- Submissions

Full Text
Advancements in Bioequivalence & Bioavailability
Bioequivalence Study of Tadalafil Tablets in Healthy Chinese Volunteers under Fasting Conditions
Guanghui Ren1, Zhiying Qin1, HuitingSong1, Yanan Tan1, Yonghan Wang1, Danni Hao1, YangLu1, Ning Li2, Di Zhao1 and Xijng Chen1*
1 Clinical Pharmacokinetics Laboratory, China Pharmaceutical University, China
2 National Experimental Teaching Demonstration Center of Pharmacy, China Pharmaceutical University, China
*Corresponding author: Xijng Chen, Clinical Pharmacokinetics Laboratory, China Pharmaceutical University, Nanjing, Jiangsu Province 211198, China
Submission: April 12, 2018; Published: June 18, 2018

ISSN 2640-9275Volume1 Issue4
Abstract
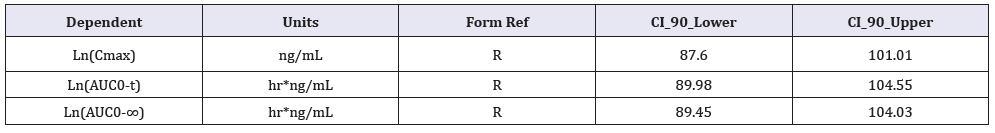
A two way, randomized cross-over bioequivalence study was conducted to comparing the rate and extent of absorption of two formulations of tadalafil tablets after a single dose of 20mg. The study was carried out using healthy male volunteers (N=29) under fasting conditions. A highly sensitive liquid chromatography-tandem mass spectrometry method was developed and validated, which was employed to determine the tadalafil in human plasma. Phoenix Win Nonl in 8.0 was used for pharmacokinetic analysis and bioequivalence between the two Tadalafil Tablets was determined by calculating 90% confidence intervals (90% C.I.) for the ratio of Cmax, AUC0-t and AUC0-∞ values for the test and reference products. The 90% confidence intervals for the ratio of Cmax (87.60-101.01%), AUC0-t (89.98-104.55%) and AUC0-∞ (89.45-104.03%) values for the test and reference products. These values were within the acceptable range of 80.00%-125.00%, proposed by FDA. It was concluded that there was no significant difference between the rate and extent of absorption of the two Tadalafil Tablets.
Introduction
Approved by FDA in 2003 for the treatment of penile erectile dysfunction and in 2009 for the treatment of the pulmonary arterial hypertension, tadalafil is a selective, reversible inhibitor of PDE5 and it is the active compound of Cialis® [1,2].The therapeutic dose of tadalafil for the treatment of penile erectile dysfunction ranges from 2.5 to20mg, daily. While in the treatment of pulmonary arterial hypertension the doses of tadalafil usually at 40mg. It has been confirmed that tadalafil could improve exercise capacity and tolerance, pulmonary hemodynamic and quality of life [3-5].
In many instances, the differences of generic drugs and the original product was correlated successfully to dissimilar drug blood levels caused mainly by impaired absorption. The bioequivalent of two drug products is defined as they are pharmaceutical equivalents or pharmaceutical alternatives and their rates and extents of absorption do not show a significant difference when administered at the same molar dose of the therapeutic moiety under similar experimental conditions [6]. The purpose of this study is to evaluate bioequivalence of domestically produced Tadalafil Tablets and Cialis® from Lilly.
Method Validation
Figure 1: Chemical structures of tadalafil and tadalafil-d3.

Figure 2: Representative chromatograms of blank human plasma matrix (A) and LLOQ of tadalafil-d3 (B) and Representative Chromatograms of tadalafil (2ng/mL) (C) and Representative chromatograms of samples (D).
