiber optic probe in 2001 to perform a quantitative and qualitative evaluation of the residual contamination on the machine surface [54]. This system was developed using a benchtop FTIR which was connected to a flexible fiber-optic probe. The system successfully measured the lubricant contaminant on the machine surface.
Reflective spectroscopy is a well-known technique for collecting FTIR spectra from powder samples or a surface [55]. There are two types of reflective spectroscopy that are widely used in surface analysis a) The grazing angle b) specular reflective spectroscopy. The grazing angle mid-infrared spectroscopy is the most sensitive optical absorption technique for measuring low contaminants on the surface. Due to the higher surface area scanned by the spectrometer, the sensitivity is increased significantly. The specular reflectance FTIR has a very low scan area but with higher penetration depth the specular reflectance interface can measure a greater thickness of the sample. The specular interface is used to quantify paint thickness in the aviation industry [56]. A recent study showed that this type of analytical technique is also usable for surface cleanliness verification.
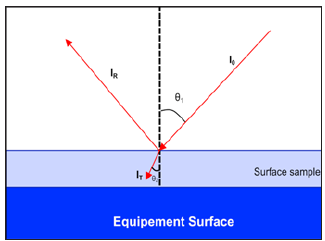
Hernandez-Rivera et al. published the use of a reflective measurement technique for surface contamination detection and demonstrated the detection capability and quantification capability on metals and plastic surfaces [57-60]. A detailed description of the reflection-absorbance spectroscopy is beyond the scope of this article; however, a good description of this topic can be found in the article written by Hoffard [59]. Hofford [59] explained the grazing angle principle., In grazing-angle spectroscopy, the IR beam incidence angle is approximately 800 and is explained by Hoffard [59] in Figure 3. Infrared energy striking a contaminated reflective substrate at a grazing angle of incidence. In specular reflective spectroscopy (Figure 4), the incident bean approaches the surface sample at an angle of approximately 450 or lower, which differentiates it from the grazing-angle. This technique allows measuring relatively thick samples at a near-normal angle of incidence.
Figure 3:Grazing angle spectroscopy used in Rem Spec Spot View FTIR.
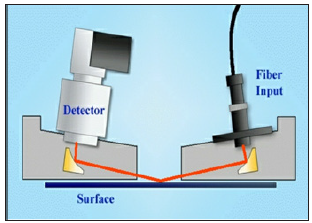
Figure 4: FPrinciple of specular reflectance spectroscopy. the incident FTIR beam represented by I0 illuminates the sample of a given refractive index (n2) and at an angle of incidence (θ1). Some of the incident beams are reflected from the sample surface, represented by IR at the incident angle (θ1).
Cleaning using FTIR to date
In 2008 The Parenteral Drug Association (PDA) carried out a survey which suggests that more than 90 percent of the companies use the swab technique to verify the surface cleanliness and to quantify the active pharmaceutical ingredient (API) residues on the surface of the manufacturing equipment [61]. It is a very common practice in the API manufacturing facilities to rinse the equipment surface and analyze the rinse sample to verify the surface cleanliness. These methods are classified as indirect methods for cleaning verification. The indirect methods are becoming less favorable as some of the API residues may have a very poor recovery, which may increase the chance of producing inaccurate results.
A spectroscopy-based cleaning verification method is able to interrogate the surface directly without any sampling. A spectroscopy-based method can be used in a place where visual inspection is not sufficient, and an analytical means is necessary to ensure the surface is clean with a high degree of confidence [60]. A number of studies have been conducted using FTIR in order to quantify the surface contaminant without any sampling [59,62-65]. Engineering Service Center (NFESC) reports indicating that a grazing angle FTIR is suitable for quantifying the surface contaminant as low 1μg/cm2. This technology is also used for low levels of explosive detection in real-time on a wide range of surfaces, including commercial shipping and passenger luggage [11]. The Mid-IR reflectance measurement technique was first developed by the sheet metal manufacturing industry to detect and quantify the amount of oil and other contaminants on the equipment’s metal, glass, or plastic surfaces [66]. Mid-infrared spectroscopy is capable of measuring the surface residue directly in real-time on the surface sample. The surface contamination result is normally reported in μg/cm2. Any results quoted in this review are expressed as μg/cm2.
