- Submissions

Full Text
Advancements in Bioequivalence & Bioavailability
Isolation and Evaluation of Antibiotic Resistivity Pattern of Faecal Coliforms Bacteria Isolated From River Wudil Kano, Nigeria
Ali M1*, Farouk SN2, Abdullahi A Minjibir,3 and Musa Ahmad A Diso4
1 Department of Microbiology, Kano University of Science and Technology Wudil, Nigeria
2 Department of Biological Sciences, Bayero University Kano, Nigeria
3 Department of Pharmaceutical Technology, School of Technology Kano, Nigeria
4 Department of Science Laboratory Technology, School of Technology Kano, Nigeria
*Corresponding author: Muhammad Ali, Department of Microbiology, Kano University of Science and Technology Wudil, Nigeria, Tel: 07032967252;Email: alimuhd4real@gmail.com
Submission: March 13, 2018; Published: May 15, 2018

ISSN 2640-9275 Volume1 Issue2
Abstract
Water borne diseases has been a major public health concern in Nigeria. This study was conducted to identify and evaluate the antibiotic resistivity pattern of faecal coliforms bacteria isolated from River Wudil in Wudil Local Government Area of Kano State, Nigeria. Four (4) water samples from different part of the river were collected for the study. The bacteriological and physicochemical analyses performed were in accordance with standard procedures. Antibiotic susceptibility patterns of the bacterial isolates were determined using modified Kirby Bauer method. The result of the study revealed that the physicochemical properties of the river are within the WHO and NIS standard. The result of microbiological analysis of the River showed that Escherichia coli, Salmonella typhi, Proteus mirabilis, Enterobacter sp. and Klebsiella pneumoneae were identified E. coli, S. typhi and Klebsiella were present in all the four water samples examined. The result of this study also showed that Gentamicin, Ciprofloxacin, Erythromycin and Levifloxacin were 80-90% effective against the tested bacteria isolates. On the other hand, the isolates were 100% resistant to Ampicillin, 80% resistant to Rifampicin and 60% resistant to Chloramphenicol. Statistical analysis of the results showed significant different on the susceptibility of the isolates to the antibiotics used at p< 0.05. The use of the river water for drinking may be hazardous. The study therefore, stresses on the need to control the faecal pollution of the water before use.
keywordsAntibiotics; Microbiological; Physicochemical; River water; Wudil
Introduction
Water is one of the most essential natural resources needed by every living thing. Whether it is used for drinking, bathing, food production or recreational purposes, portable and accessible water supply is crucial for public health [1]. Water is the commonest solvent for many substances and it rarely occurs in its pure nature. Water can be obtained from a number of sources, among which are streams, lakes, rivers, ponds, rain, springs and wells [2]. Drinking water has always been a major issue in many countries, especially in developing countries like Nigeria. In Nigeria, majority of the rural populace do not have access to potable water and therefore, depend on well, stream and river water for domestic use [3]. Contaminated water sources are vehicles for the transmission of waterborne diseases such as cholera, shigellosis, and Campylo bacteriosis [4]. The World Health Organization (WHO) estimated that about 1.1 billion people globally drink unsafe water and the vast majority of diarrheal diseases in the world (88%) are attributable to unsafe water, sanitation and hygiene. Approximately 3.1% of annual deaths (1.7 million) and 3.7% of the annual health burden worldwide (54.2 million) are attributable to unsafe water, sanitation and hygiene [5].
Untreated water sources such as surface waters (streams, rivers, lakes, etc.) or unprotected open wells are vehicles for waterborne bacterial diseases such as cholera and typhoid fevers [4]. Untreated waters may also play a role in the transmission of water washed viral enteric diseases such as hepatitis, gastroenteritis, as well as an unknown number of ill-defined diseases caused by other enteric viruses. The fecal-oral route is probably the major route for transmission of these bacterial and viral diseases as well as of many parasitic diseases in poor sanitary conditions. An improvement of water quality and water usage for improving sanitary conditions should result in a decrease of waterborne as well as water-washed diseases [4,5].