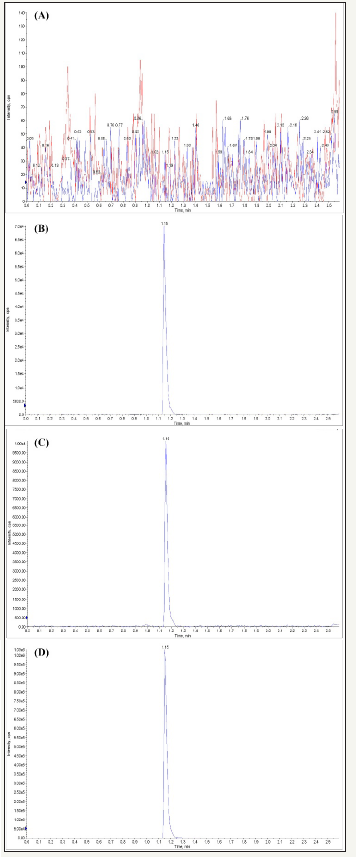
A rapid, selective, sensitive, and reliable UPLC-MS/MS based method for the determination of tadalafil in human plasma was developed and validated to support the bioequivalence trial. Tadalafil was extracted from human plasma by protein precipitation with acetonitrile. In this paper; tadalafil-d3 was used as the internal standard of tadalafil Figure 1. A good chromatographic separation of tadalafil was achieved on a Shim-pack Giss C18 column (2.1×50mm, 1.9μm), with the analytical time 2.6min for each sample Figure 2. The linearity range of the assay was from 2 to 500ng/ml. The intra-day precision and inter-day precision ranged from 3.19 to 6.08%, and the accuracy (RE) was within±11.0%. The extraction recoveries of tadalafil from human plasma were 97.6-102%, and the overall CV calculated for the concentration of matrix effects was 1.56-1.92%, respectively.
Bioequivalence Trial
The study was an open, randomized, two-period, two-group crossover trial, with two weeks washout interval. During the first period, the group A received a single 20mg dose of Cialis® (reference product), while the group B received a single 20mg dose of test product. At the second period, the procedure was repeated on the groups in reverse. The study was performed according to the guideline of Good Clinical Practice. The protocol of this study was approved by Ethical Committee of Nanjing First Hospital. The tablets were given to the volunteers in the morning, after an overnight fast, with 240ml of water. Volunteers did not ingest any alcoholic drink, coffee or other xanthine-containing drinks during the trial. Furthermore, they did not take any other drug, 3 week before the study and during its execution. Blood samples were collected at 0 (pre-dose) and at 0.25, 0.5, 1.0, 1.5, 2.0, 2.5, 3, 3.5, 4.0, 5.0, 6.0, 8.0, 12, 24, 36, 48, and 96h post-dose. The samples were centrifuged and the plasma was stored at -70 °C until quantification
Quantification of Plasma Samples
Calibration standards of 2, 5, 10, 20, 50, 100, 200 and 500ng/ml and quality-control samples of 6, 60 and 400ng/ml were prepared by spiking blank human plasma with standard solutions of tadalafil of the samples, 11.11% were reanalyzed (116 out of 1044). Incurred sample reanalysis performance was 98.2%. The ISR result meets the acceptance criteria.
Bioequivalence Evaluation Results
Average concentration versus time curves after given reference (Cialis® 20mg, Lilly) and test (20mg) products to 29 healthy volunteers is shown in Figure 3. The average values of pharmacokinetic parameters after given reference (Cialis® 20mg, Lilly) and test (20mg) products to 29 healthy volunteers is shown in Table 1. The 90% confidence intervals (90% C.I.) for the ratio of Cmax, AUC0-t and AUC0-∞ values for the test and reference products were calculated by Phoenix Win Nonlin 8.0, is shown in Table 2.
Figure 3: Average plasma concentrations of tadalafil after given reference (Cialis® 20mg, Lilly) and test (20mg) products to 29 Chinese healthy volunteers. Bars indicate standard deviations (upper bars for reference and lower bars for test).