Description of the Method
The requirement for the surface contamination analysis is to introduce the FTIR signal on the surface sample and capture the reflected signal using a suitable FTIR detector for processing and quantification [67]. Hamilton [68] demonstrated the principle and the opportunity of using the FTIR for surface cleanliness verification in the pharmaceutical industry in 2007 in her thesis published in University Canterbury [12]. To date, the pharmaceutical industry has not used the technique to release the manufacturing equipment for use in production, although the cleanliness verification of the manufacturing equipment is a very hot topic in the pharmaceutical industry. Pharma manufacturing is a regulated industry; using new technology in GMP regulated manufacturing facilities could be very challenging and has to be extensively demonstrated and validated prior to implementation according to ICH & GMP guidelines. There are a number of key aspects involved in validating the FTIR for cleaning application:
A. Preparation of calibration standard.
B. Data collection and model development and validation.
Preparation of calibration standard
The challenges involved in the preparation of the calibration standard are described by Hamilton in his thesis [12]. Hamilton investigated two sample deposition techniques a) direct deposition using soft edge plastic spatula and b) spraying. The direct deposition technique was extremely non-uniform, and the uniformity is solvent dependent. He experimented with a number of modified direct smearing techniques and stated, “Confidence in the uniformity and consistency of a particular technique is important, and the basic smear technique did not meet these requirements while the modified methods did not result in any significant improvements. The spray technique was able to produce a visually more uniform sample compared to the smear technique. However, the amount of sample deposited by the spray method was unknown, and he stated the following: ‘Since the amount of material deposited by the spray method is difficult to predetermine, an independent method is needed to establish the true loading.”
PolyPico technology Ltd. claims their Piezoelectric based nanodrop device is capable of depositing the known amount of sample uniformly on the surface. However, the instrument measures the individual droplet size using a microscopic camera to calibrate the drop size. An image-based drop size measurement technique may cause some uncertainty, especially when the camera is capturing the droplets of a few nano-litters in size. The accuracy of the measurement is very critical in a GMP-regulated laboratory; also, the software used to calculate the drop size needs to be 21 CFR 11 compliance. Therefore, the PolyPico deposition system may not be used in a GMP-regulated industry. Similarly, the SPT lab-tech has developed a microdot printer that is capable of printing samples on the surface uniformly, but the quantification of the deposited amount is calculated by using a windows-based software which is not 21 CFR 11 compliance. Therefore, many pharmaceutical companies will be very cautious about using these technologies in a GMP-regulated manufacturing facility.
The chemical printer was developed by Photon system Inc in a joint collaboration effort with Pfizer, which is capable of printing samples on multiple surfaces uniformly (Figure 5) and the total amount of sample deposited can be verified visually using the NIST certified syringe reader provided with the Cham Cal. This printer does not use software to verify the deposition volume, and it is in compliance with GMP regulation [55]. This technology can be very useful in validating the FTIR according to the ICH guidelines.
Figure 5: (A) Sample on stainless steel surface and Surface heat map displaying the non-uniform distribution of the sample on the surface when sample was deposited using a microliter syringe. (B) Sample on stainless steel surface and Surface heat map showing the near-uniform distribution of the sample when the Chem-Cal was used to deposit the sample.

Data collection
FTIR spectroscopy requires a blank background, and the best way to collect a background is to use similar material as the manufacturing equipment or targeted surface. The consistency in the background, as well as the roughness of the sampling surface, should be considered. Hamilton et al. [68] and Micheal et al. have investigated the influence of varying surface roughness on the spectra collected using FTIR. Their overall finding is that increased surface roughness tends to degrade the model quality; in particular, there is a high degree of error associated with the predicted value [11,68-72]. For qualitative information where the risk of contamination is low, the FTIR can be used as an addition to the visual inspection. A semi-quantitative method based on the comparative peak area or height of the related peak has been used in the pharmaceutical setting. Where a fully quantitative method is required, it is suggested in several studies performed by Hamilton et al. [68] that the chemometric-based multivariate model should be used to encounter the variability for the method development [68].