Major factors affecting microbiological quality of surface waters are discharges from sewage works and runoff from informal settlements. Indicator organisms are commonly used to assess the microbiological quality of surface waters and faecal coliforms (FC) are the most commonly used bacterial indicator of faecal pollution [6]. They are found in water that is contaminated with faecal wastes of human and animal origin. Total coliforms (TC) comprise bacterial species of faecal origin as well as other bacterial groups (e.g. bacteria commonly occurring in soil). The coliforms are indicative of the general hygienic quality of the water and potential risk of infectious diseases from water [6].
Faecal contamination of water is established by the isolation of an organism that occurs only in faeces, never free-living in nature. There are several such organisms like Echerichia coli, Salmonella, Shigella and Streptococcus faecalis. The finding of E. coli or Clostridium perfringens and S. faecalis is sufficient evidence that the water in question is not safe, since enteric pathogens may be presumed present [7]. The World Health Organization and many other authorities continue to support the use of bacterial indicator levels and their isolation as a basis for judging and verifying drinking water quality [8]. The study was therefore aimed to identify and evaluate antibiotic resistant pattern of bacteria isolated from River Wudil in Kano, Nigeria.
Materials and Methods
Study site
Wudil Local Government Area is geographically located in the south-eastern part of Kano state along Maiduguri – Kano way with a distance of about 41 Kilometer from the State capital. It is located at Latitude 110 49’ 0” N and Longitude 80 51’ 0” E. It covers an area of about 362 Km2 of land and population of about 185,189 according to 2006 census [9]. Wudil Local Government shares common boundaries with Warawa (North), Dawakin-kudu (West), Garko (South) and Gaya Local Government (East).
Sample collection
Four (4) water samples from different part of the river were collected at river Wudil in Wudil Local Government Area Kano State, Nigeria. The water samples (250ml) were collected in sterile bottles, transported to the water laboratory Department of Geography, Bayero University Kano for analysis. Microbiological analysis of water samples conducted at Microbiology Laboratory of Kano State University of Science and Technology Wudil, and was conducted as soon as possible after collection to avoid unpredictable changes in the microbial population
Physico-Chemical analysis
The physico-chemical tests included the determination of temperature, turbidity, odour, colour, total solid, total dissolved solid, total suspended solid, PH, conductivity, acidity, total hardness and chloride content using the methods of FAO [10].
Microbiological analysis
Membrane filters (MF) method was used. The method described by Genthe and Franck [11] was followed; 100 ml of each water sample was filtered through sterile membrane which retained the bacteria on its surface. The membrane was removed aseptically and placed on a Nutrient agar (NA) as a basal medium and MacConkey agar as a differential medium to determine coliforms bacteria. All the plates were incubated at 37°C for 24 hrs. Each colony was isolated in a pure form by sub culturing in fresh Mac Conkey agar plates for further studies and identification. Distinctive morphological properties of each pure culture such as colony form, elevation of colony and colony margin were observed. Further microbial identification was based on the methods of Holt et al. [12].
Characterization and identification of bacterial isolates
Presumptive colonies were confirmed by gram staining and biochemical (Indole, Methyl-red, Vougues Proskeaur, Citrate utilization and Oxidase) tests and each plate were graded as positive or negative. Bacteria isolates were identified and characterized according to Bergy’s manual of systemic determinative Bacteriology by Holt et al. [12].