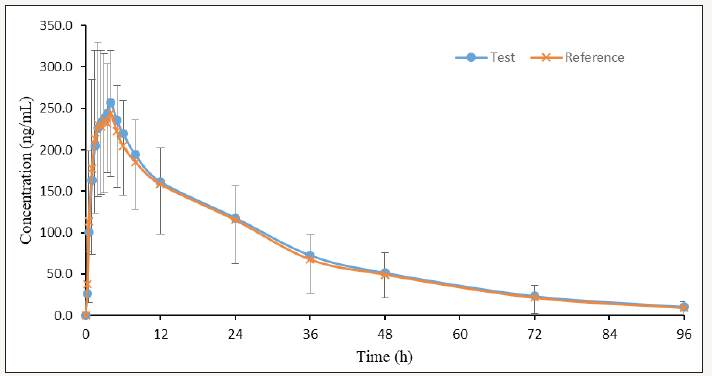
Table 1: Pharmacokinetic parameters after given reference (Cialis® 20mg, Lilly) and test (20mg) products to 29 Chinese healthy volunteers.
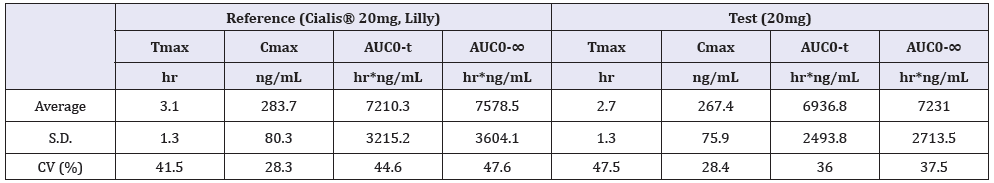
Table 2: Bioequivalence evaluation of two Tadalafil Tablets in healthy Chinese volunteers (n=29).
Conclusion
The 90% confidence intervals for Cmax (87.60-101.01%), AUC0-t (89.98-104.55%) and AUC0-∞ (89.45-104.03%), are within the 80-125% interval proposed by regulatory agencies (FDA). It was concluded that the two Tadalafil Tablets are bioequivalent in their rate and extent of absorption. Thus, two Tadalafil Tablets can be used interchangeably, without any prejudice of therapeutic effect.
Acknowledgement
This work was supported by Natural Science Foundation of China (No. 81503148, No. 81473272).
References
- Bansal UK, Jones C, Fuller TW, Wessel C, Jackman SV (2018) The Efficacy of tadalafil daily vs. on demand in the treatment of erectile dysfunction: a systematic review and meta-analysis. Urology 112: 6-11.
- Abu El-Enin MA, Al-Ghaffar Hammouda Mel S, El-Sherbiny DT, El Wasseef DR, El Ashry SM (2016) Validated spectrofluorimetric method for determination of two phosphodiesterase inhibitors tadalafil and vardenafil in pharmaceutical preparations and spiked human plasma. Luminescence 31(1): 173-178.
- Forgue ST, Patterson BE, Bedding AW, Payne CD, Phillips DL, et al. (2006) Tadalafil pharmacokinetics in healthy subjects. Br J Clin Pharmacol 61(3): 280-288.
- Bhadoriya, Dasandi B, Parmar D, Shah PA, Shrivastav PS (2018) Quantitation of tadalafil in human plasma using a sensitive and rapid LCMS/ MS method for a bioequivalence study. Journal of Pharmaceutical Analysis.
- Proenca P, Mustra C, Marcos M, Franco JM, Corte-Real F, et al. (2013) Validated UPLC-MS/MS assay for the determination of synthetic phosphodiesterase type-5 inhibitors in postmortem blood samples. J Forensic Leg Med 20(6): 655-658.
- Chow SC, Liu J (2008) Design and Analysis of Bioavailability and Bioequivalence Studies, (3rd edn), CRC Press, Florida, USA.
© 2018 Xijng Chen. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