Hamilton also showed that if the contamination level is 1μg/cm2 or higher, a simple method using peak height was able to produce the correct result. However, they found that at lower concentrations, the calibration model was nonlinear, and the interference from the atmospheric water was affecting the applicability of the univariate model. The surface reflective FTIR is an open path technique; therefore, the atmospheric CO2 may also affect the spectra collected.
Advantages and Disadvantages
The FTIR-based analytical technique is significantly faster to produce an analytical result when compared to the swab or rinse technique. According to Figure 6, the result can be achieved in near real-time. The specular reflectance FTIR is also very accurate, and the technology is capable of predicting ±15% of the actual concentration [72]. The FTIR-based cleaning verification technique allows the pharmaceutical industry to bring the laboratory analysis to the production floor. Furthermore, no advanced knowledge in analytical chemistry is required as all the data analysis is performed by the instrument software. A portable or hand-held FTIR with a specular reflectance interface can be very beneficial for usability perspective as well as to measure the manufacturing equipment directly on the production floor. The FTIR systems evaluated so far are too big for a lot of the manufacturing equipment. Therefore, many hard-to-clean locations in small manufacturing equipment may not be accessible by the instruments. The technology is at its early development; therefore, any regulatory challenges must be overcome in order to use the technology in a GMP-regulated manufacturing facility. In addition, the FTIR system evaluated to date is not capable of quantifying a molecule with a RAL of less than 1μg/cm.
Figure 6: The figure on left shows the current cleaning verification process which could takes more than 52 working hours. The figure on right shows the process proposed by Sarwar et al. [72] for real time analysis.

Conclusion
The current cleaning verification method is capable of verifying the vessel cleanliness; however, due to the lack of efficiency and accuracy, the pharmaceutical industry is constantly searching for an efficient way to measure surface cleanliness. The accuracy and repeatability of the analytical method for cleaning verification are very important as the regulators are enhancing focus on the cleaning verification more due to the increasing number of product contaminations and recalls. Efficiency is also key from the pharmaceutical manufacturer’s perspective as the demand for medicine is ever-increasing, and there is a greater need for more rapid equipment changeovers. FTIR with either the grazing angle or specular reflectance mode could be an extremely useful tool for surface cleanliness measurement. A significant amount of work has been done to establish a full range of qualitative, semi-quantitative, and fully quantitative applications of FTIR in pharmaceutical cleaning verification. Further research to identify possible interference from the surface and other chemical residues is required to use FTIR for cleaning verification in a complex environment such as the pharmaceutical industry.
Acknowledgment
The authors gratefully acknowledge support from Seamus O’Neill (Pfizer) and the Irish Research Council (IRC) for sponsoring the project. Also, the authors would like to thank Dr. Anna Hogan from University College Cork; for reviewing the paper.
References
- World Health Organization (2006) Quality assurance of pharmaceuticals: A compendium of guidelines and related materials. 2 Good manufacturing practices and inspection. (2nd edn).
- (2016) Guidance of aspects of cleaning validation in active pharmaceutical ingredient plants.
- Dubey N, Dubey N, Mandhanya M, Jain DK (2012) Cleaning level acceptance criteria and HPLC-DAD method validation for the determination of Nabumetone residues on manufacturing equipment using swab sampling. Journal of Pharmaceutical Analysis 2(6): 478-483.
- Yang P, Burson K, Feder D, Macdonald F (2005) Method development of Swab sampling for cleaning validation of a residual active pharmaceutical ingredient. Pharmaceutical Technology.
- (2019) 21- current good manufacturing practice for finished pharmaceuticals.
- Robert L, Aaron U (2018) Pharmaceutical spectroscopic cleaning validation. Science Direct Working Paper No S1574-0331(04)70625-6.
- Mollah H (2008) Cleaning validation for biopharmaceutical manufacturing at Genentech, Inc. Part 2. BioPharm International 21(3): 10-17.
- Jenkins M, Vanderwielen AJ (1994) Cleaning validation: An overall perspective. Pharm Technol 18.
- Forsyth RJ (2016) Best practices for cleaning validation swab recovery studies. Pharmaceutical Technology 40(9): 40-53.
- Kalelker S (2010) Why the swab matters in cleaning validation? In recent years, increased emphasis has been placed on the development of validated and robust cleaning protocols. Controlled environments.