Antibiotic sensitivity test
The bacteria isolates were subjected to antibiotic susceptibility testing using the agar disc diffusion method as described by Bauer et al. [13]. Mueller Hinton agar (MHA) plates were inoculated with overnight culture of each isolate by streak plating. The standard antibiotic sensitivity discs were then aseptically placed at equidistance on the plates and allowed to stand for 1 hour. The plates were then incubated at 37°C for 24 hours. Sensitivity pattern of the isolates to Augmentin (30 μg/disc), Erythromycin (10 μg/disc), Streptomycin (30 μg/disc), Amoxicillin (30 μg/disc), Gentamicin (20 μg/disc), Oxacillin (10 μg/disc), Ofloxacin (30 μg/disc), Neomycin (20 μg/disc), Ciprofloxacin (10 μg/disc) and Septrin (30 μg/disc), produced by Abtek pharmaceutical limited, were determined. Isolates were divided into three groups based on the zone of inhibition produced by the antibiotic disc; susceptible, intermediately susceptible and resistant according to the Clinical and Laboratory Standards Institute (CLSI) guideline; Performance Standards for Antimicrobial Susceptibility Testing [14].
Statistical analysis
The data of average zones of inhibition produced by the isolates against the antibiotics used was analyzed using One-Way ANOVAs using statistical program SPSS 21.0 (Statistical Package for the Social Sciences). Significance level for the differences was set at p< 0.05.
Results
Physicochemical analysis of water
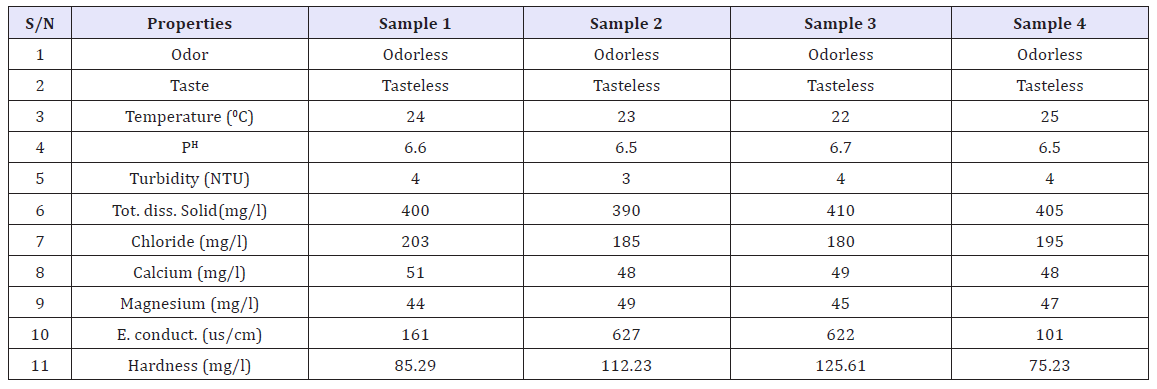
The average physicochemical analysis of the water samples is presented in (Table 1). Both physical and chemical properties of water are presented. All of the samples have turbidity of 4NTU except sample 2, which indicated that the samples have relative turbidity. The PH of the water samples ranges from 6.5-6.7. Both the water samples were colorless and odorless.
Table 1: Physico chemical analysis of water.
Bacteriological analysis of water
The bacterial isolates recovered from water samples are presented in (Table 2). The result showed that Escherichia coli, Salmonella typhi, Proteus mirabilis, Enterobacter sp. and Klebsiella pneumoneae identified. Escherichia coli, Salmonella typhi, and Klebsiella pneumoneae were present in all the water samples examined.
Table 2: Bacteriological analysis of water.
