- Submissions

Full Text
Annals of Chemical Science Research
Inhibiting Effect of Different Carboxylic Acid on Uncatalysed and Co2O3 Catalysed Autoxidation of SO2 in Alkaline Medium- Brief Comparision
Sharma H, Sharma AK and Prasad DSN*
Department of Chemistry, Govt. P.G. College Jhalawar, India
*Corresponding author: Prasad DSN, Department of Chemistry, Govt. P.G. College Jhalawar, India
Submission: March 03, 2020Published: March 16, 2020

Volume2 Issue1January, 2020
Abstract
The results of the different carboxylic acids inhibited Co2O3 catalysed autoxidation of S(IV) in alkaline medium have been shows that the reactions follow the free radical mechanism.
Introduction
The study of air pollution has been the interest area to the scientific community, since the end of the World War II. The earliest perceived problems were those related to the incomplete combustion of coal, soot and ash abounded in the major industrial cities of the world [1-5]. The oxidation of sulfur dioxide has been one of the most frequently studied reactions in aqueous atmospheric droplets. Three reaction pathways are considered to be dominantly responsible for oxidation of SO2 in atmospheric water droplets. These are the oxidation of dissolved SO2 by H2O2, O3 and O2 in the presence of transition metal ions as catalysts [6-10]. Recent studies show that the sulfur (IV) oxidation in atmospheric water droplets can be affected by other reactions. Organic compounds may dissolve into water droplets and react with sulfoxy radicals and transition metal ions, and thus alter the rate of catalytic S(IV) oxidation [11-15]. In most of the studies the role of organics has been reported in the metal ion catalysed autoxidation of sulfur (IV) in aqueous medium [16-17]. Very few studies are available on the role of organics on the metal oxide catalysed autoxidation of sulfur (IV) in aqueous medium. This led us to investigate the kinetics of sulfur (IV) autoxidation catalyzed by Co2O3 in the pH range 7.8-9.4. and the effect of various carboxylic acids have been studied in alkaline media to delineate the nature of the mechanism.
Experimental
The experimental procedure was exactly the same as described earlier [18-20].
Product Analysis
The qualitative tests showed sulfate to be the only oxidation product [21-22].
Results
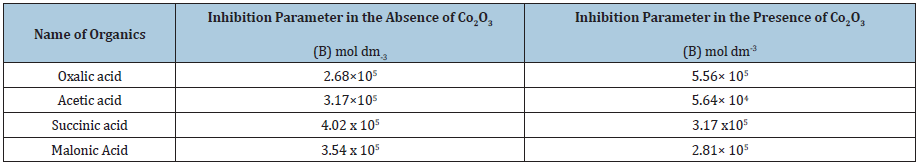
The major aim of the present study was to examine the effect of organic inhibitors on the autoxidation of S(IV) in alkaline medium. For this purpose, oxalic acid, acetic acid, succinic acid, and malonic acid were chosen as the organic inhibitors. On varying the (carboxylic acid) from 1×10-7 to 4×10-3mol L-1, the rate of the reaction become decelerated. The nature of the (S(IV))-dependence in presence of carboxylic acid did not change and remained first order. By plotting a graph between 1/Kinhv/s (carboxylic acid) gives a linear line with non-zero intercept [23-25]. The value of intercept=1/kcat and slope=B/kcat from these values the value of inhibition parameter B can be calculated, inhibition parameter B=slope/intercept (Table 1).
Table 1: Calculated value of B (Inhibition parameter) in absence and presence of Co2O3.
Conclusion
As reported by Gupta et al. [1] a radical mechanism operates in those reactions in which the inhibition parameter lies in the range 103-104. In this study the value of inhibitor parameter is found to be 104-105, which lies in above the range [26,27]. These results strongly support the radical mechanism. In contrast the results conclusively show that (oxalic acid, acetic acid, succinic acid and malonic acid) act as inhibitors for S(IV) autoxidation and the order in uncatalyzed reaction is -
oxalic acid<acetic acid<malonic acid< succinic acid
In case of Co (III) catalysed reaction the order is -
acetic acid<malonic acid< succinic acid<oxalic acid
Hence it is concluded that succinic acid is better inhibitor out of the four organics in uncatalysed reaction, whereas oxalic acid is better inhibitor among the four organics in Co (III) catalysed reaction.
Acknowledgement
The authors pay their sincere gratitude to Principal, Govt. P.G. College, Jhalawar- Rajasthan (India) for providing necessary research facilities to accomplish this study.
References
- Gupta KS, Singh R, Saxena D, Manoj SV, Sharma M (1999) Role of manganese dioxide in the autoxidation of sulfur (IV) in oxic and anoxic suspensions. Ind J Chem 38A: 1129-1138.
- Sharma AK, Sharma R, Prasad DSN (2017) Acid rain chemistry, catalysis and inhibition of SO2 in environment. P Lambert Academic Publishing, Germany.
- Sharma AK, Prasad DSN, Sharma R (2018) Catalysis and inhibition of SO2 in atmospheric environment- a perspective of acid rain chemistry. Analytical techniques in Chemical and biological sciences Discovery publishing house Pvt Ltd, New Delhi, India.
- Sharma AK, Acharya S, Sharma R, Saxena M (2012) Recovery and reuse of SO2 from thermal power plant emission. In: Mukesh K (Ed.), Air Pollution-Monitoring, Modelling, Health and Control. In Tech Open Access Publisher, University campus, Croatia.