- Hamilton L (2007) Applications of grazing-angle reflection absorption Fourier transform infrared spectroscopy to the analysis of surface contamination. University of Canterbury, New Zealand.
- Mittal KL (1979) Surface contamination: An overview. Plenum Publishing 1: 3-45
- Zurbrügg D (2006) Cleanliness testing and identification of residues on polymer medical devices. JASTM Intl 3(2): 1-9.
- Choudhary A (2017) Sampling in cleaning validation in pharmaceutical industry. Pharmaceutical guidelines 06.
- Forsyth RJ, Nostrand VV (2005) Application of visible residue limit for cleaning validation in a pharmaceutical manufacturing facility. Pharm Technol 29(10): 152-161.
- Forsyth RJ, Hartman J, Nostrand VV (2006) Risk-management assessment of visible-residue limits in cleaning validation. Pharm Technol 30(9): 101-114.
- Forsyth RJ (2014) Qualifying personnel to visually inspect cleaned equipment. Pharm Technol 38(1).
- Kaur S, Bala I, Kamboj A, Jain U (2017) Development and validation of analytical method for estimation of aripiprazole in swab sample on pharmaceutical manufacturing equipment surface for cleaning validation. International Journal of Pharmacy and Pharmaceutical Sciences 9(5): 141-151.
- Karukhnishvili RN, Loria K, Dvali N (2015) Validation of swab sampling and HPLC methods for determination of meloxicam residues on pharmaceutical manufacturing equipment surfaces for cleaning validation. Turkish Journal of Pharmaceutical Sciences 12(3): 40-52.
- Raj A (2014) Cleaning validation in pharmaceutical industries. J Atom and molecules 4(4): 779-783.
- Pagan M (2014) Why are your swab recoveries so low? Pharmaceutical Online.
- Forsyth RJ, Haynes D (1998) Cleaning validation in a pharmaceutical research facility. Pharm Technol 22(9): 104-112.
- Forsyth RJ (2007) Materials of construction based on recovery data for cleaning validation. 31(10).
- Chudzik GM (1999) General guide to recovery studies using swab sampling methods for cleaning validation. J Val Technol 5(1): 77-81.
- Valavala S, Seelam N, Tondepu S, Sundaramurthy V (2017) Cleaning method validation for estimation of dipyridamole residue on the surface of drug product manufacturing equipment using swab sampling and by high performance liquid chromatographic technique. Turk J Pharm Sci 17(2): 182-189.
- Kohli R (2012) Chapter 3-Methods for monitoring and measuring cleanliness of surfaces. Developments in Surface Contamination and Cleaning 4.
- Rizoulieres MF, Majoor FA (2014) Cleaning-in-place’, Ch. 10.
- (1993) FDA guide to inspections validation of cleaning processes.
- Jenkins KM, Vanderwielen AJ, Armstrong AJ, Leonard LM, Murphy GP, et al. (1996) Application of total organic carbon analysis to cleaning validation. PDA J Pharm Sci & Tech 50(1): 6-15.
- Strege MA, Stinger TL, Brett TF, Avinash LL (1996) Total organic carbon analysis of swab samples for the cleaning validation of bioprocess fermentation equipment. BioPharm 9(4): 42-45.
- Wallace BN, Stevens R, Purcell M (2004) Implementing total organic carbon analysis for cleaning validation. Pharmaceutical Technology Aseptic Processing 2004(3).
- Wallace BN, Purcell M, Furlong J (2002) Total organic carbon analysis as a precursor to disinfection byproducts in potable water: Oxidation technique considerations. J Environ Monit 4(1): 35- 42.
- Glover C (2006) Validation of the Total Organic Carbon (TOC) swab sampling and test method, PDA. Journal of Pharmaceutical Science and Technology 60(5): 284-290.
- Queralt M, Montoya EG, Lozano PP, Negre JMS, Miñarro M, et al. (2009) Total organic carbon (VCSN and VWP) and HPLC analysis for cleaning validation in a pharmaceutical pilot plant. PDA Journal of Pharmaceutical Science and Technology 63(1): 42-57.