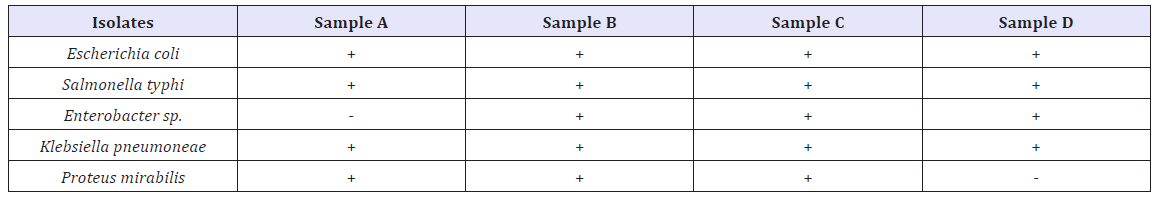
Keywords: + = Present; - = Absent
Antibiotic sensitivity test
The mean zone of inhibition of antibiotic sensitivity disc against the bacterial isolates is presented in (Table 3). Most of the antibiotics are active against the isolates. Enterobacter spp. is susceptible to the entire antibiotic used except Streptomycin and ampicillin. S. typhi is resistant to Norfloxacin, Rifampicin, Ampicillin and Chloramphenicol. E. coli also showed resistivity to Ciprofloxacin Amoxicillin, Rifampicin and Ampicillin. Klebsiella is resistant Amoxicillin, Rifampicin, Ampicillin and Chloramphenicol. Proteus mirabilis is susceptible to the entire antibiotics but resistant to Rifampicin and Ampicillin and intermediate for Norfloxacin, Streptomycin and Erythromycin.
Table 3: Mean zone of inhibition of antibiotics against of bacterial isolates

Keywords: CIP = Ciprofloxacin; NOR = Norfloxacin; GEN = Gentamicin; AMO = Amoxil; STR = Streptomycin; RIF = Rifampicin; ERY = Erythromycin; AMP = Ampicillin; LEV = Levifloxacin; CHL = Chloramphenicol
Susceptibility and resistivity status of the isolates
The sensitivity pattern of the isolates against the antibiotics used is presented in (Table 3). Isolates were divided into three groups based on the zone of inhibition produced by the antibiotic disc; Susceptible S (above 18mm), intermediately susceptible I (11- 17mm) and Resistant R (below 11mm).
Discussion
The Wudil river water was neutral with pH ranges from 6.5- 6.7. Results from this study suggested that the pH values fell within acceptable levels and the values are not toxic to aquatic life according to WHO report. All the PH values of the samples fell within the WHO and NIS permissible limits of 6.50-8.50 and 6.50-7.50 respectively.
The well buffered nature of the river water can be attributed to the fact that, normally, running waters are influenced by the nature of deposits over which they flow [15]. The temperature of the water ranges from 22-250C. The temperature of the river water would not affect its use for domestic and recreational purposes, and the aquatic ecosystem.
Turbidity values ranged from 3-4 NTU. The background levels for turbidity vary from 0–5 NTU [5]. All the samples had the range for turbidity of which also conforms to the NIS and WHO standards. Soil erosion and runoff from the catchments could be the source of turbidity in the river water [16]. The electric conductivity of all the samples ranged from 101-161 μs/cm in all the samples analyzed, all the electrical conductivity values were within the WHO and NIS standards. The conductivity of a river is lowest at the source of its catchments and, as it flows along the course of the river, it leaches ions from the soils and also picks up organic material from biota and its detritus. The average value of typical unpolluted rivers is approximately 350μs/cm [17]. Therefore, the parameter does not give cause for concern and it makes the water suitable for direct domestic use. The fluctuations in electrical conductivity correlated positively with the total dissolved solids (TDS). Suspended solids (SS) and TDS are common indicators of polluted waters. TDS values ranged from 390-410 mgl-1. These values were relatively low compared with WHO guideline value of 1000 mgl-1.