- Sharma H, Sharma AK, Kumar M, Prasad DSN (2019) The influence of succinic acid on the kinetics of the atmospheric oxidation of dissolved SO2 Catalysed by Co2O3. International Journal on Emerging Technologies 10(3): 82-86.
- Wilkosz I, Mainka A (2008) Mn (II)-catalysed S(IV) oxidation and its inhibition by acetic acid in acidic aqueous solutions. J Atmos Chem 60: 1-17.
- Ziajka P, Pasiuk-Bronikowska W (2003) Autoxidation of sulfur dioxide in the presence of alcohols under conditions related to tropospheric aqueous phase. Atmos Environ 37(28): 3913-3922.
- Sharma AK, Sharma R, Prasad DSN, Parashar P, Gupta AK (2015) Formic acid inhibited Ag (I) catalysed autoxidation of S(IV) in acidic medium. J Chem Chem Sci 5(12): 760-771.
- Sharma AK, Sharma R, Prasad DSN, Parashar P, Gupta AK (2016) Ag (I) catalyzed autoxidation of S(IV) and its inhibition by isopropyl alcohol in acidic medium. Chem Sci Rev Lett 17(5): 14-23.
- Sharma AK, Sharma R, Prasad DSN (2015) Kinetics and mechanism of uncatalysed and Ag (I) catalysed autoxidation of S(IV) and its inhibition by isoamyl alcohol in acidic aqueous solutions. Int J Mod Sci Eng Technol 2(12): 31-40.
- Sharma AK, Sharma R, Prasad DSN, Parashar P (2016) The inhibitive action of aniline on the autoxidation of sodium sulfite in acidic medium. J Anal Phar Res 17(5): 14-23.
- Sharma AK, Sharma R, Prasad DSN (2017) The effect of atmospheric aromatic amides on the Ag(I) catalyzed S(IV) autoxidation in aqueous solution. The Experiment 40(1): 2354-2363.
- Sharma AK, Sharma R, Prasad DSN, Parashar P (2017) Ag(I) catalyzed oxidation of S(IV) in aqueous solution differing effect of benzoate ions in acidic medium. Curr Phy Chem 7(2): 338-347.
- Sharma AK, Prasad DSN (2017) Influence of pH and organics on autoxidation of S(IV) catalyzed by Ag(I). Recent Adv Petrochem Sci 3(1): 1-2.
- Prasad DSN, Rani A, Madnavat PVS, Bhargava R, Gupta KS (1991) Kinetics of surface catalyzed oxidation of sulfur (IV) by dioxygen in aqueous suspensions of cobalt (II) oxide. J Mol Catal 69(3): 393-405.
- Sharma H, Sharma AK, Prasad DSN (2018) Kinetics and mechanism of oxalic acid inhibited and heterogenous Co2O3 catalysed autoxidation of S(IV) in atmospheric water. J App Chem 7(5): 1442-1449.
- Hussain F, Begam S, Singh J, Sharma AK, Prasad DSN (2018) Kinetics of aniline inhibited uncatalysed and Co2O3 catalysed autoxidation of S(IV) in atmospheric environment. Asian J Chem Env Res 11: 60-67.
- Hussain F, Begam S, Sharma AK, Prasad DSN (2018) Effect of isopropyl alcohol on autoxidation of S(IV) catalyzed by Co2O3 in alkaline medium. Bull Pure appl Sc 37(1): 9-18.
- Hussain F, Begam S, Sharma AK, Prasad DSN (2018) Co2O3 catalyzed oxidation of SO2 in aqueous solution differing effect of benzamide in alkaline medium. Chem Sc Trans 7(4): 600-609.
- Hussain F, Begam S, Sharma AK, Prasad DSN (2018) Kinetics and mechanism of Co2O3 catalysed autoxidation of sulphite and inhibition by sodium benzoate. J Inst Chemists 90(4): 104-119.
- Begam S, Hussain F, Singh J, Sharma AK, Prasad DSN (2018) Kinetics of sodium sulphite oxidation catalyzed by Co2O3 and inhibited by ethylene glycol. Asian J Res Chem 11(3): 610-616.
- Sharma H, Sharma AK, Parashar P, Prasad DSN (2019) Effect of acetic acid on Co2O3 catalyzed autoxidation of aqueous sulphur dioxide in alkaline medium. Sch Acad J Biosci 7(3): 117-124.
- Sharma AK, Sharma R, Prasad DSN (2017) Role of organics in atmospheric catalytic autoxidation of aqueous Sulphur dioxide in acidic medium. Malaysian J Chem 19(1): 1-12.
- Sharma AK, Sharma R, Prasad DSN (2017) Kinetics of isoamyl alcohol and aniline inhibited uncatalysed and Ag (I) catalysed autoxidation of S(IV) in acidic medium. Asian J Res Chem 10(3): 251-258.
- Sharma AK, Sharma R, Prasad DSN (2018) Effect of aliphatic mono carboxylic acids and alcohols on Ag(I) catalyzed oxidation of SO2 in aqueous solution. J Mat Env Sci 9(6): 1829-1837.
- Sharma AK, Prasad DSN (2020) Copper catalyzed autoxidation of Sulphur dioxide and inhibition by methanoic acid. Current Phy chem 10: 33-46.
- Sharma H, Sharma AK, Prasad DSN (2019) Dynamics of malonic acid inhibited uncatalysed and Co2O3 catalysed autoxidation of S(IV) in alkaline medium. Think India Journal 22(14): 7168-7181.
© 2020 Prasad DSN. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