- Schmidt HA (2006) Validated HPLC method for the determination of residues of acetaminophen, caffeine, and codeine phosphate on swabs collected from pharmaceutical manufacturing equipment in support of cleaning validation. J Liq Chrom & Rel Technol 29(11): 1663-1673.
- Boca MA, Apostolides Z, Pretorius E (2005) A validated HPLC method for determining residues of a dual active ingredient anti-malarial drug on manufacturing equipment surfaces. J Pharm & Biomed Anal 37(3): 461-468.
- Czubak L, Krygier A, Małecka BT (2014) Gas chromatography method of cleaning validation process for 2-propanol residue determination in pharmaceutical manufacturing equipment. International Journal of Pharmaceutical Sciences and Drug Research 6(3): 243-245.
- Mirza T, George RC, Bodenmiller JR, Belanich SA (1998) Capillary gas chromatographic assay of residual methenamine hippurate in equipment cleaning validation swabs. Journal of Pharmaceutical and Biomedical Analysis 16(6): 939-950.
- Chevalier F, Sommerer N (2011) Analytical methods | mass spectrometric methods. Encyclopedia of Dairy Sciences (2nd edn), pp. 198-205.
- Carr G (2013) Using tandem LC-MS for cleaning validation. Pharmaceutical Technology 37(8).
- Liu L, Pack BW (2007) Cleaning verification assays for highly potent compounds by high performance liquid chromatography mass spectroscopy: Strategy, validation and longer performance. Journal of Pharmaceutical and biomedical analysis 43(4): 1206-1212.
- Newton B (2008) Ionic contamination and analytical techniques for ionic contaminants, developments in surface contamination and cleaning. Fundamentals and Applied Aspects pp. 653-673.
- Lakowicz JR (2010) Principles of fluorescence spectroscopy (3rd edn).
- Chullipalliyalil K, Lewis L, McAulif MAP (2020) Deep UV laser-induced fluorescence for pharmaceutical cleaning validation. Anal Chem 92: 1447-1454.
- Bublitz J, Prozessanalytik K (2015) Fluorescence spectroscopy: A powerful method for surface analysis. Photonics.com.
- Fenske RA, Leffingwell JT, Spear RC (1986) A video imaging technique for assessing dermal exposure. II. Fluorescent tracer testing. Am Ind Hyg Assoc J 47(12): 771-775.
- Fenske RA (1988a) Correlation of fluorescent tracer measurements of dermal exposure and urinary metabolite excretion during occupational exposure to malathion. Am Ind Hyg Assoc J 49(9): 438-444.
- Fenske RA (1988b) Visual scoring system for fluorescent tracer evaluation of dermal exposure to pesticides. Bull Environ Contam Toxicol 41(5): 727-736.
- Fenske RA (1990) Non-uniform dermal deposition patterns during occupational exposure to pesticides. Arch Environ Contam Toxicol 19: 332-337.
- Franklin CA, Fenske RA, Greenhalgh R, Mathieu L, Denley HV, et al. (1981) Correlation of urinary pesticide metabolite excretion with estimated dermal contact in the course of occupational exposure to Guthion. J Toxicol Environ Health 7(5): 715-731.
- https://photonsystems.com/wp-content/uploads/2017/06/2017-IFPAC-talk_New-Cleaning-Validation-V11.pdf
- Durkee JB (2007) Cleanliness verification: Grazing angle FTIR (GAFTIR) vs. OSEE. Controlled Environments Magazine, pp. 29-30.
- Behrning S, Bley H (2001) A flexible method for detecting and quantifying organic films on sheet metal. In: Dufluo JR, Geiger M, Kals HJJ, Shirvani B, Singh UP (Eds.), Sheet Metal 2001 Proceedings of the Ninth International Conference on Sheet Metal, Katholieke Universiteit Leuven, Belgium, pp. 451-458.
- Thomson MA (2015) Developments in surface contamination and cleaning contamination sources. Measurement, validation, and regulatory aspects. Chapter 2 Mid-IR Spectroscopy as a tool for cleanliness validation. pp. 51-67.
- Kahrim K, Daveri A, Rocchi P, Cesare GD, Cartechini L, et al. The application of in situ mid-FTIR fibre-optic reflectance spectroscopy and GC-MS analysis to monitor and evaluate painting cleaning. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy 74(5): 1182-1188.