Coliforms are the most frequent bacteria in water responsible for water borne diseases such as cholera, dysentery, diarrhea, typhoid fever which is responsible for mortality across the world especially in Africa [15,17]. All the samples analyzed show positive test for E. coli, Salmonella typhi, Proteus mirabilis, Enterobacter spp. and Klebsiella pneumoneae. This indicates high pollution of the river Wudil. The poor microbiological quality might be due to contamination caused by human activities and livestock [18]. It is a common practice for people living along the river catchment to discharge their domestic and agricultural wastes as well as human wastes into rivers. In addition to using the river as a source of drinking water people use the source for bathing, washing of clothes and for recreational purposes such as swimming [19]. Wild and domestic animals seeking drinking water can also contaminate the water through direct defecation and urination [20]. The result of this study was in conformity with several results of similar findings which showed the presence of coliforms such as E. coli, Salmonella typhi, Proteus mirabilis, Enterobacter spp. and Klebsiella pneumoneae in most water sources [21-23]. Olorode et al. [24], study the physicochemical and microbial analysis of some rivers in Rivers State, Niger-delta in Nigeria, the result of microbial analysis show the presence of bacteria such as Escherichia coli, Campylobacter, Pseudomonas, Salmonell, Vibrio, Proteus, Shigella, Enterococcus. The bacteriological quality of most of the stream waters in the tropics is poor, mainly due to pollution from widespread and indiscriminate human and animal defecation and very poor waste disposal practices [25]. A study on Physico- Chemical and Microbiological Analysis of Well Water Samples in Settlements around Akperan Orshi College of Agriculture, Yandev in Benue State, Nigeria was conducted by Mwekaven et al. [16].
The results of microbiological analysis of the samples indicates that most of the wells were grossly contaminated with bacteria pathogens especially, Escherichia coli (100%), proteus species (47%) and salmonella specie (7%). This result supported the present study.
,p>On the sensitivity pattern of the isolates against the antibiotics used, most of the antibiotics are active against the isolates. Klebsiella was found resistant to 40% of the antibiotics used. It is resistant to Amoxicillin, Rifampicin, Chloramphenicol and Ampicillin. Salmonella typhi showed 50% resistivity, it showed resistance to Rifampicin, Norfloxacin, Levifloxacin, Chloramphenicol and Ampicillin. Escherichia coli were found resistant to Ciprofloxacin, Amoxicillin, Rifampicin, Ampicillin and Chloramphenicol. The result of this study also showed that Gentamicin, Ciprofloxacin, Erythromycin and Levifloxacin were 80-90% effective against the tested bacteria isolates. On the other hand, the isolates were 100% resistant to ampicillin, 80% resistant to Rifampicin and 60% resistant to Chloramphenicol. Similar studies on susceptibility or resistivity of bacterial isolates against antibiotics were reported. A study conducted by Nwadioha et al. [26] also reported that E. coli were highly susceptible ciprofloxacin and amoxicillin/clavulanic acid but were resistant to ampicillin and Chloramphenicol. This finding can be correlated with the present study partially. Sarathbadu et al. [27] in his study reported that all Klebsiella species isolated from urine, pus and sputum were highly susceptible to Ciprofloxacin and Gentamicin. This support the results present study. Anguzu et al. [28] reported that most of the Gram-negative bacteria isolated in their study were resistant to ampicillin; this finding correlate with the present study. The result of this study justified the result of Nnebe agumadu et al. [29] who reported that all Gram negative organisms isolated from otitis media patients were susceptible to ciprofloxacin and Gentamicin but highly resistant to ampicillin. The study showed an alarming resistance of Gram-negative organism to beta lactam antibiotics which is a serious problem that should be looked into. This beta lactam antibiotics resistance could be by vertical as well as horizontal transfer of resistance genes [30].Several factors have been reported to be responsible to antibiotics resistance in bacterial. Some of the reasons includes: Reduced access to target due to slow porin channels; increased antibiotics expulsion due to multiple drug efflux pumps; inactivating enzymes due to β-lactamases, aminoglycoside-modifying enzymes; mutational resistance due to regulatory mutations that increases the expression of intrinsic genes and operons which is variable in certain circumstances [31]. Although the manner of acquisition of resistance may vary among bacterial species, resistance is created by only a few mechanisms: Antibiotic inactivation-direct inactivation of the active antibiotic molecule [32], Target modification-alteration of the sensitivity to the antibiotic by modification of the target [33], Efflux pumps and outer membrane (OM) permeability changesreduction of the concentration of drug without modification of the compound itself [14], Target bypass-some bacteria become refractory to specific antibiotics by bypassing the inactivation of a given enzyme [34].
Conclusion
The result of this study has shown that most of the physicochemical parameters of water in River Wudil were found below the WHO and NIS standards for drinking and Agricultural activities. However, the water was found to be grossly polluted as a result of microbial contamination due to feacal coliforms such as E. coli Salmonella typhi, Proteus mirabilis, Enterobacter spp and Klebsiella pneumoneae. The sensitivity pattern of the isolates against the antibiotics used showed that most of the antibiotics are active against the isolates but with some degree of resistance especially to Ampicillin, Rifampicin and Chloramphenicol. It is recommended that necessary treatment procedures should be applied to raise the quality of the water to the WHO standards for safe drinking. Public health education aimed at improving personal, household and community hygiene is also important.
Acknowledgement
The authors wish to acknowledge the staff of Soil and Water Laboratory, Bayero University Kano. We also like to acknowledge the technical staffs of Microbiology department Kano University of Science and Technology Wudil for their support and use of the department’s laboratory facilities.
References
- Eboh JO, Ogu GI, Idara MU (2017) Micro biological quality of borehole and well water sources in Amai kingdom, ukwuani local government area of delta state, Nigeria. International Journal of Advanced Academic Research Sciences, Technology Engineering 3(7): 1-10.
- Okonko IO, Adejoje OD, Ogunnusi TA, Fajobi E, Shittu OB (2008) Microbiological and physicochemical analysis of different water samples used for domestic purposes in Abeokuta and Ojota, Lagos, Nigeria. African Journal of Bio technology 7(5): 617-621.
- Shittu OB, Olaitan JO, Amusa TS (2008) Physico-chemical and bacteriological analyses of water used for drinking and swimming purposes in Abeokuta, Nigeria. African Journal of Biomedical Research 11(2008): 285-290.
- Burgess JE, Pletschke BI (2010) Microbiological water quality assessment (catchment to tap). water and health 2(1): 1-10.
- World Health Organisation (2004) Guidelines for drinking-water quality 2004 (3rd edn), World Health Organization, Geneva, Switzerland.
- Antony RM, Renuga FB (2012) Microbiological analysis of drinking water quality of ananthanar channel of kanyakumari district, Tamil Nadu, India. Ambi Agua Taubaté 7(2): 42-48.
- Sinton LW, Finlay RK, Hannah DL (1998) Distinguishing human from animal faecal contamination in water a review. New Zealand journal of marine & fresh water research 2(32): 323-348.
- . Mark DS, Frederic KP (2002) Evaluation of the H2 S method for detection of faecal contamination of drinking water. World Health Organization 23(2): 1-10.
- National Population Commission (2006) National population census result, 2006 Abuja, Nigeria.
- Food and Agriculture Organisation (1997) Chemical analysis manual for food and water, (5th edn), FAO, Italy 1(2): 20-26.
- . Genthe B, Franck B (1999) A tool for assessing microbial quality in small community water supplies, WRC report pages to water research commission by division of water, environment and forestry technology, CSIR. Stellenbosch 33(961): 1-99
- . Holt JG, Krieg NR, Senath PHA, Staley JT, Williams ST (1994) Bergey’s manual of determinative bacteriology (9th edn), Williams and Wilkins, Baltimore, USA.
- Bauer AW, Kirby WM, Sherris JC, Turck M (1966) Antibiotic susceptibility testing by a standardized single disk method. Am J Clin Pathol 45(4): 493-496
- Melvin PW (2010) Performance standards for antimicrobial susceptibility testing. CLSI 2(6): 1-10.
- Raji MIO, Ibrahim YKE (2011) Prevalence of water borne infection in northwestern Nigeria: A Retroepective Study. J pub Health Epidimiol 3(8): 382-385.