- Rivera SPH, London P, Suarez JRC, Pedrozo OMP, Rivera HF (2011) Remote Raman and infrared spectroscopy detection of high explosives. In: Janssen TJ (Ed.), Explosive materials: Classification, composition and properties. Nova Science Publishers, New York, USA, pp. 231-258.
- London LCP, Pedrozo OMP, Rivera SPH (2011) Evaluation of samples and standards of energetic materials on surfaces by grazing angle-FTIR spectroscopy. In: Rees OJ (Ed.), Fourier transform infrared spectroscopy: Developments, techniques and applications. Nova Science Publishers, New York, USA, pp. 159-175.
- London LCP, Santiago A, Pujols J, Pedrozo OMP, Mattei A, et al. Characterization of layers of tetryl, TNB, and HMX on metal surfaces using fiber optics coupled grazing angle-FTIR. In: Andresen BF, Fulop GF, Norton PR (Eds.), Proceedings of SPIE 6542, Infrared Technology and Applications XXXIII, SPIE, Washington, USA.
- Hoffard TA (2003) Grazing-angle fourier transform infrared spectroscopy for surface cleanliness verification. Technical Report TR-2217-SHR, Department of Defense, Naval Facilities Engineering Service Center, Port Hueneme, California.
- Leblanc D (2008) PDA survey results: Cleaning validation sampling practices. PDA Lett 44: 11.
- Forsyth RJ (2010) Do visible residue limits make the 10-ppm carryover limit obsolete? Pharm Technol 34(2): p. 60.
- Chalmers JM, Griffiths PR (Eds.), Handbook of vibrational spectroscopy. John Wiley & Sons, Chichester, UK, pp. 983-998.
- Reddy KB, Teelucksingh N (2005) Quantification of active pharmaceutical ingredients on metal surfaces using a mid-IR grazing-angle fiber optics probe-an in-situ cleaning verification process. Spectroscopy 20: 16.
- Rivera SPH, London LCP, Pedrozo OMP, Ruiz O, Feliciano YMS, et al. (2007) Vibrational spectroscopy of chemical agents’ simulants, degradation products of chemical agents and toxic industrial compounds. Int J High Speed Electron Syst 17(4): 827-843.
- Claybourne M (2002) Mid-infrared external reflection spectroscopy. In: Chalmers JM, Griffiths PR (Eds.), Handbook of vibrational spectroscopy. John Wiley & Sons, Chichester, UK, pp. 969-981.
- Perston BB (2006) Infrared reflection-absorption spectrometry and chemometrics for quantitative analysis of trace pharmaceuticals on surfaces. University of Canterbury, Christchurch, New Zealand.
- Beecroft MT, Szczesniak MM, Smith BJ, Matsumoto JF, Ferguson JP (2007) Hand-held Spectrometer. US Patent 7: 236-243.
- Hamilton ML, Perston BB, Harland PW, Williamson BE, Thomson MA, et al. (2006) Fiber- optic infrared reflection absorption spectroscopy for trace analysis on surfaces of varying roughness: Sodium dodecyl sulfate on stainless steel. Appl Spectrosc 60(5): 516-520.
- Perston BB, Hamilton ML, Harland PW, Thomson MA, Melling PJ, et al. (2008) Fiber-optic infrared reflection absorption spectroscopy for trace analysis on surfaces of varying roughness. Part II: acetaminophen on stainless steel. Appl Spectrosc 62(3): 312-318.
- Perston BB, Hamilton ML, Williamson BE, Harland PW, Thomson MA, et al. (2007) Grazing- angle fiber-optic Fourier transform infrared reflection-absorption spectroscopy for the in situ detection and quantification of two active pharmaceutical ingredients on glass. Anal Chem 79(3): 1231-1236.
- Hamilton ML, Perston BB, Harland PW, Williamson BE, Thomson MA, et al. (2005) Grazing-angle fiber-optic IRRAS for in situ cleaning validation. Org Process Res Dev 9(3): 337-343.
- Sarwar A, McSweeey SC, Temmermans J, Smith M, Moore E (2020) An alternative approach for cleaning verification in pharmaceutical industry. Anlyst.
© 2021 Eric Moore. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