- . Mwekaven SS, Aorkwagh MT, Gundu EG, Yange T (2017) PhysicoChemical and microbiological analysis of well water samples in settlements around akperan orshi college of agriculture, yandev. IJST 6(1): 1-8
- Adeyinka SY, Wasiu J, Akintayo CO (2014) Review on prevalence of water borne diseases in nigeria. J adv med life science 1(2): 1-3.
- Agbogu VN, Okuofu CA, Smith SI, Ameh JB (2005) Study of the bacteriological and physicochemical indicators of pollution of surface waters in Zaria, Nigeria. African Journal of Biotechnology 5(9): 20-30.
- Olabisi OE, Awomeso AJ, Adebayo OJ (2008) Assessment of bacteria pollution of shallow well water in Abeokuta, southwestern Nigeria. Life Science Journal 5(1): 59-65.
- Ahiarakwem CA, Onyekuru SO (2011) A comparative assessment of the physico-chemical and microbial trends in njaba river, niger delta basin, southeastern Nigeria. Journal of Water Resource and Protection, 3(1): 686-693.
- Ndamatso MM, Idris S Likita, Jimoh MB, Ajai OT, Bala AA (2013) Physicochemical and escherichia coli assessment of selected sachet water produce in some area of minna, niger state Nigeria. Int J Water Res Environ Eng 5(3): 134-140.
- Anake WU, Ehi Eremosole CO, Siyambola TO, Edebor Osoh A, Adeniyi IO (2013) Physicochemical and microbial assessment of different water resources in ota, ogun state, Nigeria. Int J Current Res 5(7): 1797-1801
- Aminu T, Amadi AN (2004) Bacteriological contamination of ground water from zango local government area katsina state northwestern Nigeria. J Geoscience Geomatic 2(5): 186-195.
- Olorode OA, Bamigbola EA, Ogba OM (2015) Comparative studies of some river waters in port harcourt based on their physico - chemical and microbiological analysis, niger delta region of Nigeria. International Journal of Basic and Applied Science 3(3): 29-37.
- Ezeigbo HI (1989) Ground water quality problems in parts of imo state, Nigeria. Nigerian Journal of Mining and Geology 25(5): 1-9.
- Nwadioha SI, Nwokedi EDP, IKeh I, Egesie J, Kashibu E (2010) Antibiotic susceptibility patterns of uropathogenic bacterial isolates from AIDs patients in a Nigeria tertiary hospital. JMMS 1(11): 530- 534.
- Sarath BR, Ramani TV, Bhaskara K, Supriya P (2012) Antibiotic susceptibility patterns of Klebsiella pneumoniae isolated from sputum, urine and pus. J Pharm Bio Sci 1(2): 4-9.
- Anguzu JR, Olila D (2007) Drug Sensitivity Patterns of Bacterial Isolates from Septic Post-Operative Wounds in a Regional Referral Hospital in Uganda. Afr Health Sci 7(3): 148-154.
- Nnebe AU, Okike O, Orji I (2011) Childhood suppurative otitis media in Abakaliki: isolated microbes and in-vitro antibiotic sensitivity patterns. Nig J Clin Prat 14(2): 159-162.
- Romanus II, Emmanuel NA, Ngozi AF, Onyinyechi UE, Chidiebube NA, et al. (2013) Antibiotic susceptibility patterns of bacterial isolates from hospitalized patients in Abakaliki. International Research Journal of Basic and Clinical Studies 1(4): 46-52.
- Nikaido H (2003) Molecular basis of bacterial outer membrane permeability revisited. Microbiol Mol Biol Rev 67(4): 593-656.
- Wright GD (2005) Bacterial resistance to antibiotics: enzymatic degradation and modification. Adv Drug Deliv Rev 57(10): 1451-1470
- Lambert PA (2005) Bacterial resistance to antibiotics: modified target sites. Adv Drug Deliv Rev 57: 1471-1485
- Kumar A, Schweizer HP (2005) Bacterial resistance to antibiotics: active efflux and reduced uptake. Adv Drug Deliv Rev 57: 1486-1513.
© 2018 Ali M. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






